Embargo Nature of CuO-PANI Composite Against Corrosion of Mild Steel in Low pH Medium
Article information
Abstract
Incorporation of CuO nanoparticles during the polymerization of aniline in the presence of ammonium peroxydisulphate as an oxidizing agent and sodium salt of dodecylbenzene sulphonic acid as dopant as well as surfactant yielded water soluble CuO-PANI composite. Comparison of recorded spectra like FTIR, XRD and SEM with reported one confirm the formation of the composite. Analysis by gravimetric method exposes that the synthesized composite is having resistivity against corrosion, with slight variation in efficiency on extending the time duration up to eight hours in strong acidic condition. OCP measurement, potentiodynamic polarization and EIS studies also confirms the suppression ability of composite against corrosion. Riskless working environment could be provided by the synthesized composite during industrial cleaning process.
1. Introduction
Industries using metal as machinery parts face the problem of corrosion due to the working environment adopted either inside or outside the instrument. Shortfall in economic profit and loss in energy are the important causes of corrosion [1]. Low pH solutions are employed as eviscerating agent in industrial cleaning process [2]. Unexpected electrical, mechanical and chemical properties of metal nanoparticles attracted the researchers for more than a decade [3]. Most of the transition metal oxides are employed as semiconductor in numerous technical sites such as solar energy conversion and magnetic storage devices. Among all transition metal oxides there is special attention for copper oxide nanoparticles due to their efficacy as nanofluids in heat transfer execution [4]. CuO nanoparticles are used as gas sensor [5], Catalyst [6], photocells [7] and p-type semiconductors [8]. Based on the applications, required size of CuO-nanoparticles were prepared by solid state technique [9], sonochemical [10] and hydrothermal method [11]. CuO-nanofluid was prepared by different method [12]. Core/shell inorganic nanoparticle polymer exhibits dual physical properties of the core and shell substance [13]. Polypyrrole composites with nanoparticles of SnO, CeO2, SiO2 and Fe2O3 prepared by electrochemical process showed dual character [14–17].
For few decades, conducting polymeric substances have been investigated as resources for corrosion prevention. One such polymer is polyaniline[PANI]. Adsorption of PANI on metal surface reduces metal loss to a considerable amount [18]. The conducting nature of CuO as evidenced from the literature discussed above encouraged to assimilate it with PANI to explore it adroitness against corrosion inhibition. The present paper deals with the preparation of water soluble CuO-PANI composite and its proficiency against corrosion of mild steel in low pH environment.
2. Experimental
2.1 Materials
Cupric sulfate pentahydrate, sodium hydroxide, sodium salt of dodecyl benzene sulfonic acid, ammonium peroxydisulphate and HCl of analytical grade were purchased from Merck industry and used as such. Aniline of analytical grade was purified by distillation with pinch of zinc dust before polymerization.
2.2 Instrumentation
Perkin-Elmer 337 spectrometer was used to record the FTIR in the frequency range of 450-4000 cm−1 using KBr pellets. XRD spectra were recorded using Rigaku Maniflex X-ray diffractometer. Structural morphology of the samples were characterized by a JSM-6390 scanning electron microscope. ECLAB 10.37 model was adopted to carry out electrochemical impedance and potentiodynamic polarization studies. The open circuit potential measurements were made using CHI electrochemical analyzer instrument 1200B model.
2.3 Synthesis of CuO-PANI composite
The synthesis of CuO nanoparticle was carried out by adapting little modification to the procedure reported in the literature [19,20]. To 0.05M solution of copper sulphate, desired amount of 6M solution of NaOH was added in drops with stirring. The mixture was stirred vigorously until the color of the reaction mixture turns to black. The precipitated black colored CuO particles were filtered, washed with distilled water, acetone and then dried in hot air oven at 80°C. Modified form of in-situ chemical oxidative polymerization method adopted in our earlier reports [21,22] was used to synthesis CuO-PANI composite. About 0.7954 g/ 10 ml (0.1 M) of CuO was dispersed in required amount of 0.1M DBSA solution using oscillation frequency of 42 kHz for forty five minutes in a sonicator. In another beaker, a mixture containing 25 ml of 0.5M aniline and 75 ml of 0.5M hydrochloric acid was kept under stirring for one hour. Above two solutions were mixed together for polymerization at 0–5°C, along with the drop-wise addition of 50 ml (0.1M) ammonium peroxydisulfate with constant stirring for six hours. The green paste obtained was dried in air for two days and then kept at 70°C for eight hours in an oven.
