Contribution of Carbon Dot Nanoparticles in Electrocatalysis: Development in Energy Conversion Process
Article information
Abstract
Modern electrochemical energy devices involve generation and reduction of fuel gases through electrochemical reactions of water splitting, alcohol oxidation, oxygen reduction, etc. Initially, these processes were executed in the presence of noble metal-based catalyst that showed low overpotential and high current density. However, its high cost, unavailability, corrosion and related toxicity limited its application. The search for alternative with high stability, durability, and efficiency led scientists towards carbon nanoparticles supported catalysts which has high surface area, good electrical conductivity, tunable morphology, low cost, ease of synthesis and stability. Carbon nanoparticles are classified into two groups based on morphology, one and zero dimensional particles. Carbon nanoparticles at zero dimension, denoted as carbon dots, are less used carbon support compared to other forms. However, recently carbon dots with improved electronic properties have become popular as catalyst as well as catalyst support. This review focused on the recent advances in electrocatalytic activities of carbon dots. The mechanisms of common electrocatalytic reactions and the role of the catalysts are also discussed. The review also proposed future developments and other research directions to overcome current limitations.
1. Introduction
The rapid increase in worldwide energy consumption has led to detrimental impact on the eco-system and economy, and therefore the need for exploration of renewable energy sources. The alternative energy storage and conversion technologies are based on supercapacitors, lithium/sodium ion batteries, metal-air batteries, fuel cells and electrocatalytic water splitting [1]. Electrochemical conversion plays a significant role in energy conversion for production of sustainable fuels and chemicals [2]. Electrochemical devices for the energy conversion and storage are dependent on the constituent material, hence, discovery and modification of suitable materials have been prime concern for the scientists. Rapid advance in nanotechnology provided better options as nanoparticles have high surface to volume ratio and tunable morphology that apparently improves the efficiency for electrochemical processes. Metal nanoparticles have been widely used for electrocatalysis; however, they are expensive and not readily available. The high electron transfer capability, nontoxicity, low cost, simple synthetic route made the carbon nanoparticles an efficient alternative material. Carbon nanoparticles and carbon supported metal nanoparticles have been experimented widely to accomplish a sustainable catalyst. As the electrocatalysts are material dependent, the morphology and dimensionality of the materials are also important.
Carbon nanoparticles act significantly at two (2-D), one (1-D) and zero (0-D) dimensional morphologies. However, the high electropotential of the 0-D particles (carbon dots) have grown intense interest for the scientists. Carbon dots (CD) are fascinating class of recently discovered nanocarbons that comprise discrete, quasi-spherical nanoparticles within the dimension of 10 nm [3]. CD particles were first discovered in 2004 by Xu et al. [4] during electrophoretic analysis and purification of single walled carbon nanotubes. Later in 2006, Sun et al. [5] showed that surface passivation introduced an intriguing fluorescence of the CD particles. Since their discovery, the research on CD particles have flourished through modification of their optoelectronic properties. CD particles possess tunable emission and wide excitation spectra that have facilitated their use in wide range fluorescence imaging [6]. Such intriguing fluorescence of CD particles originate from quantum confinement, radiative recombination, surface passivation, surface defects, and doping of hetero atoms [7,8]. The CD particles are often preferred over traditional organic dyes, metal cluster, fluorescent metal nanoparticles and metallic quantum dots due to their high quantum yield, unique optoelectronic property, photo-resistivity, water solubility, and low toxicity [8]. The adverse chemical and optical properties of CD come from the surface functional groups and the surface defects. For example, CD particles with hydroxyl and carboxylic acid functional groups show excellent solubility properties, although, they have low electron transfer ability due to dislocation of electron density around unsaturated core.
The radiative recombination and electron hole localization is modified in the presence of doped hetero atoms. The interaction within carbon core and surface functional groups facilitate the electron flow within the carbon skeleton. The surface defects and edges provide high surface area. All these factors highly influence optical properties of CD particles. The property and morphology of CQDs highly depend on their synthetic route [3,8]. CD particles have already demonstrated their viability various applications such as sensing, imaging, photovoltaics, environmental remediations, catalysis, electrochemical analysis, etc. as they suppress the limitations of traditional fluorescent probes [3]. Specifically, in the case of electrochemical systems, with their facile charge transfer ability, high surface area, electronic conductivity and chemical inertness, CD particles have prospered as an efficient catalyst.
In this review, we briefly discuss the role of different 0-D CD particles towards common electrocatalytic reactions.
2. Electrocatalysis for energy conversion
The electrochemical reactions governing the energy conversion process are hydrogen evolution reaction (HER), oxygen evolution reaction (OER) and oxygen reduction reaction (ORR) [9]. Water splitting has been employed for hydrogen generation for main component of contemporary clean-energy technologies since its discovery by William Nicolson and Anthony Carlise in 1800 [10]. However, the concept of water splitting was established by Faraday in 1833. Later, Zénobe Gramme and Dmitry Lachinov prescribed low-cost and industrial basis hydrogen synthesis from water splitting, where the generated hydrogen could be used to operate fuel cells. Hydrogen has proven to be an ideal energy carrier, for sustainable energy, due to high energy density and eco-friendly production possibilities [10]. Being a simple proton-electron transfer process without any side reaction, HER is the most studied electrocatalytic reaction [11]. HER measurements are done by a three-electrode system viz. working, reference and counter electrode. The potential at reference electrode is measured with respect to the reference electrode. The HER goes preferably in acidic medium through the following mechanism, [10]
H+ + e− →H* (Volmer step)
H+ + e− + H*→H2 (Heyrovsky step)
H* + H*→H2 (Tafel step)
In alkaline medium the HER might follow the following path [12],
H2O + e− →H* + OH− (Volmer step)
H* + H*→H2 (Tafel step)
These steps rely on the inherent chemical and electronic properties of the electrode surface. In the first step (1) the catalyst-H bond should be strong enough to cleave the water molecule whereas the next step demands easy catalyst-H bond breakage for H2 evolution.
For the Volmer step, paired d-electrons are needed to facilitate electron transfer to water molecule while the next steps would prefer partially filled d-electron to facilitate adsorption of H* moiety [13]. The experimental data about Tafel slope helps to assume the mechanism of the reaction. The HER process needs appropriate catalyst that would lower the activation energy and the overpotential. Platinum (Pt), particularly 111 facets of Pt [11]), is a widely used catalyst for HER due to their long-term durability and near-zero over potential [14]. However, the high price and low abundance compelled scientists to find alternative catalysts for HER [15]. Also, the poisoning of Pt catalyst surface by hydroxyl ion limits its application in alkaline medium [16], hence the need for replacement of Pt catalyst with high stability and causing low overpotential. Carbonaceous nanoparticle at zero dimension could act as efficient electrocatalysis along with metal and nonmetal counterpart. The counter reaction for HER during water splitting is OER. HER occurs at cathode while OER occurs at anode. Even though the theoretical voltage for HER and OER at 298K are 0 V and 1.23 V, respectively, practically, extra electricity is needed to overcome the activation energy barrier [10]. In acidic and alkaline medium, the OER follows the following steps respectively, [12],
Acidic medium mechanism for OER:
H2O ↔ HO* + H+ + e−
HO* ↔ O* + H+ + e−
O* + H2O ↔ HOO* + H+ + e−
HOO* ↔ O2 + H+ + e−
Alkaline medium mechanism for OER:
HO− ↔ HO* + e−
OH− + HO* ↔ O* + H2O + e−
O* + OH− ↔ HOO* + e−
HOO* + OH− ↔ O2 + H2O + e−
OER is a more sluggish process than HER due to the four-electron transfer process. Theoretical thermochemical studies of OER showed that binding energy of surface oxygen played a controlling role [17]. Iridium (Ir) and ruthenium (Ru) catalysts satisfied the condition and provided their low overpotential and Tafel slope [18], but the scarcity and high price limited their application. In this scenario, the 0-D carbon nanoparticle-based catalysts have emerged as their replacement.
