Effect of pH and Concentration on Electrochemical Corrosion Behavior of Aluminum Al-7075 T6 Alloy in NaCl Aqueous Environment
Article information
Abstract
In the present study, the corrosion behavior of aluminum Al-7075 tempered (T-6 condition) alloy was evaluated by immersion testing and electrochemical testing in 1.75% and 3.5% NaCl environment at acidic, neutral and basic pH. The data obtained by both immersion tests and electrochemical corrosion tests (potentiodynamic polarization and electrochemical impedance spectroscopy tests) present that the corrosion rate of the alloy specimens is minimum for the pH=7 condition of the solution due to the formation of dense and well adherent thin protective oxide layer. Whereas the solutions with acidic and alkaline pH cause shift in the corrosion behavior of aluminum alloy to more active domains aggravated by the constant flux of acidic and alkaline ions (Cl− and OH−) in the media which anodically dissolve the Al matrix in comparison to precipitated intermetallic phases (cathodic in nature) formed due to T6 treatment. Consequently, the pitting behavior of the alloy, as observed by cyclic polarization tests, shifts to more active regions when pH of the solutions changes from neutral to alkaline environment due to localized dissolution of the matrix in alkaline environment that ingress by diffusion through the pores in the oxide film. Microscopic analysis also strengthens the results obtained by immersion corrosion testing and electrochemical corrosion testing as the study examines the corrosion behavior of this alloy under a systematic evaluation in marine environment.
1. Introduction
Aluminum based alloys have been utilized in moderation due to their superiority as lightweight alloys with excellent combination of mechanical properties [1]. The combination has proved them to be suitable for variety of applications but their most common has been explored in the field of aerospace engineering [2,3]. However, it has been keener to investigate the electrochemical behavior of aluminum alloys [4] because of their extensive use in marine and aerospace applications [5,6]. For example Al7075-T6 is a light-weight aluminum alloy with unique ageing treatment being utilized in aerospace and marine applications owing to its superior mechanical properties due to precipitation-hardening [7,8]. These alloy have been shown to be highly sensitive to heat treatment in terms of their microstructure and the composition (Table 1) of the matrix which is effected by the type, size and shape of the precipitates formed in different alloys [9–11].
Over the course of studies there have been efforts to investigate the electrochemistry of these alloys under different heat treatments in natural environmental conditions and modified conditions to effectively understand the kinetics of corrosion and methods of corrosion inhibition [12–14]. Some studies have focused on the effect of alloying additions on the corrosion behavior of different aluminum alloys as well as the heat treatment-led precipitate formation [15–17]. Aluminum alloys generally form passive film which are relatively stable and hence corrosion resistant. The as such formed passive layer, being oxide in nature, is a good insulator, and its stability and formation is dependent on the composition of the alloy. What is known, in addition to the effect of elements on formation and stability of passive layer, is the cathodic behavior of the precipitates formed in the alloys due to ageing and temper treatments which lead to local cell formation with the matrix. Hence, in this manner localized pitting behavior is observed on the alloy surface given that pitting attack is also influenced by other factors such as ion concentration, temperature, pH and turbidity. Corrosion behavior of Al-7075 [18] as well as Obert [19] who studies the effect of certain resin coatings on corrosion whereas localized corrosion has been quantified [20]. However to the authors’ knowledge, there is no published literature evaluating the electrochemical performance of Al 7075 alloy which has been tempered to “T6 condition” under varying pH. [13,21,22]. And the effect of pH and Cl− ion concentration on the pitting behavior on T6 tempered condition had not yet been evaluated to an in-depth extent as tempering leads to formation of precipitates which influences the mechanical properties of the alloy [23–25]. This has propelled the direction of this research work to investigate the electrochemical behavior of T6 tempered 7075 alloy under simulated sea water environment. Previous studies have showed that T6 aging treatment of Al-7075 alloy leads to formation of multiple second phases in the Al matrix. This includes primarily Al7Cu2Fe and (Al, Cu)6(Fe, Cu) particles, also primarily known as GP zones with a diameter of 2 to 3.5 nm, which are electrochemically more cathodic in nature than the matrix and therefore lead to dissolution of matrix around the periphery of the precipitates [26]. In addition to this, further development of ή particles (MgZn2 precipitates) also occurs at the grain boundaries during the T6 aging process. Contrary to GP zones which form within the matrix, the ή particles form at the grain boundary. Since they are more anodic than the matrix, their formation promotes intergranular corrosion [27].
The present work is aimed to evaluate the electrochemical corrosion and pitting behavior of the Al7075-T6 aluminum alloy as the function of variations in NaCl concentration and the pH of the testing media. 3.5% NaCl and 1.75% NaCl concentrations were used to prepare the electrolytes solution and the pH values were adjusted at 2, 7 and 12 by using respective buffer solutions.
2. Experimental
2.1 Sample Preparation
From Al-7075 T6 alloy the specimens of size 10 mm×10 mm×3 mm were cut via electric discharge machine (EDM) followed by washing & cleaning. Then, the samples were cold mounted for experimentation with epoxy resin so that the total exposed area was 1 cm2 for all the samples. The samples were then grinded up to 4000 grit silicon carbide paper followed by fine polishing with 1 μm alumina paste. To prevent oxidation, the samples were stored in desiccator. Samples were rinsed with distilled water after each stage of grinding and polishing.
2.2 Solution Preparation
All the experiments were performed in 1.75% and 3.5% NaCl (Sigma Aldrich) aqueous solutions. The pH of the electrolyte solutions was varied from 2 to 12 using hydrochloric acid and sodium hydroxide (Sigma Aldrich) solutions both 1 molar in concentration and was controlled using a Milwaukee pH 55 pocket Size pH/Temperature meter which was dual calibrated with pH 7.01 and pH 4.01 standardized buffers. The solution volume was kept constant at 150 mL for all the samples.