3. Results and Discussion
3.1 Characterization of CuO-PANI composite
3.1.1 FTIR Spectral analysis
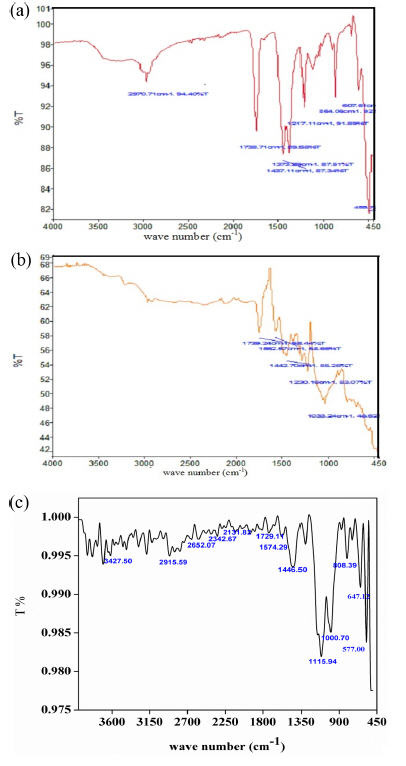
Earlier workers assigned the absorption bands appeared around 3100-2970 cm−1, 1738 cm−1, 1473 cm−1, 1373 cm−1 and 1217 cm−1 [Fig. 1(a)] to the stretching in water molecules associated with CuO, C=O stretching vibration, C-H bending, CuO wagging vibration and C-H wagging vibration respectively. The bands appeared between 607 and 485 cm−1 where allocated to Cu-O stretching modes [20,23–25].
The absorption bands of PANI [Fig. 1(b)] at 1562 and 1443 cm−1 are ascribable to stretching vibration of quinoid and benzenoid ring. The peak observed at 1739 cm−1, is due to N-H bending. The peaks at 1230 and 1033 cm−1 are owing to C-N stretching modes. The characteristic peak around 3400 cm−1 is attributed to N-H stretching of secondary amine [26,27].
The spectrum of PANI-CuO composite shown in Fig. 1(c) exhibit the characteristic peaks of CuO and PANI [20]. The peaks of PANI appeared with slight shift in wave number [3400 cm−1→3428 cm−1, 1739 cm−1 → 1729 cm−1, 1562 cm−1 → 1574 cm−1, 1443.57 cm−1 → 1446.50 cm−1, 1033 cm−1 → 000.70 cm−1]. Similarly the peaks of CuO also exposed with minimal shift in wave number [2970 cm−1 →2915 cm−1, 1738 cm−1 → 729 cm− 1, 1443.70 cm− 1→1446.10 cm− 1, 1217.10 cm−1→1115.9 cm−1, 864 cm−1→806 cm−1, 607 cm−1→647 cm−1, 488 cm−1→577 cm−1]. Above observation indicates that the PANI has been adsorbed over the surface of CuO.
3.1.2 XRD spectral analysis
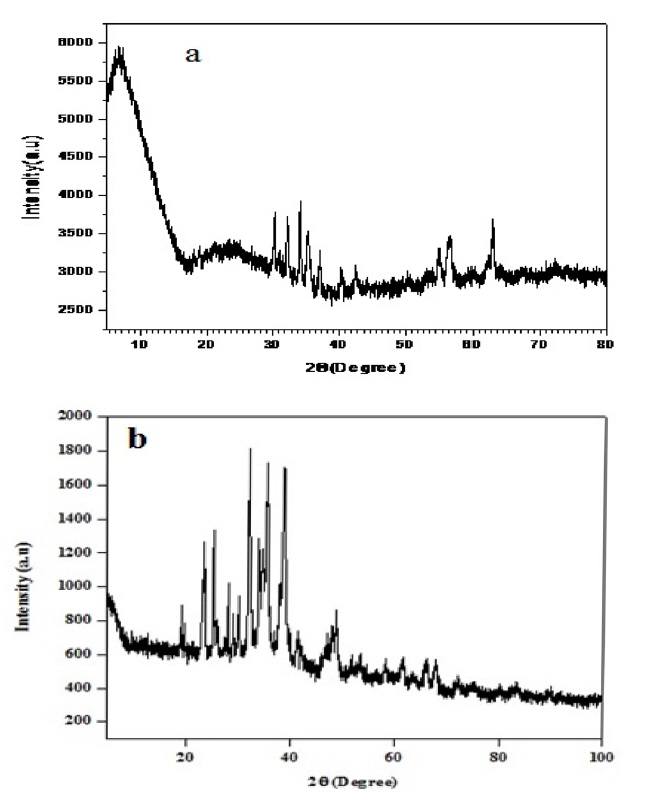
The broad peaks of pure PANI observed [Fig. 2(a)] around 2θ=10.3°,19.5° and 23.5° values were assigned for the periodicity parallel and perpendicular to the polymer chain[28,29]. The XRD of CuO-PANI [Fig. 2(b)] exhibit the characteristic peaks of PANI with slight shift in 2θ value about 22°. The peaks earmarked around 2θ values 32°, 36°, 45° and 48.81° responsible for CuO (110), (111), (112) and (11-2) monoclinic phase, are also appeared with little blue shift [30,31] in the XRD of CuO-PANI. This reveals that the PANI has coated over the surface of CuO nanoparticles.