ORR is an important cathodic phenomenon that happens in energy-conversion devices like fuel cells and zinc/air battery etc. [19]. Fuel cells were invented by William Grove and Christian Friedrich Schönbein in early 1800s [1]. Fuel cells, operated by taking hydrogen or methanol as fuels and oxygen as redundant at room temperature, are solutions to electric vehicles and portable devices [1].
ORR follows four and two electron transfer mechanisms in both acidic and alkaline medium [12],
Acidic medium mechanism for ORR:
O2 + 4H+ +4e− → 2H2O (Four-electron step)
O2 + 2H+ +2e− → H2O2 (Two-electron step)
H2O2 + 2H+ + 2e− → 2H2O
Alkaline medium mechanism for ORR:
O2 + 2H2O +4e− → 4OH− (Four-electron step)
O2 + H2O +2e− → HO2− + OH− (Two-electron step)
H2O + HO2− + 2e− → 3OH−
ORR is also a sluggish reaction as more than one electron is involved. Pt-based electrocatalysts are widely used for ORR. But the unavailability and high price of Pt compelled scientists to find replacements. The electrode structures for these reactions are shown in Fig. 1.
Apart from the HER, OER and ORR, reduction of CO2, alcohol is also used for fuel cells.
The electrolytes used in the reaction have proved to have specific role in terms of stability of catalyst and electrical conductance, e.g. for HER, the electric conductance is higher in acidic than in alkaline and then phosphate buffer medium. The choice of electrolyte influences the reactivity [10]. In modern days the energy related problems can be solved by replacing the conventional energy sources by artificial energy-conversion devices. The efficiency of the catalysts in these reactions is examined by monitoring the reaction overpotentials, onset potential, Tafel plots, Koutecky-Levich (K-L) plots. Parameters such as low overpotential and Tafel slope values indicate effective catalysis. The catalytic activity of the CD particles is described using these values.
3. 0-D CD nanoparticles for electrocatalysis
The electropotential of carbon nanoparticles varies with dimensionality. The kinetics of electron transfer increases from 0-D dot particles to 2-D sheet particles [20]. In this context the dot particles with length to width ration of 1 have become the center of attention. CD particles are classified as carbon nano dot (CND), graphene quantum dot (GQDs), and polymer dots (PDs), which are all 0-D, however, differ in chemical structure and certain electronic properties [21]. CND particles are spherical in shape and consist sp2/sp3 hybridized carbon core and high oxygen content. CND particles have amorphous nature with no crystal plane, but when crystallinity is present with a lattice parameter of ~0.24 nm, the particles are termed as carbon quantum dots (CQDs) [22]. The photoluminescent origin of CND and CQD particles vary. CND and CQDs are generally synthesized from bio-molecules through top down and bottom up process such as hydrothermal, microwave, laser ablation, chemical reduction, reflux, etc. [8]. CQD particles are commonly synthesized from biomolecules, biomass and organic compounds. Graphene is an allotrope of carbon consisting of honeycomb lattice of carbon atoms with sp2 hybridization [23]. The limitation of zero band gap semiconducting property of graphene can be tuned by change in its electronic property through doping, substrate interaction, change in dimension and morphology [24]. GQD particles overcame the limitations of graphene sheets and are proved to be efficient material for photovoltaics, bio-applications, imaging, devices etc. Figure 2 shows the classification of CD particles [22].
The designing of CD based electrocatalysts needs modification of CD structure [25]. The process includes,
Doping of hetero atoms: Doping of electronegative and electropositive hetero atoms such as, nitrogen (N) and boron (B), breaks the electroneutrality in carbon core by creating electron excess or deficiency and induces facile electronic interaction.
Surface passivation: CD particles contain different surface functional groups that help in facile electron transfer and adhesion of substrate. The electrons donating and accepting functional groups have great role in tuning the property of CD particles.
Such modifications in carbon skeleton of CD particles initiates charge transfer within the carbon skeleton and distort the electronic environment. The energy gap within HOMO and LUMO is modified along with radiative recombination process. The electrocatalytic activity of CD particles are modified through change in the electronic properties based on exposure and generation of active sites. These properties also help CD particles to act as efficient support material towards metal and carbon (graphene, graphitic carbon nitrides etc.) by boosting their surface area to volume ratio and electronic properties [26–31]. Since their first application as electrocatalysts, there has been rapid development in CD based electrocatalysis, specifically, metal free carbon-based catalysts. In the following sections the examples of CD and CD supported catalysts towards electrocatalysis have been discussed.
3.1 CND/CQD nanoparticles
The CND/CQD particles with sp2/sp3 hybridized carbon core act as efficient catalysts. Most CND/CQD electrocatalysts are hetero atom doped and their activities are often better than commercial metallic catalysts. The electronic behavior of N-doped CNDs (termed as SP2) was utilized by Gao et al. for oxygen reduction catalysis (ORR) through a four electron path in 0.1 M KOH medium [32]. The SP2 particles, the conglomeration of nanodots, showed a conductivity of 0.466 S cm−1. A characteristic ORR peak appeared at ca. ~0.22 V in the O2 saturated solution, while a featureless character was observed in the O2-absent medium indicating initialization of ORR by SP2 (Fig. 3).