2.3 Weight Loss Measurements
The immersion tests (10 days of immersion time) on Al7075 T6 alloy samples were performed in 1.75% NaCl and 3.5% NaCl at 3 different pH 2, 7 and 12. The weight measurements before and after the immersion were done using a Shimadzu analytical balance in which each sample was weighed prior to immersion. The post immersion weight measurements on samples were done after carefully removing the scale by submersing in 67% nitric acid for 2.5 minutes followed by thoroughly rinsing in distilled water and drying [28].
2.4 Electrochemical Measurements
The electrochemical measurements were performed by using G750 Potentiostat by Gamry USA. A silver/silver chloride electrode was used as reference electrode and graphite electrode was used as counter electrode. A constant solution volume of 150 mL was used for each test. After three hours of scanning at open circuit potential (OCP), the potentiodynamic polarization measurements were done at a scan rate of 1 mV/s between −0.5 V to 1.5 V of potential vs. OCP. For cyclic polarization experiments, both forward and reverse scans were set at 1 mV/s and the initial voltage was set at −0.5V vs. the OCP. The scans were set to reverse at 0 V and terminated at −1 V vs. OCP. For electrochemical impedance spectroscopy, again the OCP scans were run for 3 hours followed by Nyquist/Bode scans in the frequency range of 100 KHz to 10 MHz. All the experimentation was performed at ambient temperature conditions.
2.5 Microscopy Analysis
The microscopic studies on corroded surfaces were carried out using an Olympus optical microscope and a scanning electron microscope by Philips (XL-30). Analysis of the crystal structure was carried using Philips PW3710 X-ray diffractometer.
3. Results and Discussion
3.1 Weight Loss Measurements (Effect of pH and Concentration)
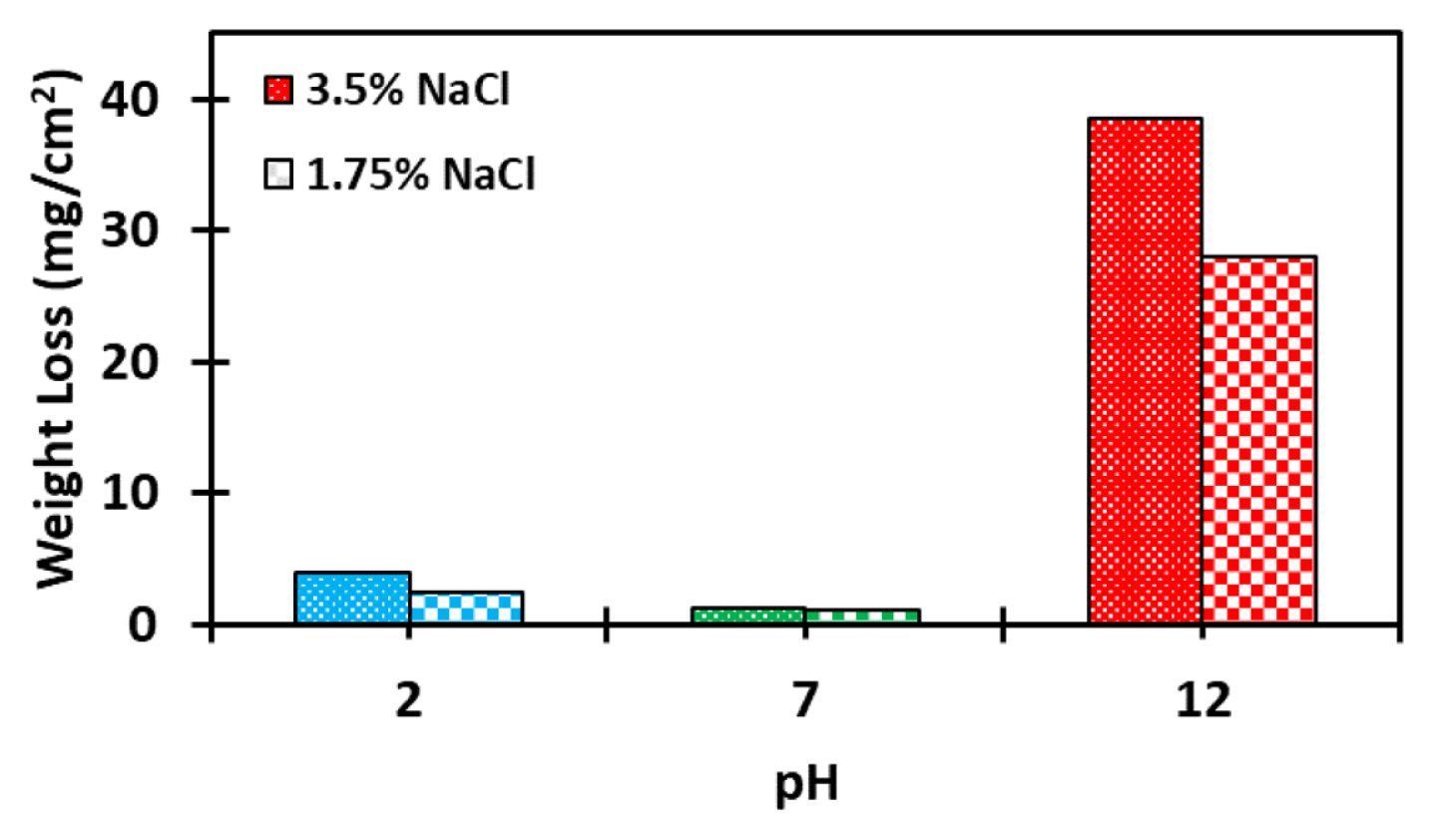
The weight losses of Al7075 T6 alloy specimens after immersing for 10 days in the electrolyte solutions at different pH values, are presented in Fig. 1. The results depict that samples submerged in neutral solution (pH=7) have least susceptibility to corrosion, exhibiting least weight loss after immersion. But the corrosion rate increases as the pH values shift to acidic or alkaline side indicating increased susceptibility of the alloy to corrosion under acidic and alkaline pH. This is in accordance with the previous studies revealing that aluminum and its alloys are amphoteric in terms of their reactivity and they react with both alkaline and acidic media and at higher rates [29,30]. This can be further explained by the mechanism of reactions occurring in NaOH solution when reacted with water. The cathodic reaction is:
Whereas the anodic reaction is:
The overall reaction is given as:
The reaction mechanism clearly shows the dissolution of water to produce H2 gas which can be seen in the form of effervescent on the surface of the aluminum sample. This signified high turbulence of the exposed surfaced due to more rigorous gas evolution and resulted in higher degree of pitting formation. As a result of the occurring turbulence, the product layer could not stabilize thereby increasing the weight loss of the alloy in the sample [31].