3.1.3 SEM spectral analysis
The surface morphology of pure copper oxide and CuO-PANI composite are given in Fig. 3(a) & (b). The SEM of CuO shows a uniform flakes structure with diameter starting from 180 nm to 270 nm. The composite SEM appears like cluster due to dispersion of CuO particles in PANI matrix [32] with increase in diameter ranging from 630 nm to 910 nm. This exposes that CuO particles have been covered with the layer of PANI [33,34].
3.2 Preparation of Electrode material
The composition of mild steel coupons used as working electrode contains C: 0.21%, Si: 0.035%, Mn: 0.25%, P: 0.082% and remaining part as iron. They were cut into pieces of size 4 cm × 2 cm × 0.2 cm and were polished with different grinding papers, having grit up to 1200, made of silicon carbide. The abraded specimens were washed with DD water, acetone and then dried in desiccators. Inhibition efficiency measurements were performed with newly polished coupons.
3.3 Preparation of Dissolver solution
The dissolver, one molar and two molar sulphuric acid solutions were prepared by diluting analytical grade sulphuric acid. Desired amount of composites were added to the dissolver to prepare test solutions having concentration of 250 ppm −1000 ppm.
3.4 Assessment of inhibition property
3.4.1 Estimation of corrosion by weight loss measurement
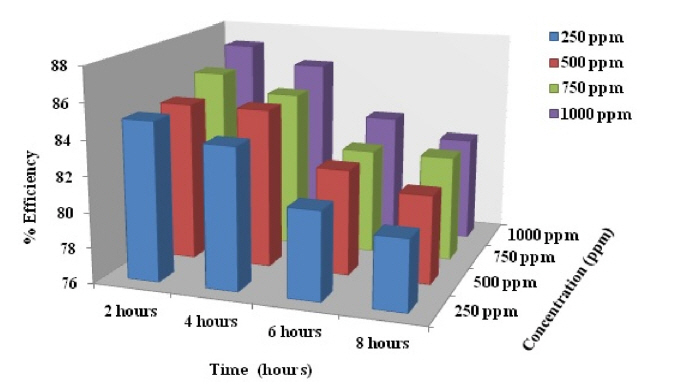
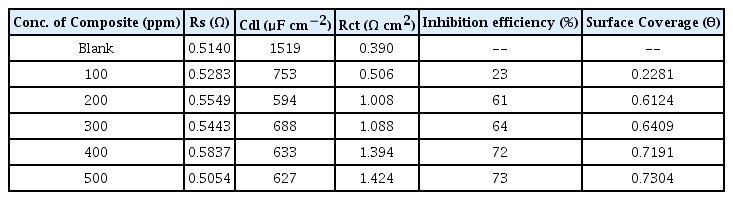
In general, evaluation of corrosion is monitored by assessing loss in weight of the electrode after keeping it in a dissolutive for required time. Pre-weighed electrodes were fully immersed in 250 ml blank and test solutions, having one molar and two molar concentration, for eight hours. The specimens were retrieved at two hours interval, washed with distilled water, acetone, dried and re-weighed. Surface coverage and inhibition efficiency calculations were adopted using the formula reported earlier [35]. The calculated values of percentage inhibition efficiency and surface coverage area are provided in Tables 1 and 2.
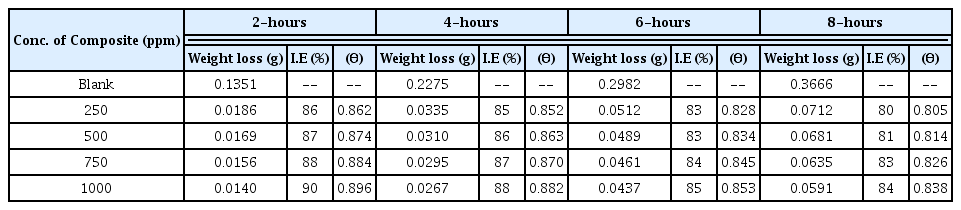
Inhibition efficiency and surface coverage values obtained from the weight loss measurement in 1M blank and test solutions.