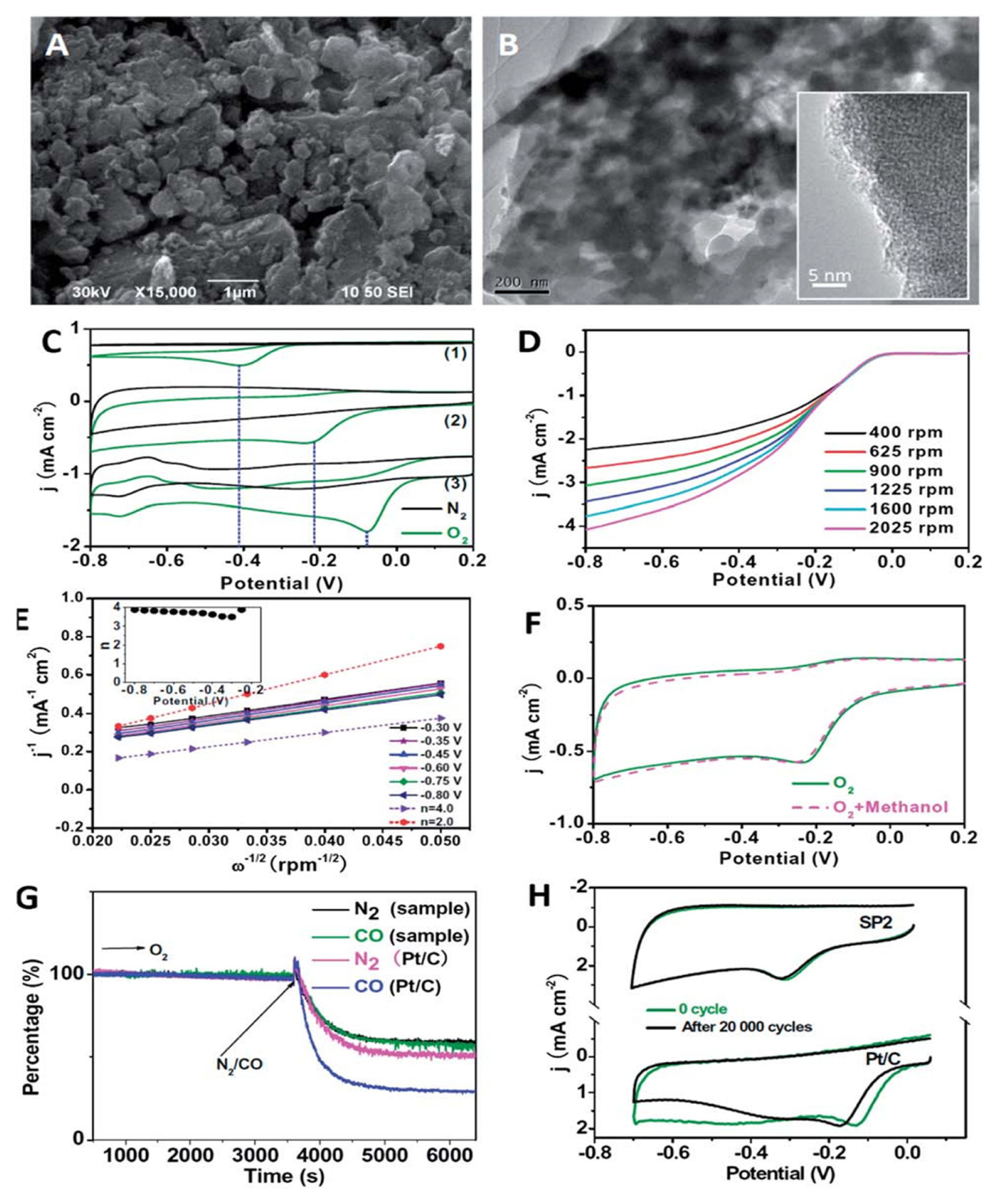
(A and B) SEM and TEM images of SP2, respectively. (C) CVs of the bare glassy carbon (1), SP2 (2), and Pt/C (3) electrodes in N2- and O2-saturated 0.1 M KOH at a scan rate of 10 mV s1. (D) Linear sweep voltammograms (LSVs) for the ORR on the SP2 electrode in an O2-saturated 0.1 M KOH solution at a scan rate of 10 mV s−1 and a series of rotation rates from 400 to 2025 rpm. (E) The K-L plot (J−1 vs. ω1/2) at various potentials based on the corresponding LSV data, and the theoretical K-L plots corresponding to n = 2 and 4, respectively. The inset plot is the n of the ORR on the SP2 electrode. (F) CV curves of SP2 in O2-saturated 0.1 M KOH solution with and without 10 vol% methanol at a scan rate of 10 mV s−1. (G) The percentage of current density (j) vs. time chronoamperometric responses obtained on SP2 and 20 wt% Pt (JM) electrodes at 0.30 V in O2-saturated 0.1 M KOH. The arrow indicates the introduction of N2 or CO into the electrochemical cell. (H) Electrochemical stability of SP2 and 20 wt% Pt/C as determined by continuous CV scanning in O2-saturated 0.1 M KOH. Scan rate: 50 mV s−1. Reprinted with permission from Reference [32]. Copyright © 2014, The Royal Society of Chemistry.
Lie et al. [28] synthesized uniformly N-doped CQDs where they used solvent (N-methyl-2-pyrrolidone and dimethyl-imidazolidinone, with cyclic structures and rich amino groups) as carbon source. They examined its catalytic activity towards ORR in O2-saturated 0.1 M KOH with 3 M CH3OH. The peak potential was −160 mV which remained almost unaltered after 1000 cycles. The presence of pyridine N-atoms caused efficient activity for ORR. Kakaei et al. used CQD to fabricate a gas diffusion electrode for reduction of oxygen in 0.5 M KOH [33]. CQD was pasted on carbon paper as an electrode for ORR in fuel cell. At high current region, the Tafel slope was calculated as 111 mV/decade, indicating the significant activity of the catalyst. The results were attributed to the ability of CQD to enhance the solubility and permeability of oxygen in the reaction layer due to its high porosity. Bhattacharyya et al. [34] synthesized a N-doped CQD from a MgII based anionic MOF, {[Mg3(ndc)2.5(HCO2)2(H2O)]·[NH2Me2]·2H2O·DMF, ncd=1,4-naphthalenedicarboxylate}, to ensure the high porosity and ease of doping. Traces of metallic Co nanoparticles were deliberately incorporated as a part of post synthetic treatment that helped the activity of as-synthesized CQDs for ORR reaction. It was reflected in the Tafel slope values; at higher and lower current density, the slopes were 141 mV dec−1 and 63 mV dec−1, respectively. Atchudan et al. synthesized N-doped CQDs from unripe peach and applied them for electrocatalytic ORR reaction in alkaline medium [35]. They found a prominent O2 reduction peak at 0.45 V vs. RHE for the O2 saturated KOH solution with an onset potential of ORR at 0.94 V. They obtained a two electron pathway of O2 reduction to hydrogen peroxide. Martínez-Periñán et al. [36] reported the effect of surface functional groups on ORR in acidic and alkaline medium. They fabricated amide (CD-CONH2) and carboxylic acid functionalized (CD-COOH) CND particles and examined their electrocatalytic properties towards ORR in acidic and alkaline medium. A suitable onset potential of −0.28 V (vs. SCE) was obtained for CD-COOH modified the screen-printing electrode in alkaline medium. Datta et al. [37] showed a metal free water oxidation by CQD with entrapped 5-hydroxymethyl-2-furaldehyde (5-HMF) at carbon skeleton. They proposed that the 5-HMF molecules in close vicinity to the catalytically active sites containing C=O groups could extract the ·OH/·OOH radicals and increase the in situ H2O concentration to facilitate the O2 evolution. The electrocatalytic behavior of the CQDs was dependent on 5-HMF concentration. The highest concentration of 5-HMF decently exhibited low 0.21(±0.03) V overpotential and generated current density up to 33.6 (±2.3) mA cm−2. Shen et al. [38] prepared a CND from melamine and glutaraldehyde and used them for electrocatalytic ORR in alkaline medium with an onset potential and current density of −0.169 V and 2.69 mA cm−2, respectively. The CND particles were then coupled with silver nanoparticles to achieve superior catalytic activity with onset potential of −0.081 V and a current density of 4.09 mA cm−2 at −0.5 V.
3.2 CND/CQD supported metallic and nonmetallic nanoparticles
In their study, Shen et al. [38] reported that the composite of the as-synthesized N-doped CND with silver nanoparticle (AgNP) provided better activity towards ORR compared to N-CND. The CND possessed both p-and n-type conductivities from the Mott-Schottky relationship. The catalytic property of metal was synergistically improved by the high surface area and facile electron transport property of CND particles. There are certain other reports of such synergistic interaction within CND-metal/metal oxide/metal phosphide etc. towards electrocatalysis. Along with that, nonmetal carbonaceous counter parts have also been studied as the alternative for metal. Karim Kakaei reported the enhanced activity of platinum (Pt) nanoparticles combined with CQDs towards ORR [39]. The activity of Pt/CQD composite was compared with Pt/C catalyst. The durability was checked by chronoamperometric measurements at 0.5 V vs Ag/AgCl in an O2-saturated 0.1 M HClO4 electrolyte (89.4% relative current persisted in acidic medium after 21000s compared to 81.6% for Pt/C), which showed that the composite can further be used for fuel cells. Bao et al. [40] prepared CQD/nickelcobalt phosphide (CQDs@NiCoP) catalysts through a facile annealing method for electrocatalytic hydrogen evolution reaction (HER), which yielded a lower initial overpotential than pure for NiCoP (108 vs.182 mV) and a lower Tafel slope (80 mVdecade−1). CQD aided high catalyst conductivity and significantly accelerated charge transfer. Li et al. found CQD loaded ruthenium nanoparticles (Ru@CQDs480) to be efficient electrocatalyst for reduction of hydrogen at alkaline medium [41]. They varied the medium pH and observed that even at extreme alkaline condition the as-formed Ru@CQDs480 exhibited significant catalytic behavior with onset overpotential of 0 mV, a Tafel slope of 47 mV decade−1, and good durability (Fig. 4).