In neutral environment, the surface oxidized uniformly in conjunction with Cl− attack which is minimal and hence the oxide layer remains stable on the alloy resulting in minimal weight loss. The acidic environment is succeeded by high concentration of Cl− attack which is facilitated by the high degree of reactivity of the Al3+ ions from the matrix of the alloy. However, in comparison to a weight loss of neutral media in 3.5% NaCl of 1.31 mg/cm2, the weight loss for pH 2 solution is 4 mg/cm2, an increase of 3 times observed but in alkaline solution the weight loss is 38.56 mg/cm2. This substantial increase in comparison to the neutral media is owing to the similarity of the nature of the attack on the alloy in the pH 12 solution. This is in conjunction with the open circuit potential analysis at 3.5% NaCl which shows that the stabilization time of the alloy in the pH 12 solution is the longest owing to the relatively high concentration of OH− ions. This extends the equilibrium time by steady dissolution of Al3+ ions via oxide formation due to strong nature of attack of OH− ions, hence resulting in maximum weight loss [32,33].
With lowered concentration, the weight loss decreases owing to primarily the reduction in the concentration of the Cl− ions. This reduces the selective attacking (pitting) on the alloy hence corrosion mechanism in all three pH conditions is driven more by relatively more uniform dissolution with increased concentration of OH− ions [11,34] as can be observed from Fig. 2.
3.2 Effect of pH and Concentration on OCP
The OCP vs. time plots as the function of pH and NaCl concentration are given in Fig. 2. The variation in pH of the solution causes a shift in OCP to more noble values of the Al-7075 T6 alloy because of stable passive oxide layer formation and the solution stabilizes after some finite amount of time. On comparison, as shown in Fig. 2 (a), samples tested in solutions of pH 2 and pH 7 had a close, similar potential of approximately −785 mV whereas the sample tested in solution of alkaline pH 12 had a relatively more negative OCP of −1146 mV.
The stabilization of the OCP also depends on the composition of the electrolyte itself [35]. Previous research has identified the pattern that the time to stabilization of the OCP decreases with the concentration of the corrosive species in the electrolyte, which in turn effects the formation of the passive of oxide layer on the surface, preventing further corrosion [28]. It is observed that lower concentration of the Cl− in the solution shifts the potential to more anodic values, as depicted in Fig. 2 (b), and leads to early stabilization of the system as less aggressive interaction and dissolution of Al3+ ions occurs [36].
3.3 Potentiodynamic polarization behavior as a function of pH and NaCl concentration
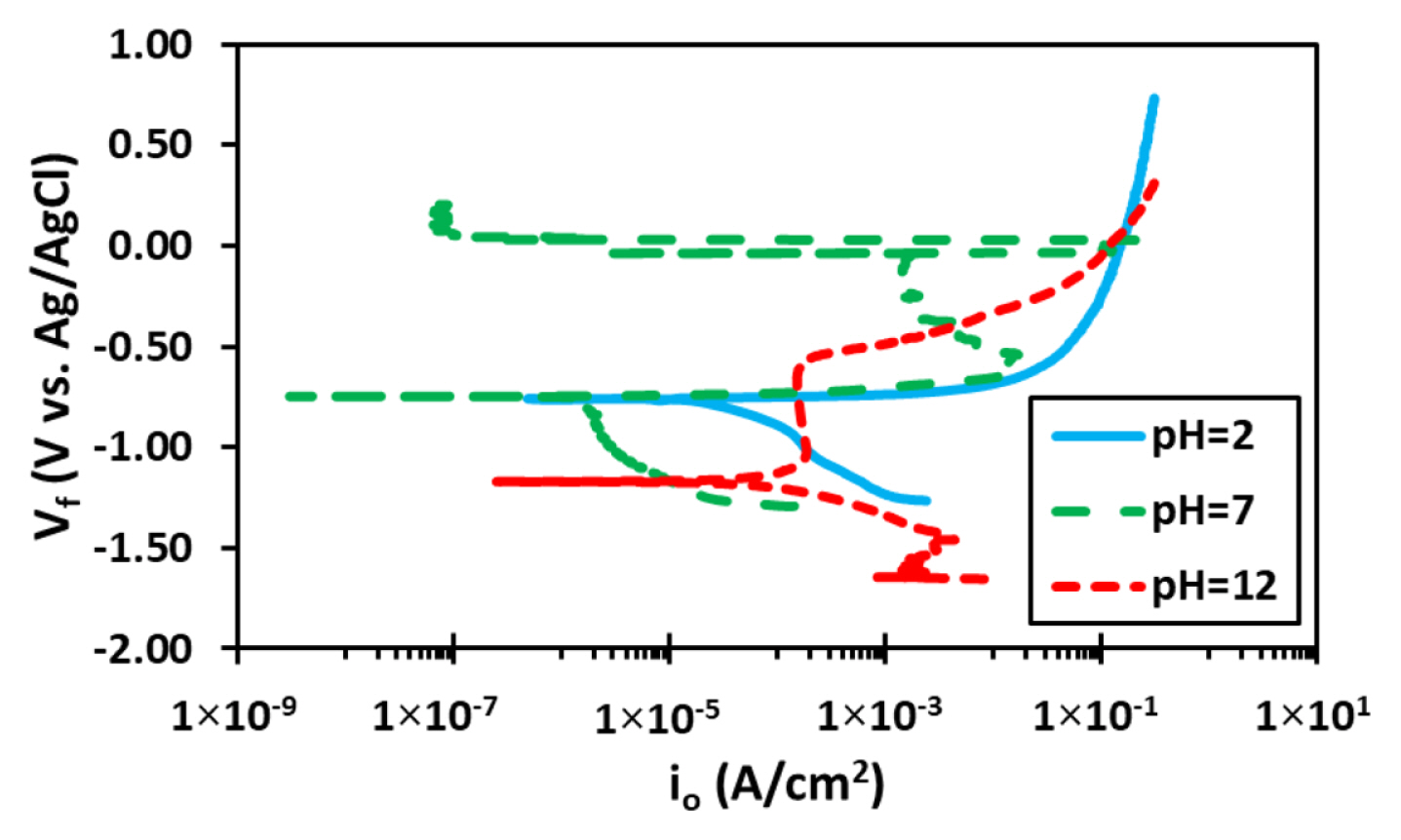
Fig. 3 shows the plot of potentiodynamic polarization results of the samples performed in 3.5% NaCl solution in ambient conditions at varying pH. The quantitative results, as evaluated by Tafel extrapolation method, are presented in Table 2.