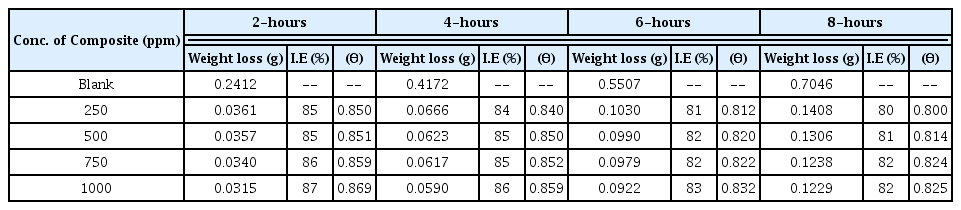
Inhibition efficiency and surface coverage values obtained from the weight loss measurement in 2M blank and test solutions.
Scrutiny of the Figs. 4 and 5 and also the data’s given in the Tables 1 and 2 exposes that there is no appreciable changes in inhibition efficiency and surface coverage ability of the CuO-PANI composite after 250 ppm concentration. This is contrary to our previous reports[21,22] and also exposes that CuO-PANI composite is more efficient than the previously reported composites.
Minimum concentration of the material of present interest is capable of giving better protection even up to eight hours duration. High performing ability might be due to the positive reduction potential value of copper than the other metals used in our previous investigations.
3.4.2. Open Circuit Potential Measurement
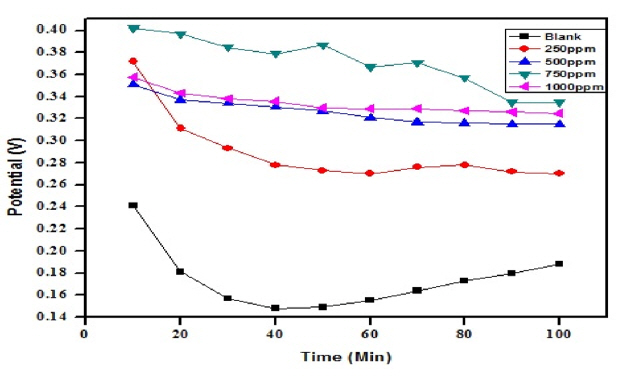
CHI-Electrochemical instrument model 1200B was used to record the OCP values up to two hours. Three electrodes compartment cell having 1 cm2 area of metal under study as working electrode, standard platinum electrode as counter electrode and saturated calomel electrode as reference electrode was used in the analyzer. Fig. 6 and 7 represents the results obtained on OCP measurement in 1M and 2M H2SO4 dissolver with various concentrations of inhibitor ranging from 250 ppm to 1000 ppm.
Earlier reports [26,36] suggest that shift in OCP values towards positive potential side than it was observed for blank solutions could imply the ability of the inhibitor. Addition of CuO-PANI composite to the dissolver shift the OCP values as expected and also only slight changes are noticed with increase in time. This hinted that prepared composite could act as an inhibitor in aggressive conditions.
3.4.3 Electrochemical measurements
The electrochemical measurements like potentio-dynamic polaization and electrochemical impedance were obtained from conventional three electrodes cell assembled to Potentiostat /Galvanostat EC-LAB Analyzer 10.37 model. The working electrode was mild steel ASTM 415 sample with 1 cm2 area and the rest of the area was fully covered with araldite epoxy resin. The counter electrode was platinum electrode and the reference electrode was saturated calomel electrode. Potentiodynamic polarization studies were perceived from −200 to +200 mV with scan rate of 0.5 mV s−1. For Electrochemical impedance measurement 100 kHz – 10 mHz was used as the frequency range with amplitude of 10 mV AC sine wave.