(a) HER polarization curves for the Ru@CQDs480 and CQDs, Ru powder, 5% commercial Ru/C and 20% commercial Pt/C. (b) Durability test for Ru@CQDs480 in 1 m KOH before and after 10000 cycles. (c) Current-time curve of the Ru@CQDs480 before and after the addition of SCN− ions in 1 m KOH. (d,e) HER polarization curves (d) and Tafel plots (e) for the Ru@CQDs annealed at different temperatures and Pt/C catalyst. f) Nyquist curves for the Ru@CQDs annealed at different temperatures and Pt/C catalyst at η = 10 mV. g) Current density as a function of the scan rate for the Ru@CQDs annealed at different temperatures. h) XRD (i) and Raman pattern of Ru@CQDs calcinated at different temperature. (j) Schematic illustration of the synthesis of the Ru@CQDs480 electrocatalyst. Reprinted with permission from Reference [41]. Copyright © 2018, WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Li et al., reported CQD supported enhanced activity of ruthenium (Ru) nanocatalyst for HER [31]. The catalytic activity largely depended on the crystallinity and confinement effects. The experimental results showed that Ru nanoparticles were successfully anchored by strong coordination interactions between the d orbitals of Ru and the surface functional groups of CQDs, which is beneficial to the confinement of Ru nanoparticles around the CNDs and to prevent their aggregation. The Tafel slope with the catalyst at its optimized condition was ~ 63 mV decade−1. From the experimental data, they proposed Volmer-Heyrovsky mechanism for HER. A rhodium (Rh)/silicon quantum dot/CQD nanocomposite was reported by Dang et al. for HER [42]. The silicon quantum dots limited the size of Rh and prevented the aggregation and the CQDs greatly increased the electron transportation during the catalytic process. The catalyst showed a low overpotential of 36 mV at 10 mAcm−2, low Tafel slope of 26 mV.dec−1 and long-term stability. Nanocomposites based on transition metal oxide and carbon nanostructure are of low cost and provide high activity and stability. Liu et al., prepared a general route for synthesis of Ru-M (M=Ni, Cu, Mn) bimetallic nanoparticle and CND hybrid, and out of three kind of composite, RuNi/CQD exhibited highest performance for HER. [43]. The overpotentials of 13, 58, and 18 mV were shown by RuNi/CQDs at a current density of 10 mAcm−2 in 1M KOH, 0.5M H2SO4, and 1M PBS, respectively, for Ru loading at 5.93 μgRucm−2. The role of CQD particles was tested by replacing CQD with other carbon materials, but it was observed that CQD based particles showed high activity and durability. This might be due to strong interaction within d-orbital of Ru and functional groups of CQD which further prevented the aggregation of the particles. Theoretical studies revealed that Ni doping caused a moderate weakening of the hydrogen bonding energy of nearby surface Ru atoms, that played a critical role in improving the HER activity. Li et al. [44] prepared a CQDs decorated Ba0.5Sr0.5Co0.8Fe0.2O3−δ-perovskite nanofibers (CQDs@BSCF-NFs) catalyst for OER. The catalyst achieved a low overpotential (0.35 V at 10 mA cm−2), high current density (140.8 mAcm−2 at a potential of 1.65 V vs. RHE), and high stability. The high catalytic activity was attributed to high surface area of hollow nanofibers further increased by CQD and the numerous surface oxygen vacancies as well as the synergy between CQD and BSCF. Tian et al. [45] incorporated CQDs in MnO2 nanoflower, increasing the surface area, electron transfer speed, and electrical conductivity of the catalyst and used for OER in alkaline medium: with an overpotential of 343 mV and a low Tafel slope of 43.6 mV dec−1, it was a good catalysis. Zhang et al. [46] showed enhanced specific surface area and electric conductivity in a N,S-codoped CQD supported Co9S8 nanoparticles (Co9S8/CD@NSC) where CQD helped in smooth distribution of Co9S8 nanoparticles in the matrix. The obtained Co9S8/CD@NSC exhibited an excellent ORR and OER bifunctional catalytic activity and a great long-term durability, with a half-wave potential of 0.84 V vs. reversible hydrogen electrode (RHE) for the ORR and a low potential of 1.62 V versus RHE at 10 mA cm−2. These results were better than those exhibited by commercial Pt/C and RuO2 catalysts. Later, they used the Co9S8/CD@NSC catalyst for zinc/air battery with low charge/discharge voltage gap of 0.62 V and great cycling stability of over 125 h at 10 mA cm−2. Zhao et al prepared CQD/SnO2-Co3O4 composite for highly efficient electrochemical water oxidation [47]. Their experimental results showed that the Co atoms were considered as the active center, the nano sized SnO2 enhanced electronic conductivity and the insoluble CQDs layer on the surface effectively protected the catalyst. CQD also enhanced the electrocatalytic property with surface modification and doped hetero atoms. Tang et al. [48] reported a CQD-NiFe layered double-hydroxide (NiFe-LDH) composite for electrocatalytic water splitting with a high electrocatalytic activity (overpotential of ~235 mV in 1 M KOH at a current density of 10 mA cm−2) and stability for oxygen evolution. CQD increased the electroactivity and durability of NiFe-LDH through the bonding and interaction between the CQDs and the pure NiFe-LDH nanoplate. Canton-Vitoria et al. synthesized a CQD/MoS2 ensemble and used it for HER [49]. The quenching in CD fluorescence by MoS2 indicated electronic interaction between the two components. Yang et al. [16] prepared a nickel nanoparticle/CQD hybrid for hydrogen evolution in alkaline medium. The obtained hybrid showed good catalytic ability for HER with an onset potential comparable to that of platinum wire and a low Tafel slope of 98 mV dec−1. The good catalytic activity might be attributed to the synergistic interaction between nickel nanoparticles and CQDs at the Ni-O-C interface. Shih et al. [50] designed Cu2−xS/CQD nanoparticles that was used as cathode electrode material for ORR in direct methanol fuel cells. The ORR was carried out in acidic medium. The synthesized nanocomposite contained Cu9S5 and Cu2S. The catalyst provided a limiting density of −1.77 mA cm−2 at a scan rate of 5 mV s−1 and rotation rate of 3600 rpm. Wang et al. [51] developed a heteroatom doped CQD-metal sulfide hybrid for HER in acidic medium. They prepared a N,P-doped CQD/CoS2 hybrid through hydrothermal method, which showed long term stability and low overpotential during the HER. Zhang et al. [52] prepared a CQD/molybdenum phosphide (MoP) nanohybrid for HER in alkaline medium. The CQDs helped to alleviate the agglomeration and surface oxidation of MoP nanoparticles, ensuring that each MoP nanoparticle could be electronically addressed to elevate the electrocatalytic activity of the nanohybrid. The optimized CQDs/MoP hybrid synergistic catalysis toward HER in 1 M KOH electrolyte with a low onset potential of −0.08 V and a small Tafel slope of 56 mV dec−1. The nanohybrid showed high durability with negligible current loss for at least 24 h. Zhao et al. [53] prepared a CQD supported cobalt phosphate (CoPi) nanocomposite for electrocatalytic water splitting. The nanocomposite showed efficient catalysis for OER in both alkaline and neutral medium. The activity was a result of the synergistic interaction within CQD and CoPi. The CQD layer on the CoPi surface averted the CoPi from dissolution of enhancing the stability of the composite electrocatalyst. In neutral condition, the Tafel slope was 59 mV per decade. Cao et al. [54] prepared a N-doped CQD/graphene (N-CQD/G) composite and increased its activity for ORR by combining it with Pt nanoparticles (Pt/N-CQD/G). The composite was advanced with highly distributed Pt nanoparticles with small size, N-doping, and strong interaction between metal and support, as well as the unique structure of N-CQD/G hybrids with abundant edges and doping sites, high surface area, large pore size and high electrical conductivity. The structural advantages reflected in their high activity and durability (the mass activity decreased a ~32% after 5000 cycles). A three component electrocatalyst composed of Co3O4-CQD-C3N4 was designed by Guo et al. [55] for reduction of carbon dioxide to syngas. The composite was able to control the balance between the HER channel and CO2 reduction channel and initiated the reaction of CO2 reduction to CO in aqueous solutions at a low overpotential (0.17V) while the total current density reached up to 15 mA/cm2 at a potential of −1.0 V. A sandwich-like electrode with tungsten nitride nanosheets decorated with CQD (N-CDs@WN hybrid) was designed by Zhang et al. [56] for oxygen reduction. The hierarchical structure with large specific area, numerous active sites, and synergistic effect between CQD and WN might facilitate the conductivity and mass transfer during the ORR process. The low Tafel slope of 79 mV dec−1 indicated high catalytic efficiency. Even after 20000s, the catalyst retained ~82.3% of its original ORR performance.