The analysis of the curves is in conjunction with the weight loss experiment performed by immersion testing presented and discussed in previous section. The results show similar behavior for the alloys experimented in the neutral and acidic solutions with the cathodic behavior being nearly constant for both environments albeit the lowest cathodic current density was observed for neutral solution. The Ecorr values are similar in magnitude for solution tested at pH 2 and 7 with major difference being the anodic plot of the curves. The anodic current density for acidic condition was 2.16×10−5 A/cm2 whereas for the neutral solution it was 1.08×10−6 A/cm2, an increase in 20 times when the solution conditions become acidic [36,37].
Additionally, results show that the onset of pitting potential is inseparable from the Ecorr underlying the poor stability of the oxide film as discussed earlier. The resulting anodic plot of the neutral and alkaline highlights that pitting occurs at Ecorr hence both Epit and Ecorr can be considered equal since the anodic slope is too low for practical purposes. Hence, over this pH range little variation in pitting behavior is observed in the electrolyte solutions [22,38].
However, for the samples performed in alkaline solution the result is contrastingly different from neutral or acidic pH. In alkaline solution, owing to the higher concentration of the OH− ions, the uniform dissolution of oxide layer occurs under electrochemical attack [39,40]. Since the differential of the OH− concentration is extensively large, the anodic dissolution range increases which shifts the corrosion potential to more anodic domain [33]. This suggests more stable passivation behavior due to prolonged time for dilution of OH− concentration in the solution. It also shows reduction in the rate of uniform corrosion and favors selective pitting behavior which does not overlap with the Ecorr of the sample. The Ecorr for the alkaline condition alloy was approximately −1170 mV whereas the pitting potential was at −613 mV and pitting potential shifts to more anodic region highlighting a delay in pitting formation in comparison to the samples tested in neutral and acidic solution. This can theoretically be due to the lack of stable film formation under these pH conditions and more aggressive OH− ions attack that develops the passive film formation. This eventually delays the ion transport of Al3+ cations from the surface to the exterior of the coating itself [41]. Cathodic slope was found to be maximum for the neutral solution conversely the lowest cathodic current density was observed for alloy samples tested in neutral solution [42]. Additionally, the cathodic curve shows a sharp behavior towards diffusion-controlled phenomena for the pH 12 with fluctuating value of current density. This could be a result of metastable pitting as cathodic over potential provides necessary energy for re-passivation of such pits formed [43]. The corrosion current density was changing relative to pH in the order of pH 7 < pH 2 < pH 12 as perceived from the previous results. Additionally, the passivation potential increased to more positive (anodic) region with decrease in pH implying that the pH reduction lowers the stabilization potential of the passive layer formation due to the influence of the Cl− and OH− ions in the solutions which affect the stability of adsorption of Al3+ ions on the surface of the polarized sample.
Furthermore, the effect of Cl− concentration was evaluated at different pH on the corrosion behavior of the alloys. It can be seen from the Fig. 4 that with the reduction in chloride concentration, the corrosion potential shifts to more noble direction. This is markedly due to the relative increase in the ratio of the hydroxyl ions to account for film formation. Also the severity of Cl− attack effectively results in two phenomena: (1) the lower concentration of Cl− ions lowers the severity of pitting on the surface of the alloys as seen from the comparison of the acidic and neutral solutions and delays it more anodic potential due to the delayed response in pitting and corrosion initiation [9,44], (2) there is no distinction between the Ecorr and Epit with change in Cl− concentration so passive film formation is not stabilized to higher potential rather they overlap with the Ecorr so that the pitting behavior has no effect with change in concentration [9,45].

The potentiodynamic polarization plots as a function of NaCl concentration at (a) pH= 2 and (b) pH =7
Specifically, in the case of acidic solution (Fig. 4(a)), the corrosion rate is inhibited such that the slope corresponding to sharp increase in corrosion current density with voltage recedes to a lower range. This is due to the reason that Δio was measured to be 3.17 μA/cm2 for a NaCl concentration of 1.75% as opposed to 33.83 mA/cm2 for the 3.5% NaCl solution for acidic condition and from 16.39 mA/cm2 for 3.5% NaCl to 62.24 μA/cm2 for the 1.75% NaCl solution.
