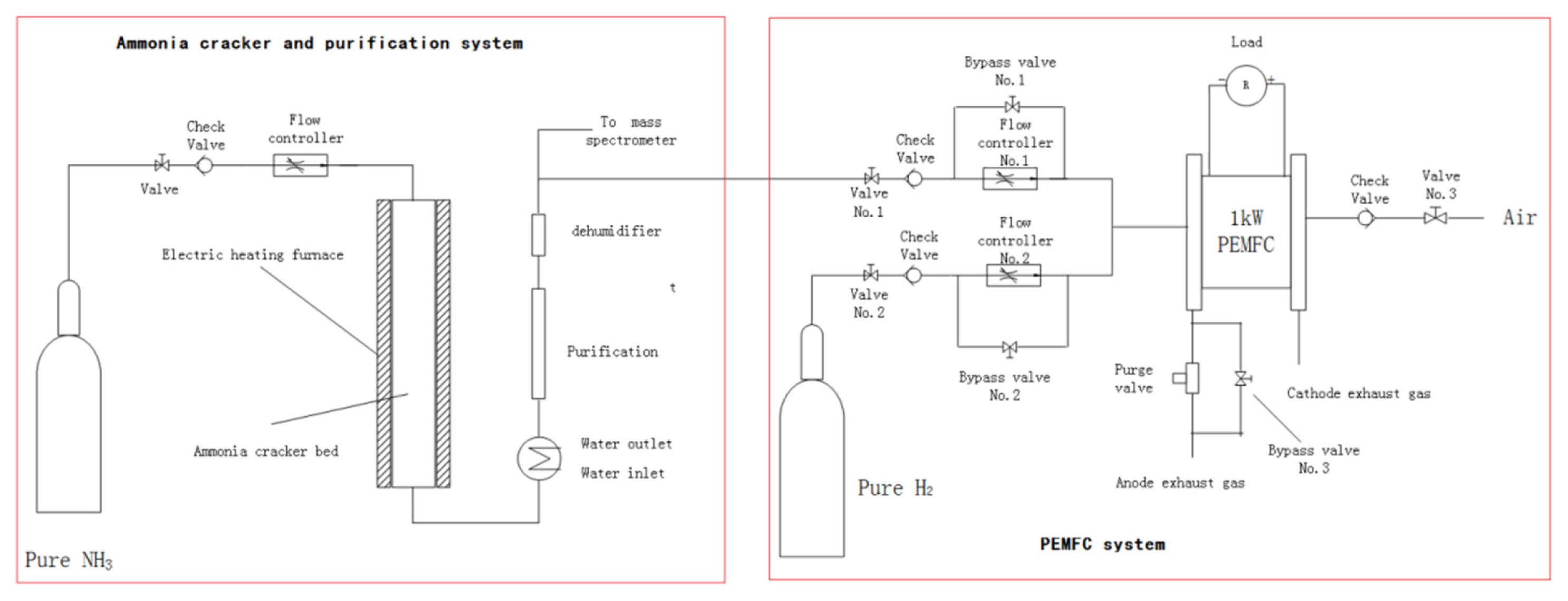
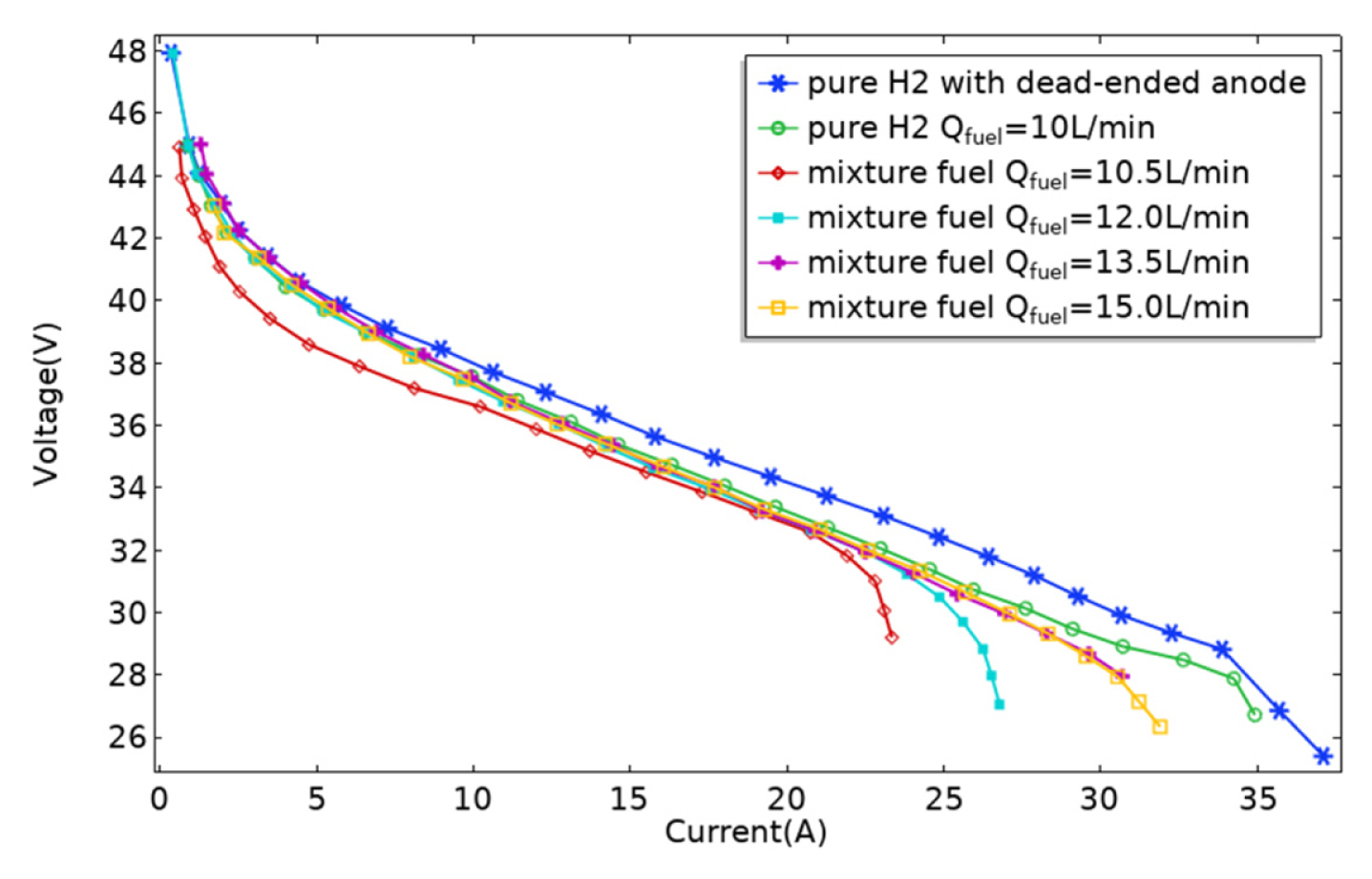