Nevertheless, metal free CQD/carbon nanoparticle composites also showed efficient electrocatalytic activity. Hu et al. reported a N-doped CQD decorated graphene composite for ORR [57]. The graphene was support and CQD provided the active sites. The catalyst showed comparable catalytic activity, durability, stability and selectivity with commercial platinum electrocatalyst during alkaline ORR. Zhang et al. [27] examined the superior behavior of CQD/reduced graphene oxide (rGO) composite compared to CQD doped rGO. They prepared a N,S-dual doped CQD/rGO nanocomposite and analyzed its catalytic activity towards ORR in N2-saturated and O2-saturated 0.1 M KOH. The doping of hetero atom played an important role in catalysis. The low Tafel slope (71 mVdecade−1) indicated high catalytic activity. Wang et al. [58] reported that the effect of defect in the CQD/graphene composite helped to achieve high catalytic activity towards ORR. The composite was used as an air cathode material for zinc/air battery. Zhou et al. [59] synthesized a CQD decorated reduced graphene oxide using microorganisms. Both CQD and graphene were doped with hetero atoms; N, S, and P. This hybrid had long term stability and significant catalytic activity towards reduction of oxygen through a four electron pathway in alkaline medium. The catalytic property was as a result of the combined effect of heteroatom doping and numerous surface/edge defects that induced the high-density distribution of highly active and stable catalytic sites. Again, the strong coupling effects between CQDs and graphene facilitated the charge transport and ionic motion during the reduction of oxygen. The electrochemical studies and fabrication process of the catalyst are shown in Fig. 5.

(a) CV curves of the CDs/M-rGO in N2-saturated and O2-saturated 0.1 M KOH solutions at a scan rate of 10 mV s1; (b) RDE curves of C-rGO, M-rGO, CDs/M-rGO and Pt/C in O2-saturated 0.1 M KOH solutions at a scan rate of 10 mV s1 and a rotation speed of 1600 rpm; (c) RDE curves of the CDs/M-rGO at different rotation rates; (d) K-L plots at different potentials for the CDs/M-rGO; (e) chronoamperometric responses to the injection of methanol into an O2-saturated 0.1 M KOH solution at the CDs/M-rGO and Pt/C electrodes; (f) chronoamperometric responses of the CDs/M-rGO and Pt/C electrodes at 0.4 V (vs. SCE) in O2-saturated 0.1 M KOH solutions, normalized to the initial current responses. Reactions and operations involved in the process of fabricating CDs/M-rGO (g); the process of fabricating C-rGO (h). Reprinted with permission from Reference [59]. Copyright © 2016, The Royal Society of Chemistry.
Niu et al. [60] prepared a nitrogen rich CQD supported graphene oxide for reduction of oxygen in alkaline medium. The as-prepared hybrid showed positive onset potential (0.13 V vs. Ag/AgCl) and high kinetic current density (up to 18.4 mA cm−2 at −0.70 V) comparable to commercial Pt/C catalyst during ORR. A N-doped CQDs supported carbon nanosheet (N-CNA) particle was used as electrocatalyst for ORR in an O2-saturated 0.1 M KOH solution by Zhang et al. [61]. The N-CNA showed a small over potential of −0.08 V that could be compared to intrinsic low over potential for ORR. The catalytic activity was comparable to commercial Pt/C. The wide current plateau within a wide potential range indicated high performance of the N-CNA electrocatalyst. Another synergistic interaction within N-doped CQD and graphene was reported by Liu et al. [62], showing that the synergistic interaction within CQD and graphene oxide boosted the catalytic activity and the doping could modify the charge density on the electrocatalyst and weaken the adsorption of reaction intermediates (O and OOH). The composite was used for ORR process, through a four-electron way mechanism, with an onset potential of −95 mV and kinetic current density of 12.7 mA cm−2 at −0.7 V (vs. Ag/AgCl). The results indicated good catalytic behavior. The catalyst also possessed significant stability (~88% retention of current density even after 40000 s), durability and methanol tolerance.
3.3 GQD nanoparticles
Colloidal GQDs are new addition to 0-D carbonaceous nanoparticles. GQDs with high electrical conductivity and active sites have been used for electrocatalysis. Li et al. [63] applied solution chemistry approach for the synthesis of N-doped GQDs and explored their size with respect to their catalytic property for ORR in 0.1M KOH. The doping of hetero atom and size of the catalyst played a major role. The linear sweep voltammetry showed that the N-doped GQDs have high, size-dependent electrocatalytic activity for the ORR They prepared three kinds of GQDS (1, 2 N-doped and 3-undoped) and demonstrated via both experimental and theoretical data, that the size and abundance of nitrogen atom with specific bonding configuration have an important role in the catalysis. Figure 6 shows the results of electrochemical studies and the structure of GQDs.