This shows that the effect of pH variation also offsets the Cl− concentration of the electrolyte in terms of the dynamics of pitting, corrosion mechanism and its rate. This is because the degree of reactivity of the oxidizing/corrosive species is dependent on the diffusivity of the Al3+ ions through the corroded layer on the surface directly to the electrolyte whose morphology is effected by the pH conditions [46]. As such the corrosion current density increases with increasing concentration for the same pH i.e. Icorr (3.5% NaCl) > Icorr (1.75% NaCl). The results have also been compared with a basic pourbaix diagram for aluminum. It can be seen from Fig. 7 that the E-pH diagram for the aluminum metal takes four species into consideration, namely: Al, Al2O3. H2O, Al3+, and AlO2−. Furthermore, the concentration is taken at the boundary of 1×10−6 mol/kg, the temperature for this diagram is taken at 25°C which is indicative of room temperature, the same at which the corrosion experiments were performed in the current study. The corrosion potential mainly lies in and around the cathodic protection regions for current work alloy samples. This shows that passive layer breakdown does not lead to pitting in neutral and acidic conditions as verified from the graphs in Fig. 3 and Fig. 4 as well.
3.4 Cyclic Polarization behavior as a function of pH and NaCl Concentration
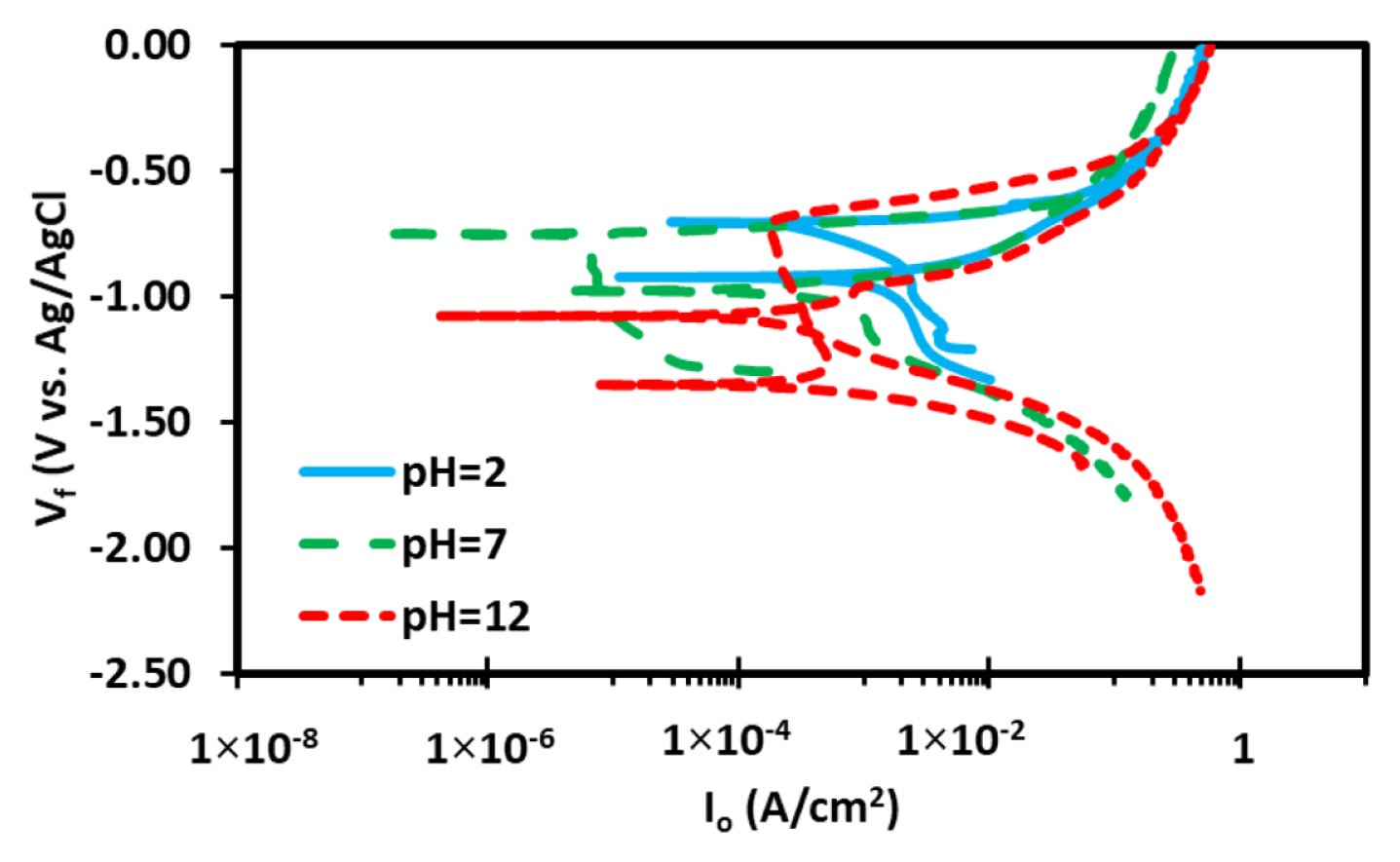
Fig. 5 shows the cyclic polarization curves performed in 3.5% NaCl at varying pH values. The purpose of the cyclic polarization is to evaluate the pitting behavior as a function of both pH and Cl− concentration of the electrolytes to evaluate the degree of both pitting and re-passivation behavior.
The analysis of the forward scan and reverse scan in conjunction with loop analysis provide an accurate description (schematically) of the pitting behavior and re-passivation of the surface in the corrosive species. For comparison purposes, the analysis of both acidic and neutral medium was carried out prior to alkaline medium as discussed in the previous sections.
Previous research explains the behavior of the reverse scan from apex potential such that the hysteresis loop and direction of reverse scan are main contributor to pitting analysis and that the degree of pitting is influenced by the loop area and direction of the reverse scan [47]. From the Fig. 5, it is seen that in case of the sample tested in neutral solution that the pitting potential (Epitt) overlaps with the corrosion potential (Ecorr) after which the corrosion current density increases sharply till it reaches the value of 1.0×10−1 A/cm2 and then increases slowly [14,48]. Additionally, upon reversing the scan from the Apex potential of 0V, the corrosion current density increases which indicates the formation of pitting in the sample. The reverse scan coincides with the forward scan to give an Erp of −977 mV, which is below the Epit (−750 mV) of the sample. This indicates that in neutral sample re-passivation behavior is improbable as the re-passivation potential is lower than the pitting potential. With regards to the acidic conditions, it can be seen that the cathodic and anodic portion of the curve and reverse scan have similar behavior pattern as that of the neutral solution. The reverse scan shows increase in corrosion density, the same occurrence for likelihood of pitting and loop area indicates pitting formation in the sample. The repassivation potential, Erp of −922 mV is lower than the Epit (−702 mV) hence the over-potential of the acidic sample being −220 mV which is approximately that of the neutral sample (−227 mV).