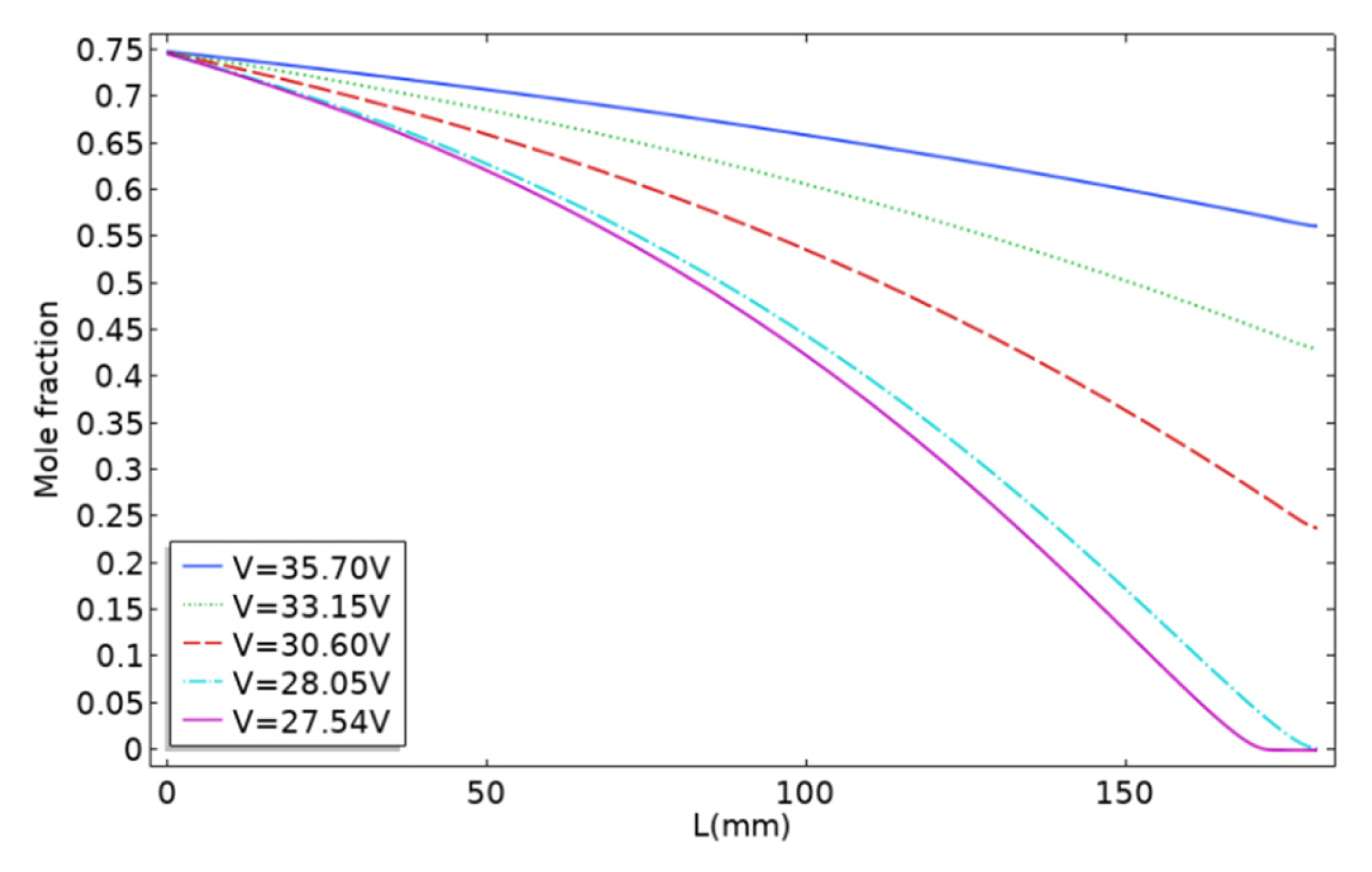