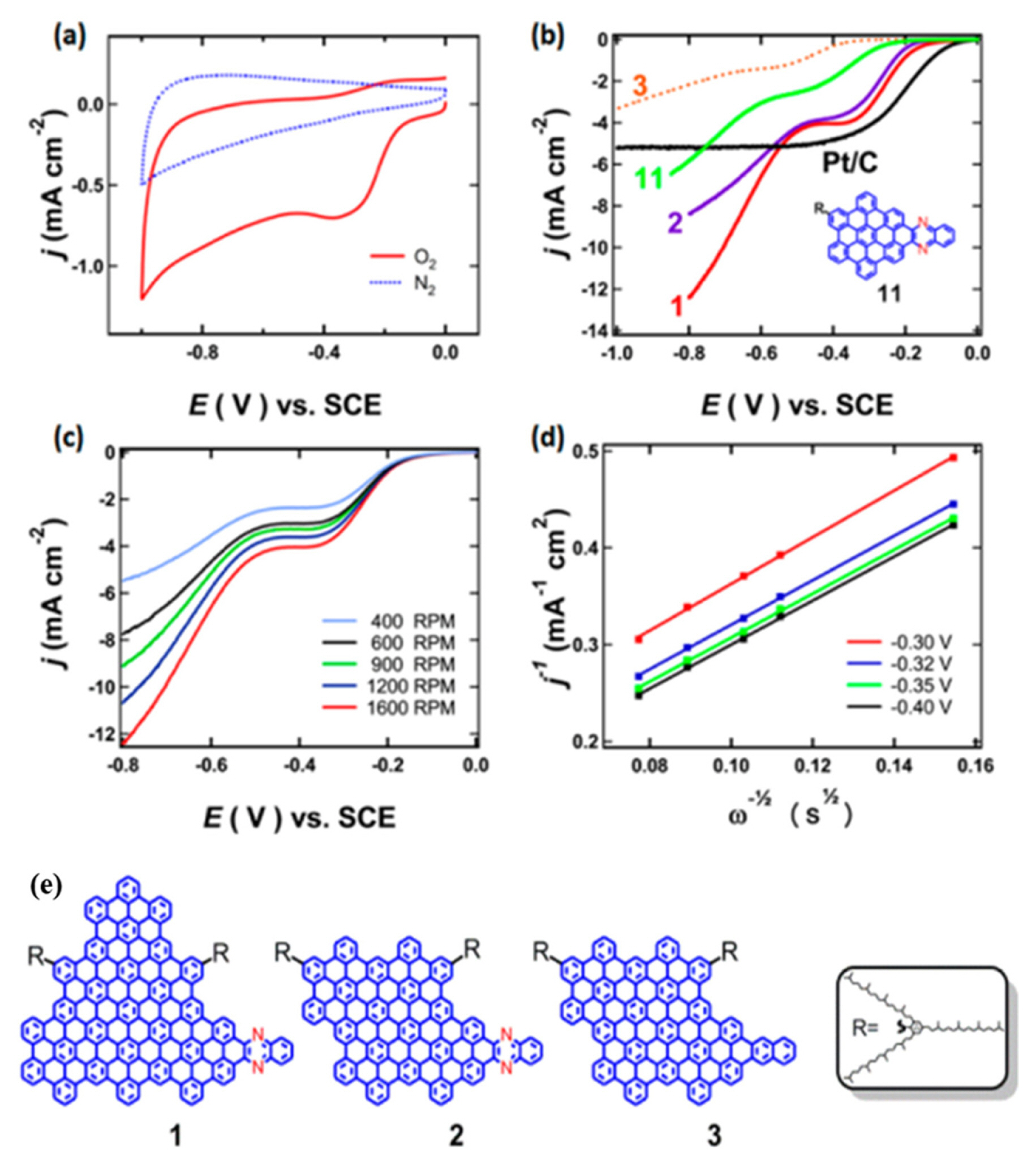
(a) CV curves (scanning rate of 50 mV s−1) of 1 on a glassy carbon RDE in a N2- and an O2-saturated 0.1 M KOH solution. (b) LSV curves (10 mV s−1) for 1–3 and Pt/C on a RDE (1600 rpm) in an O2-saturated 0.1 M KOH solution. Also shown is the LSV curve for 11, a much smaller N-substituted heterocycle with structure shown in the inset. (c) LSVs (10 mV s−1) for 1 on a RDE in an O2-saturated 0.1 M KOH solution with various rotating speeds. (d) Koutecky-Levich plots for 1 obtained from the LSV curves in (c) at various voltage values. A global fitting of the plots reveals that the number of electrons transferred per O2 molecule is 3.9. (e) Structures of N-Doped GQD 1, 2 and an Undoped GQD 3 for Comparison. Reprinted with permission from Reference [63]. Copyright © 2012 American Chemical Society
Yan et al. prepared GQDs from graphene flakes for ORR [64]. They found that the GQD catalyst showed a high electrocatalytic activity for the ORR, with a 70 times enhancement in voltammetric current in oxygen saturated KOH compared to that in N-GQD. The ORR onset potential was −0.25 V with the reduction peak at −0.45 V. The ORR followed a two-electron mechanism. Guin et al. [65] used green luminescent GQDs as impending electrocatalyst for reduction of K3[Fe(CN)6]. The standard rate constant of this heterogeneous reaction was 2.69 × 10−3 cm s−1 within a scan rate of 25–300 mV s−1. They proposed that the edge functionalities and the basal graphitic π-units facilitated the outer-sphere heterogeneous electron transfer kinetic of the bench-mark redox mediator, which could be further used for various electrochemical applications. Favaro et al. [66] synthesized singly and doubly doped graphene oxide quantum dots with N and B hetero atoms for ORR. Through DFT calculation, they determined that the selectivity of the reaction is controlled by the oxidation states of the dopants. The as-prepared GQDs follow a two-electron reduction path that leads to the formation of hydrogen peroxide, whereas after the reduction with NaBH4, the same materials favor a four-electron reduction of oxygen to water.
3.4 GQD supported metallic and nonmetallic nanoparticles
The electronic interaction between GQD and metallic nanoparticles results in the modification of the catalytic properties of the metals and makes GQD an active support. Deming et al. [67] prepared a Pd nanoparticle supported N-doped GQD for the electroreduction of oxygen in alkaline medium. The catalyst worked better than Pd supported on undoped GQDs and Pt/C. The Pd atoms had a strong interaction with the pyrrole ring. The p-type pyridinic and pyrrolic N-centers in N-doped GQDs increased the catalytic activity in terms of manipulating the Pd-oxygen intermediate. The highest specific current density of 25.4 A/m2 and best Tafel slope of 120 mV/dec were obtained for the nitrogen centers felicitated charge transfer and provided optimal binding condition within metal surface and oxygenated intermediates. Deming et al. also reported about Pd supported GQD nanocatalyst [68]. Voltammetric studies showed apparent electrocatalytic activity toward oxygen reduction in alkaline medium. The peak activity of the sample was attributed to the optimal interactions between Pd and GQD, where the GQD defects promoted charge transfer from metal to GQDs and weakened interactions with oxygenated intermediates, leading to enhanced ORR activity. The corresponding defect concentration was higher than that identified with the platinum counterparts due to the stronger affinity of oxygen to palladium. The best Tafel slope was 71.6 mV/dec and the highest kinetic current density of 1.08 A/m2 at + 0.90V for optimal interactions between the GQD structural defects and the palladium nanoparticles was recorded. He et al. [69] prepared nanosized GQDs through acid etching of carbon fibers and used these as effective substrate-supports for platinum nanoparticles (Pt/G), synthesized by thermolytic reduction of platinum (II) chloride in ethylene glycol. The as-synthesized Pt/G particles showed enhanced catalytic activity during reduction of oxygen, with an onset potential of +1.05 V. This result was 70 mV more positive than that of Pt/C, and a specific activity almost nine times higher, showing better activity of Pt/G over commercial Pt/C catalysts. These results were ascribed to the abundant structural defects of the nanosized GQDs that manipulated the dissociative adsorption of oxygen and the binding of reaction intermediates O* and HO* on platinum surfaces. Another GQD supported platinum nanocatalyst (Pt/G) was prepared by Song et al. [30] for ORR. The electrocatalytic performance was accounted by the deliberate manipulation of the adsorption of oxygen and reaction intermediates on platinum by the GQD structural defects through partial charge transfer. Pt-GQD catalysts were prepared at different conditions, however, materials at two reaction conditions (160°C for 12 h and 180°C for 6 h) exhibited maximum efficiencies (kinetic current densities of 29.2 and 24.6 A/m2 at + 0.90 V, respectively). Apart from platinum noble metal, gold has also been used for catalysis due to its lower cost and effective catalytic activity. The activity of gold nanoparticles with GQD support has been explored by Luo et al. [70]. They prepared GQD/gold nanoparticle hybrid (Au@GQDs core-shell nanostructure with an average diameter of 15.6 nm) and used it for catalytic HER in 0.5 M H2SO4 medium. The Tafel slopes were 78 and 75 mV/dec, respectively for Au@GQD and Au nanoparticles, but the former had a lower over potential compared to latter. The higher activity of the composite was attributed to the strong chemical coupling/interactions between components GQD and Au nanoparticles. The strong GQDs coating layer at Au surface prevented the aggregation of Au nanoparticles and boosted the activity. Liu et al. synthesized a Cu2O/CuO-GQD composite for alkaline electroreduction of oxygen [71]. The Cu/C ratio played an important role during catalysis. The comparison of the activity of Cu/GQD nanocomposite with Pt/GQD and Pt/C showed that the composite had the lowest onset potential towards ORR. Non-noble metal based GQD catalysts also showed enhanced catalytic activity. Koh et al. conjugated iron (II) phthalocyanine (FePC) with GQDs and used the catalyst for ORR in alkaline medium [72]. The catalyst also showed potential tolerance against methanol and carbon dioxide. The FePC-GQD catalyst followed a four-electron path and reduced