The case of alkaline condition in 3.5% NaCl showed a deviancy form the acidic and neutral behavior. This is because the pitting potential is independent of the corrosion potential as the formation of classic passivation region is observed to start from −1280 mV when the current density is at 501 mA/cm2 and the pitting initiates at −700 mV. The reverse scan shows increase in current density with exceptionally larger loop formation hence the previous results support the lager degree of pitting in alkaline environment. The exception here is that Erp of the sample is more anodic (−1050 mV) which signifies higher repassivation in alkaline environment [49,50].
For the pH=2 in Fig. 6(a) that the pitting which is weakly influenced by the Cl− of the NaCl addition reduces such that the reverse scan shows a negative hysteresis which signifies little to no pitting in the sample rather uniform thinning of the sample follows anodic polarization and film formation stabilizes on surface. The resultant oxide stable film accounts for Erp being larger than Ecorr of the sample [12].
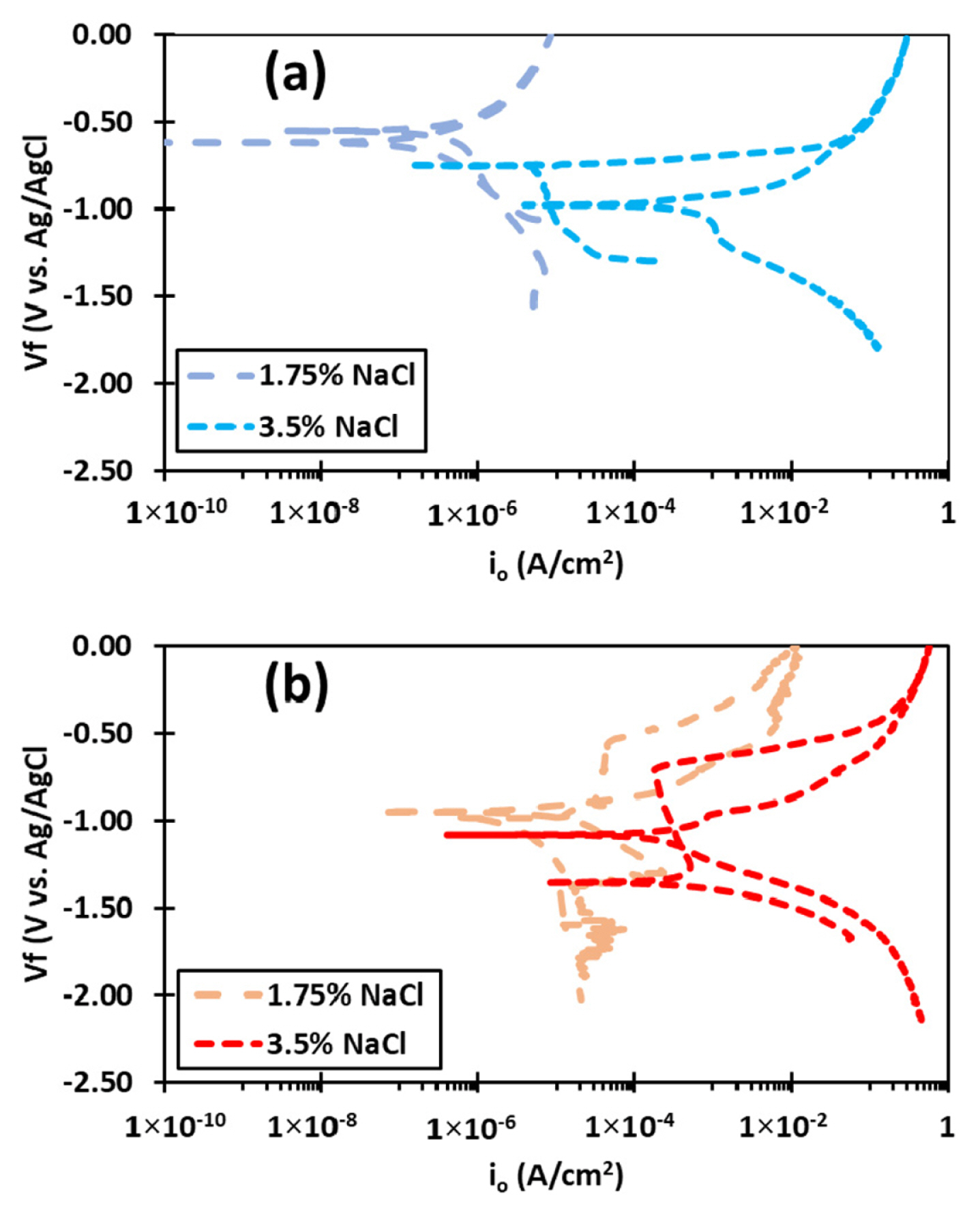
Variation in cyclic polarization behavior as a function of NaCl concentration at (a) pH=2 and (b) pH=12
In case of the alkaline pH (Fig. 6(b)), it is seen that the corrosion current density shifts to relatively noble direction, indicating less severity of corrosion and the curve shifts to more cathodic direction relative to the 3.5% NaCl highlighting the reduced severity of the uniform dissolution as a result of Cl− reduction. This, however, results in reduced stability of passivation on the surface as pitting breakdown occurs in relatively lower potential range and the area of the hysteresis loop in the reverse scan signifies greater impact of OH− based pitting attack. In quantitative terms, Erp (−946 mV) recedes closer to the Ecorr (−974 mV) [51].