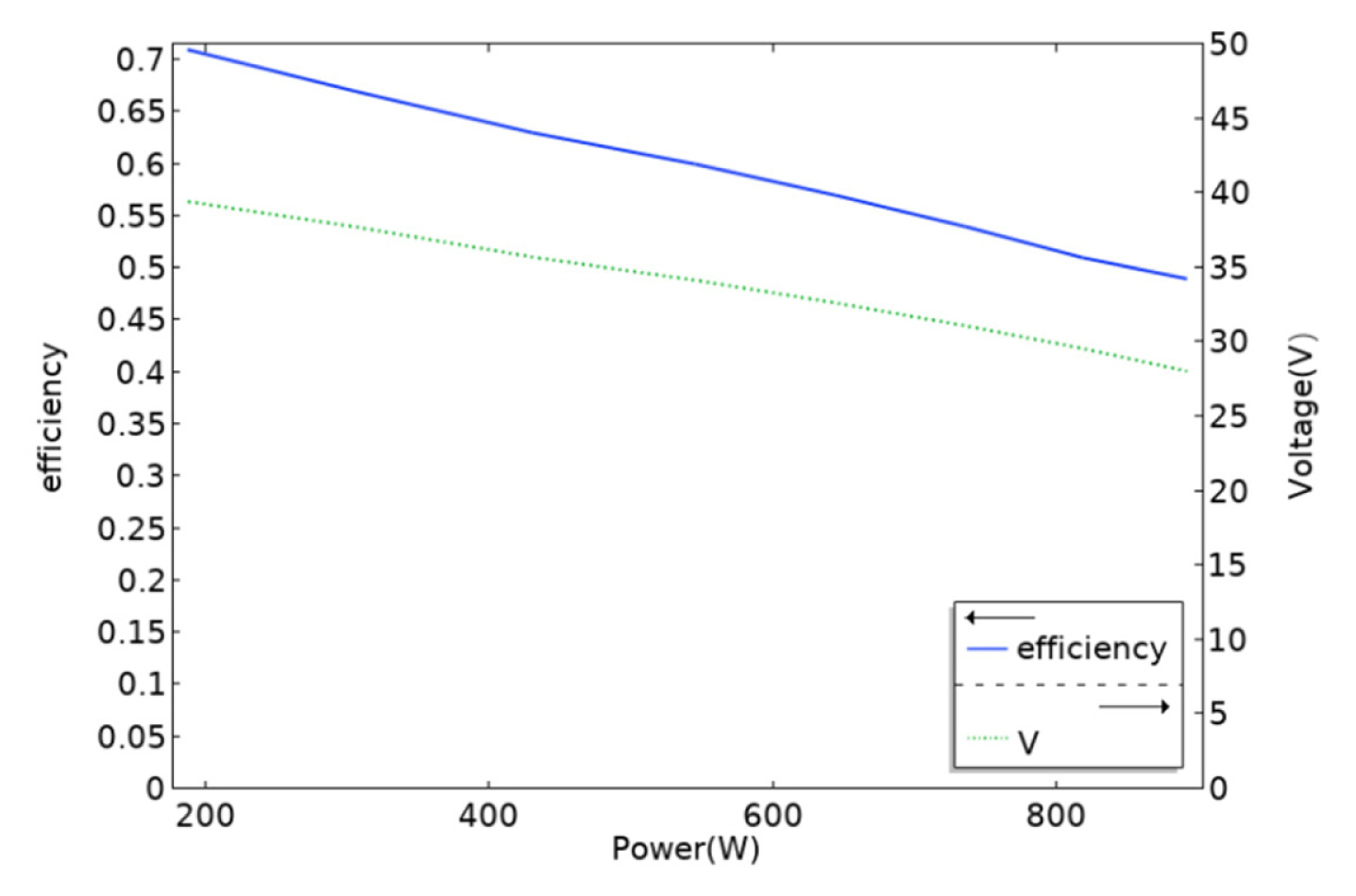
[1] ZP. Cano, D. Banham, S. Ye, A. Hintennach, J. Lu and M. Fowler,
Nat Energy,
2018,
3(
4), 279–289.