oxygen directly to OH− in alkaline electrolyte, with the Tafel slope of the reaction being 23 mV dec−1. When GQD followed a two-electron transfer process yielding HO2−, the Tafel slope was 28.9 mVdec−1, indicating the four-electron mechanism to be the preferable path. A GQD loaded and molybdenum doped porous Ni3S2 hybrid (G-Mo-Ni3S2) was in situ grown on Ni foam by GQDs and was used for overall water splitting by Li et al. [73]. The in situ coupling of GQD with metal hybrid modified the electronic behavior of the catalyst and exposed more active sites for reaction. This boosted the overpotential of HER and OER by 68 mV at 10 mA cm−2 and 326 mV at 20 mA cm−2 in 1.0 M KOH, respectively. The two-electrode cell, using G-Mo-Ni3S2, was constructed to deliver low cell voltage of 1.58 V at water-splitting current of 10 mA cm−2 with a significant stability for more than 50 h. Chen et al. used GQDs to enhance the activity of Bi2O3 nanosheet for reduction of CO2 to formate [74]. The as prepared Bi2O3-NGQDs demonstrated a nearly 100% formate Faraday Efficiency (FE) at a moderate overpotential of 0.7 V with a good stability and a high activity. The DFT calculation suggested that the increase in activity could be attributed to better adsorption of reaction intermediate at the GQD surface. In Figure 7, the experimental results and schematics of synthesis are documented.

a) CO2 adsorption isotherm for Bi2O3 and Bi2O3-NGQDs. b) Tafel plots for the Bi2O3-NGQDs, Bi2O3, and NGQDs. DFT calculations for the electrochemical reduction of CO2 to formate process over Bi2O3-NGQDs and Bi2O3. c) Calculated free-energy diagram. d) Calculated adsorption energy of CO2, OCHO*, and HCOOH (ads). e) Schematic of the synthesis process for Bi2O3-NGQDs. Reprinted with permission from Reference [74]. Copyright © 2018 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
Vinoth et al. [75] decorated N-doped GQDs (N-GQDs) on a 3D MoS2-reduced graphene oxide (rGO) framework via a facile hydrothermal method for ORR. They concluded that the chemical interaction between the electroactive N-GQDs and MoS2-rGO increased surface area and pore size of the N-GQDs/MoS2-rGO nanohybrid synergistically, which in turn improved the ORR onset potential to +0.81 V vs reversible hydrogen electrode (RHE). Moreover, the N-GQDs/MoS2-rGO nanohybrid showed better ORR stability for up to 3000 cycles with negligible deviation in the half-wave potential. Lv et al. [76] fabricated a noble metal-free electrocatalyst via in situ formation of nanocomposite of N-doped GQDs (NGQDs) and Ni3S2 nanosheets on the Ni foam (Ni3S2-NGQDs/NF). The resultant Ni3S2-NGQDs/NF served as an active, binder-free, and self-supported catalytic electrode for direct water splitting. At a current density of 10 mA cm−2, the overpotential was 216 mV for OER and 218 mV for HER in alkaline media. This bifunctional electrocatalyst enabled the construction of an alkali electrolyzer with a low cell voltage of 1.58 V vs. reversible hydrogen electrode (RHE) at 10 mA cm−2.
Apart from GQD metal/metal oxide composites, metal free GQD/carbon nanoparticle composites were also investigated for different electrocatalytic reactions. Luo et al. synthesized a GQD supported N,S-co doped reduced graphene oxide (N,S-RGO/GQDs) by microwave treatment for ORR in alkaline medium [77]. The doping enhanced the catalytic activity. The onset potential, reduction peak, and the limiting current of N,S-RGO/GQDs hybrids potential of were −0.10 V and −0.25 V, and 4.53 mA cm−2 (at −0.8 V a rotary speed of 1600 rpm), respectively. These results showed that N,S-RGO/GQDs have good catalytic activity and are potential metal-free catalyst for the ORR in an alkaline solution through a four electron mechanism. Yao et al. prepared S-doped GQD decorated carbon nanoparticles (S-GQDs/CNPs) from dehydration of fructose and H2SO4 for ORR [78]. The low reduction peak at −0.396 V and high peak current density of −2.02 mA/cm2 showed the efficiency of the composite. From the experimental data, the layer by layer modified method could inhibit the occurrence of a two-electron pathway and cause a four-electron pathway for ORR.
The S-GQDs/CNP nanohybrid showed better methanol tolerance and long-term durability than commercial 20% Pt/C. Wang et al. [79] reported synergistic within GQD and graphene hydrogen composite during electroreduction of oxygen. The nanocomposite exhibited fine ORR activity and durability in alkaline solution, which showed a comparable discharge performance of zinc/air batteries with Pt/C under a current density of 20 mA cm−2. The high performance was attributed to the rich edge defects present at the composite surface. Graphene nano ribbons (GNR) were also reported to get support from GQD and perform as excellent electrocatalyst for oxygen reduction. Jin et al. [80] prepared a GQD/GNR composite for ORR in 0.1 M KOH medium, with the numerous surface/edge defects on the GQD/GNR surfaces and at their interface acting as the active sites. This, coupled with efficient charge transfer between the intimately contacted GQDs and GNRs, rationalized the observed electrocatalytic performance. The Tafel slope was measured to be ~87.7 mV per decade, indicating high efficiency. The catalyst showed significant stability over 26 hrs. Hasanzadeh et al. prepared a GQD functionalized by chitosan (CS) and beta-cyclodextrin (β-CD) as a new support nanocomposite material for efficient methanol electrooxidation [81]. The anodic peak currents showed a linear relationship with the square root of the scan rate for characteristic diffusion-controlled process. The overall electron transfer coefficients were 0.44 and 0.5 while the Tafel slopes were 0.7 mV and 0.63 V for CS and β-CD support, respectively towards methanol oxidation. The observed peak current values indicated the essential roles of CS and β-CD in the observed electrocatalytic behavior. Zhou et al. prepared another metal free electrocatalyst for reduction of oxygen [82]. The synergistic effect of GQD and multi walled carbon nanotube (MWCNT) was used for the catalysis in O2-saturated 0.1 M KOH solution. The GQD intake was optimized by considering the ratio of lattice and adsorbed oxygen in the composite. At optimized ratio of GQD-MWCNT, the ORR peak was observed at −0.25 V. The highest current density was obtained as 3.47 mA cm−2. Fei et al. synthesized a B,N-doped GQDS/Graphene hybrid nanoplatelets for electrocatalysis of oxygen [83]. The electrochemical studies and the synthetic protocol are shown in Fig. 8.