3.4.4 Potentiodynamic polarization meassurements
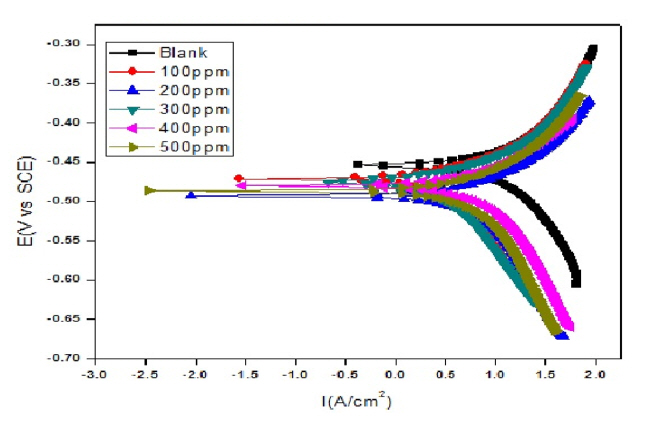
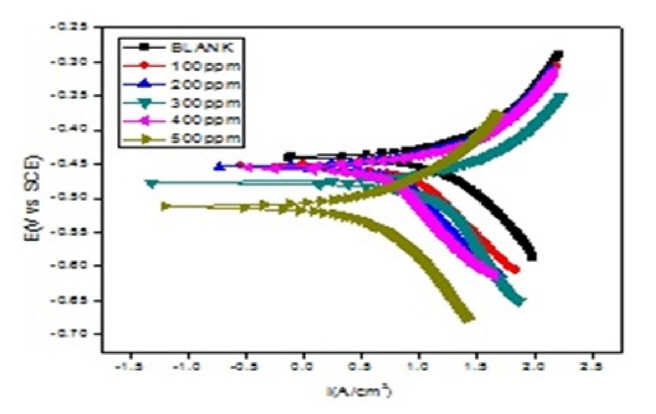
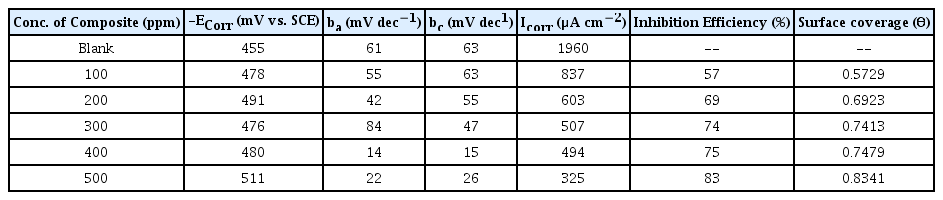
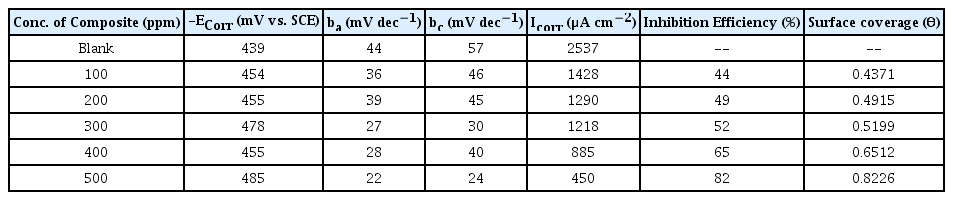
Fig. 8 and 9 reflects Tafel plots detected for blank and test solutions with various quantity of CuO-PANI composite. Various parameters representing the corrosion potential, corrosion current, inhibition efficiency anodic and cathodic slopes are depicted in Tables 3 and 4. Formula disclosed elsewhere [37] was amused to calculate the efficacy of CuO-PANI composite against corrosion.
The kinetic parameters data provided in the Tables 3 and 4 exposes that corrosion current density decreases steadily with increase in addition of composite. Though the corrosion current increases with increase in concentration of dissolver, the addition of inhibitor reduces the corrosion current. Available literature [38] suggested minimal changes on the values of Ecorr, ba and bc for mixed type inhibitors with varied concentration of inhibiting materials. Similar trend is followed by CuO-PANI composite.
3.4.5 Electrochemical impedance measurements
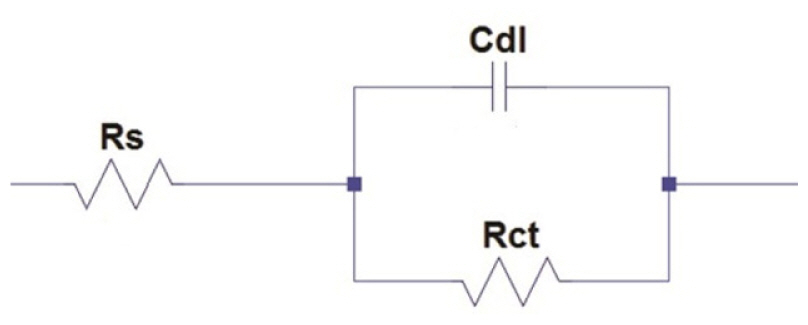
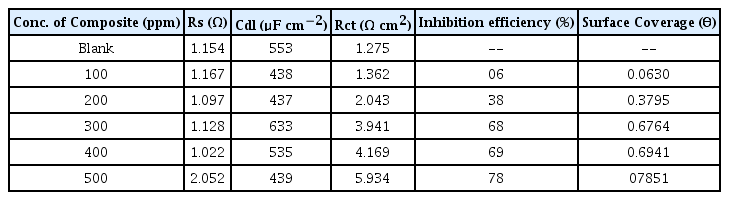
The Impedance parameters such as charge transfer resistance(Rct) and double layer capactiance (Cdl) were determined using the following equivalent circuit.