The resultant reactions occurring during the anodic polarization process were compared to the reactions documented in a Pourbaix diagram of aluminum metal since it is the primary constituent of this alloy [52]. The result shows that passivation breakdown results observed in current study comply with the boundary condition set in the diagram. The cyclic polarization curve shows that the re-passivation potential lies in the same domain as the immunity region or the cathodic protection range if the values are translated to silver/silver chloride reference electrode. This shows that re-passivation potential of the alloy samples occurs below the cathodic protection region while pitting occurs above it in the corrosion domain.
3.5 Electrochemical Impedance Spectroscopy Analysis
The EIS curves are shown in Fig. 8 (Bode plots) and Fig. 9 (Nyquist plots) which indicate that the solution resistance is less prominent in the case of acidic (pH=2) and alkaline (pH=12) media. This can be realized from the inductor elements in the circuit model (Fig. 10 (a)) and the negative inductive loop in the Nyquist plot (Fig. 9). This indicates adsorption and desorption characteristics of the Al3+ ions in the media which is owing to the greater flux of Cl− ions, thus providing stronger dissociation behavior of ions from the surface which was further complicated by the inhomogeneous crystallographic pitting and corrosion product formation. This effectively increased the surface area in contact with the solution thus promoting faster diffusion of Cl− ions. The NaCl addition promoted the stable coating formation which was effectively suppressed by the strong acidic ions flux on the surface. However, this promoted greater stability of the double layer which showed greater resistance at lower concentration [53].
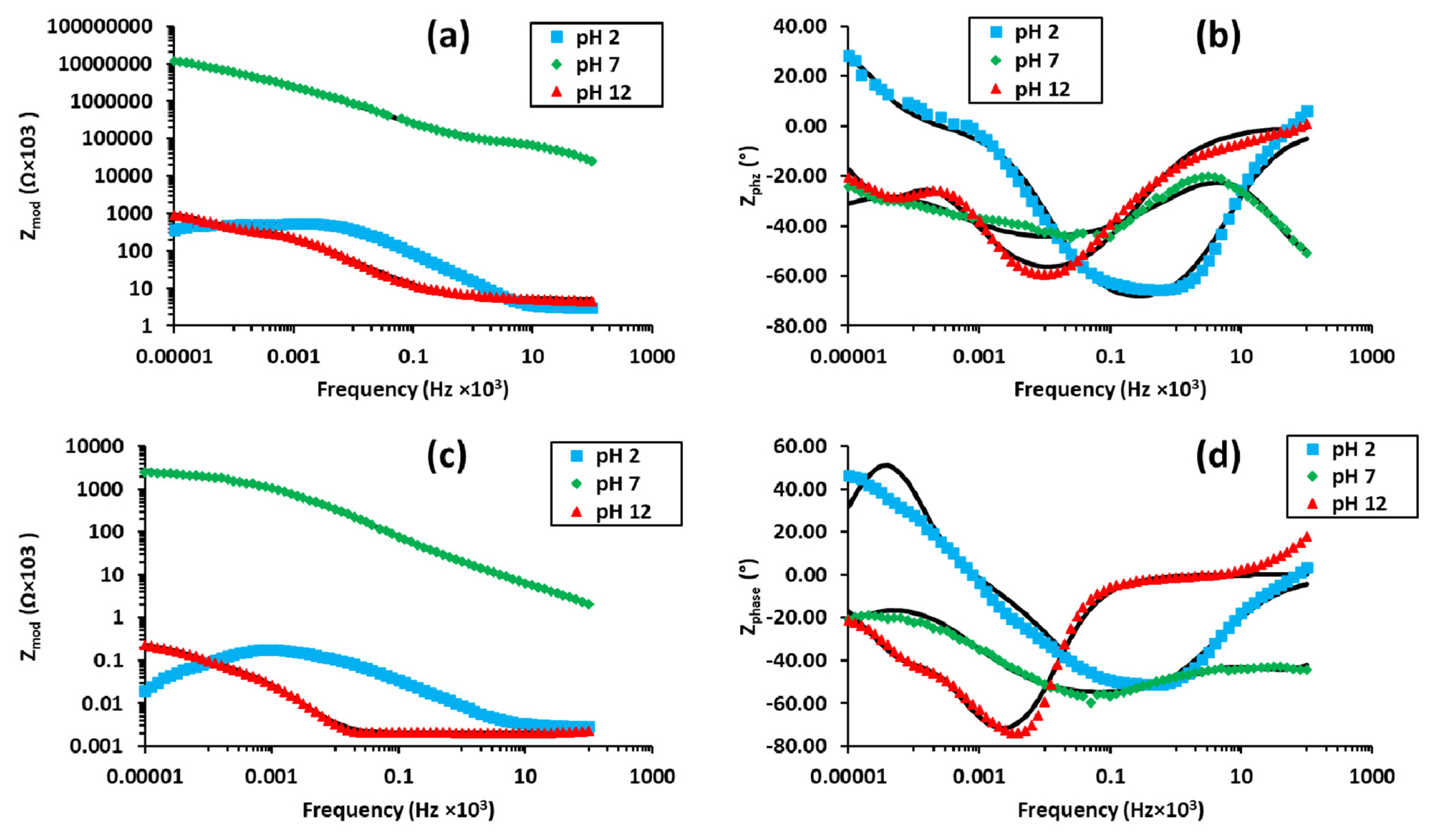
Bode plots as the function of pH in 3.5% NaCl solution (a) Impedance, (b) phase; and in 1.75% NaCl solution (c) Impedance (d) Phase.