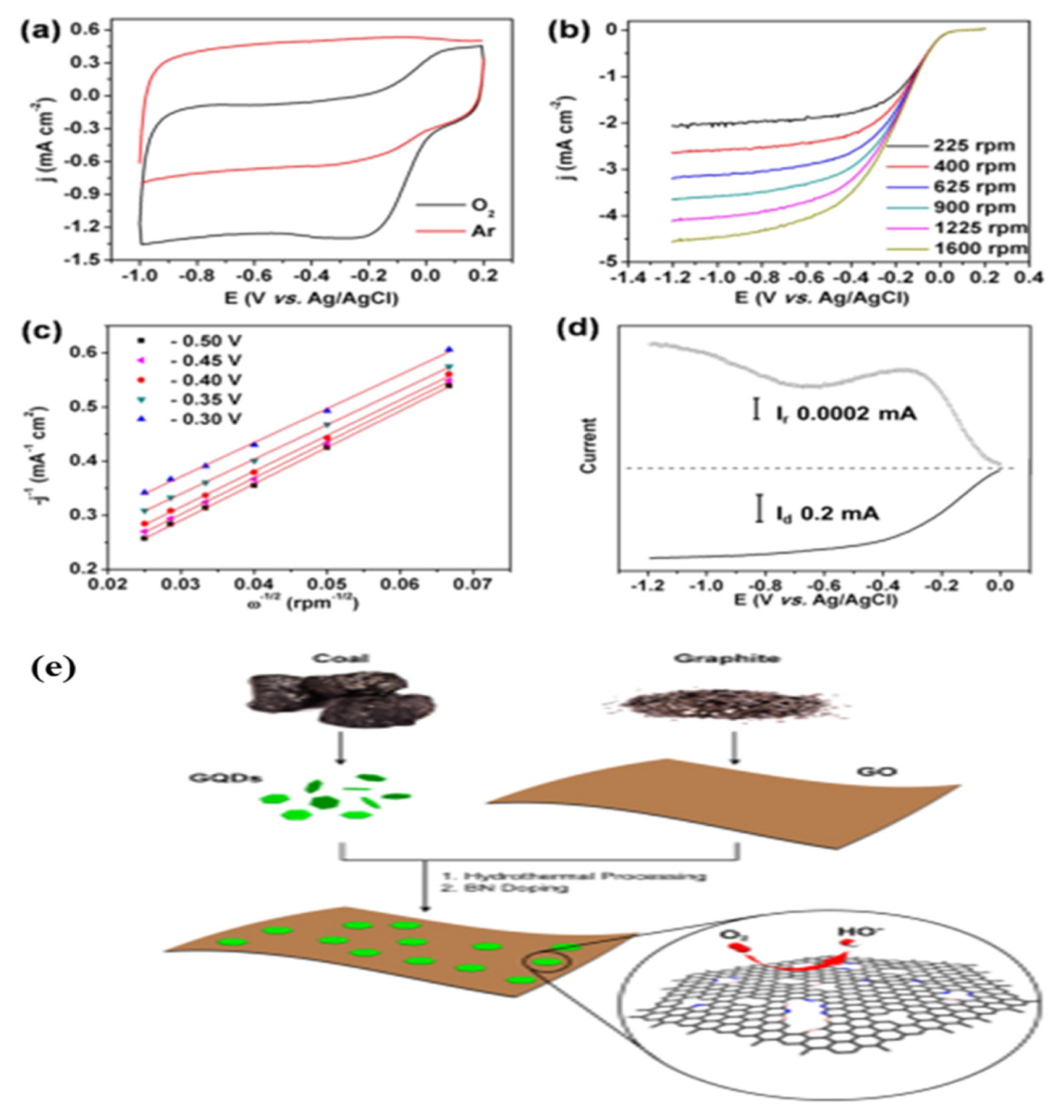
(a) Cyclic voltammograms of ORR on BN-GQD/G-30 in Ar- and O2-saturated 0.1 M KOH solution at a scan rate of 100 mV s−1. (b) RDE linear sweep voltammograms of ORR on a BN-GQD/G-30 electrode at different rotating speeds in O2-saturated 0.1 M KOH solution with a scan rate of 5 mV s−1. (c) Koutecky-Levich plots of BN-GQD/G-30 derived from RDE voltammograms (b) at different potentials. (d) Rotating ring disk electrode voltammograms of ORR on a BN-GQD/G-30 electrode with a scan rate of 5 mV s−1. (e) Illustration of the preparation procedure for the BN-GQD/G nanocomposite. Reprinted with permission from Reference [83]. Copyright © 2014 American Chemical Society.
The GQD particles self-assembled on graphene and the hybrid material combined the advantages of both components, such as abundant edges and doping sites, high electrical conductivity, and high surface area, making the resulting materials excellent oxygen reduction electrocatalyst with activity even higher than that of commercial Pt/C in alkaline media. The optimum activity of the catalyst was obtained by tuning the concentration of doped atoms. The onset potential of composite at optimized condition, measured from the rotating disk electrode was ~15 mV more positive than of Pt/C. In addition, the diffusion-limited current density and current density of the composite was larger than that of Pt/C within the range of 0.20 to −0.15 V at the same mass loading, suggesting the higher ORR activity of the composite than Pt/C. For the four-electron transfer mechanism, the kinetic current density was obtained as 11.1 mA cm−2 for the composite.
4. Conclusion and Perspective
Intense effort has been put on to find suitable and efficient catalysts for different electrochemical reactions such as HER, OER, ORR for generation of clean and sustainable energy. As the alternative to expensive platinum, ruthenium and iridium-based catalysts CD particle with or without metal hybrid/composite have emerged. The large surface area, facile electron transfer and tunable morphology of CD particles enables high catalytic action. The unique optoelectronic properties of CD particles can be tuned through their size, doping and surface functional groups. Doping of hetero atoms at the carbon core plays an important role in catalysis. The co-doping and concentration of dopant are also important. The CD supported metallic or nonmetallic nanoparticles benefit from the unique properties of CD support. Furthermore, CD support might protect from catalyst poisoning and oxidation in reaction medium. Since the activity of Pt/C catalyst depends on the Pt loading, the comparison between Pt/C and CD based catalysts might vary at different conditions. CD based catalysts are yet to overcome the mass production related limitation to achieve ultimate activity to solve the energy issues. More strategist way for carbon supported nanoparticle synthesis have to be adopted to reduce side products and achieve maximum product. Synthesis of CD particles at an industrial scale is needed to widen their applications. Stability, resistance towards corrosion of electrodes and catalyst poisoning are also persisting problems for the electrocatalytic reactions that need tactical solutions.
In future, these limitations are expected to be overcome with more innovative and facile synthetic routes and precursors. More metal free catalysts would be explored to avoid metal related toxicity and corrosion. Doping of heteroatom such as sulphur, phosphorous etc. might be explored to achieve different synergistic interactions. Organic polymer dots might be applied as electrode material along with metal or nonmetal support. These different strategies towards employing a series of 0-D CD particles might enlighten future research. The potential use of CDs might be extended by combining their optical and electrochemical properties to fabricate low-cost commercial fluorescent electrodes.
Acknowledgement
This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by The Ministry of Science, ICT and Future Planning (NRF-2019R1A2B5B02069683).