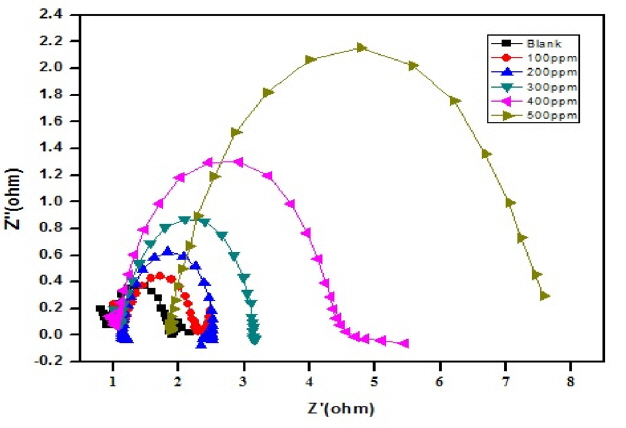
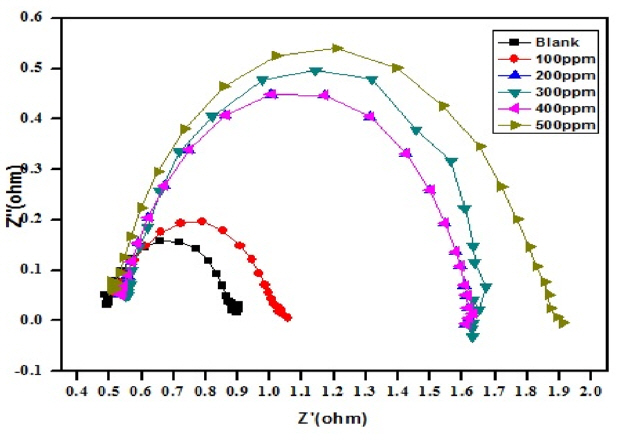
The diameter of the Nyquist plot [39] is directly related to the resistance to corrosion current. Increase in loop size implies that less flow of corrosion current in corrosive environment. Semicircle image of Nyquist plot poses out single charge transfer process [39]. Exact semicircle formation of Nyquist representation indicates the resistivity against corrosion and also reflect single charge transfer process in our studies. The increase in size of capacitive loop with increase in inhibitor concentration (Fig. 10 and 11) discloses that the composite get adsorbed over the metal surface and prevent the flow of corrosion current and thereby act as a good inhibitor in very low pH environment. The reported relationship [37] is used to calculate the IE values.
Formation of protective layer at the electrolyte/ metal interface was reported [40] for increase in Rct values and increase in electrical double layer thickness was assigned to decrease in Cdl values [41]. The behavior of the CuO-PANI composite is indistinguishable from the above reports.
4. Conclusions
The Polymer coated metal oxide composite soluble in aqueous medium is synthesized successfully by modifying the in-situ oxidative polymerization method. It manifest good protection efficacy up to 87% even in strong 2M acidic condition in weight loss studies and is also stable for 480 minutes under corrosive environment. Increasing trend in protection efficiency with respect to the concentration of composite is noticed in OCP measurements and electrochemical studies. Results imply that universal solvent soluble material can act as better shielding agent in maintenance related work.
Acknowledgement
The authors acknowledge the support from Dr.B.V., Dean for Research, SRM group of institutions, Dr. C. Jayaprabha, Associate Professor of Chemistry, University college of Engineering (Anna University) Dindigul-624622 and the Management of Rajalakshmi Engineering college, Chennai-600 105 for the electrochemical analysis of samples on free of cost. This research did not receive any specific grant from funding agencies in the public, commercial or not for profit sectors.
Notes
Conflict of Interest
The authors declare that there is no conflict of interests regarding the publication of the article.