[2] M. Khzouz, EI. Gkanas, S. Jia, F. Sher and MA. Qubeissi,
Energies,
2020,
13(
15), 3783.

[3] R. Moradi and KM. Groth,
Int J Hydrog Energy,
2019,
44(
23), 12254–12269.

[4] X. Wu, N. Li, X. Wang and W. Zhao,
IEEE Transactions on Sustainable Energy,
12(
2), 1020–1031.

[5] G. Yang and Y. Jiang,
Int J Energy Res,
2020,
44(
11), 8340–8361.

[6] H. Cheng, B. Meng, C. Li, X. Wang, X. Meng and J. Sunars,
Int J Hydrog Energy,
2020,
45(
12), 7423–7432.

[7] TY. Amiri, K. Ghasemzageh and A. Iulianelli,
Chem Eng Process,
2020,
157, 108148.

[8] S. Sengodan, L. Rong, J. Humphreys, D. Du, W. Xu, H. Wang and S. Tao,
Renew Sust Energ Rev,
2018,
82, 761–780.

[9] N. Lu and D. Xie,
Int J Chem React Eng,
2016,
14(
1), 1–31.

[10] L. Ouyang, K. Chen, J. Jiang, XS. Yang and M. Zhu,
J Alloys Compd,
2020,
829, 154597.

[11] SS. Mohammadshahi, EM. Gray and CJ. Webb,
Int J Hydrog Energy,
2016,
41(
5), 3537–3550.

[12] PM. Modisha, C. Ouma, R. Garidzirai, P. Wasserscheid and D. Bessarabov,
Energy Fuels,
2019,
33(
4), 2778–2796.

[13] PT. Aaldto-Saksa, C. Cook, J. Kiviaho and T. Repo,
JPower Sources,
2018,
396, 803–823.

[14] H. Teng, Q. Pei and C. Ping,
J Energy Chem,
2015,
24(
5), 587–594.

[15] M. Aziz, AT. Wijayanta and A. Nandiyanto,
Energies,
2020,
13(
12), 3062.

[16] AT. Wijayanta, T. Oda, CW. Purnomo, T. Kashiwagi and M. Aziz,
Int J Hydrog Energy,
2019,
44(
29), 15026–15044.

[17] N. Rajalakshmi, TT. Jayanth and KS. Dhathathreyan,
Fuel Cells,
2003,
3(
4), 177–180.

[18] IS. Girnik, YI. Aristov, H. Lund and MJ. Kaiser, Energy, 2020, 211.
[19] CP. Girotto, S. Campos and L. Campos,
Heliyon,
2020,
6(
2), e03397.

[20] GP. Holland, BR. Cherry and TM. Alam,
J Phys Chem,
2004,
108, 16420–16426.

[21] BV. Hassel, JR. Karra, J. Santana, S. Saita, A. Murray and D. Goberman,
Sep Purif Technol,
2015,
142, 215–226.

[22] P. Sajjan, V. Nayak, M. Padaki, VY. Zadorozhnyy and PA. Konik,
Energy Fuels,
2020,
34(
9), 11699–11707.

[23] A. Alkali,
J Power Energy Eng,
2020,
8(
02), 1.

[24] H. Hunter, JW. Makepeace, TJ. Wood, OS. Mylius, MG. Kibble and JB. Nutter,
J Power Sources,
2016,
329, 138–147.

[25] S. Liu, T. Chen and Y. Xie,
Int J Green Energy,
2020,
17(
4), 255–273.

[26] SA. Grigoriev, AA. Kalinnikov, NV. Kuleshov and P. Millet,
Int J Hydrog Energy,
2013,
38(
20), 8557–8567.

[27] N. Ahmadi, AD. advand, I. Mirzaei and S. Rezazadeh,
Int J Energy Res,
2018,
42(
8), 2805–2822.

[28] JX. Liu, H. Guo, F. Ye and CF. Ma,
Energy,
2017,
119, 299–308.

[29] SW. Tsai and YS. Chen,
Appl Energy,
2017,
188, 151–159.

[30] B. Wang, K. Wu, Z. Yang and K. Jiao,
Energy Convers Manag,
2018,
171, 1463–1475.

[31] S. Yesilyurt, JB. Siegel and AG. Stefanopoulou,
J Fuel Cell Sci Technol,
2012,
9(
2.


[32] M. Le Bars and MG. Worster,
J Fluid Mech,
2006,
550, 149–173.

[33] RJ. Kee, ME. Coltrin and P. Glarborg, Chemically Reacting Flow: Theory, Modeling, and Simulation. Wiley, American,
2017. p.91–149.

[34] EU. Ubong, Z. Shi and X. Wang,
J Electrochem Soc,
2009,
156(
10), B1276.