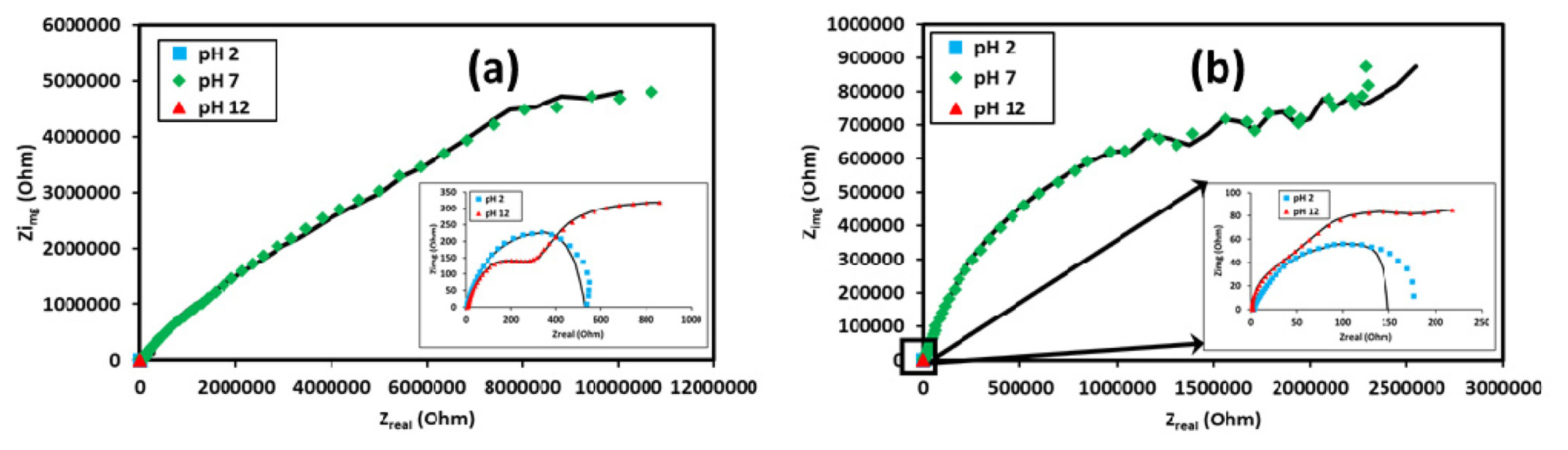
Nyquist plots as the function of pH for (a) 3.5% NaCl concentration and, (b) 1.75% NaCl. concentration
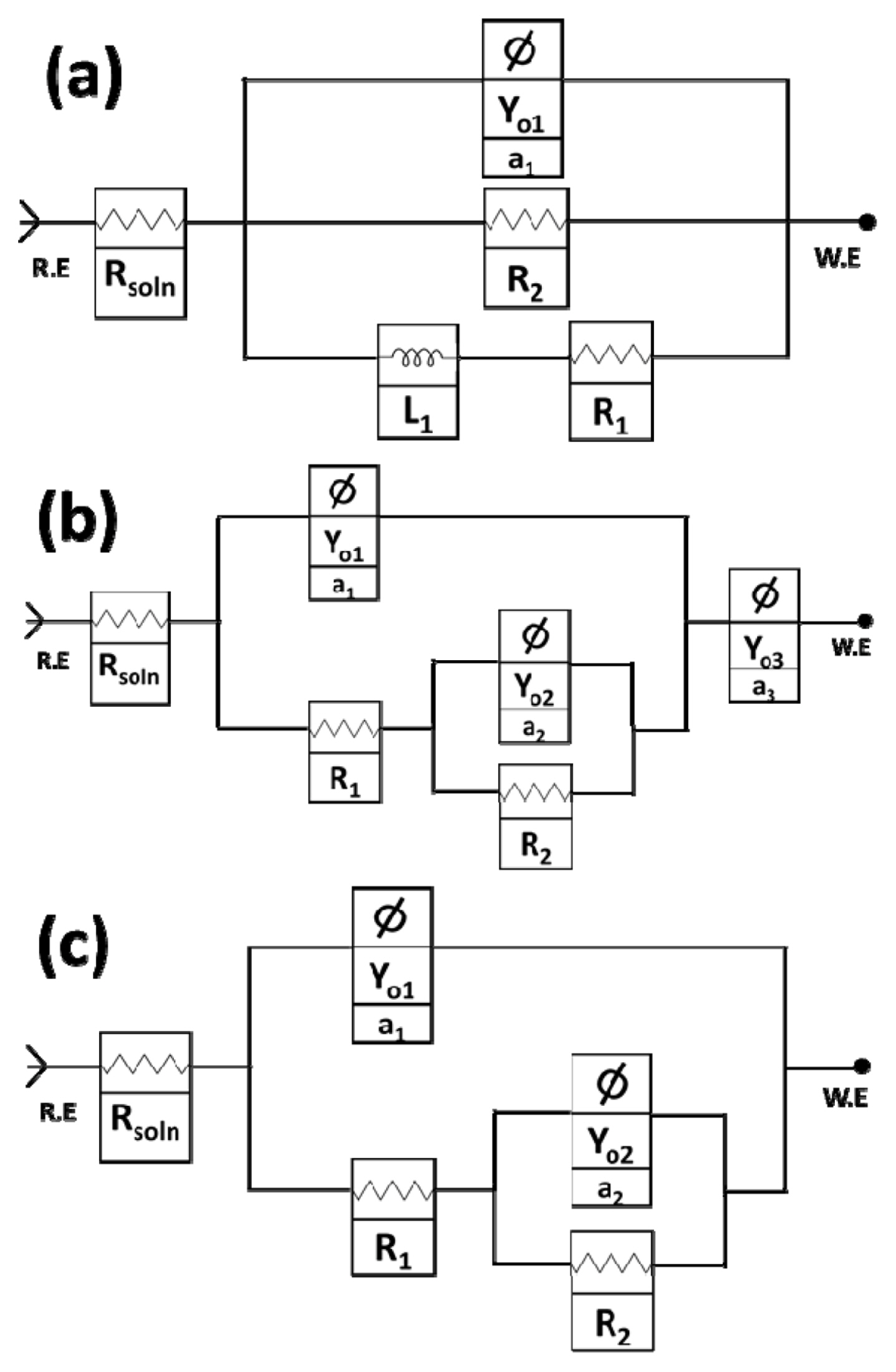
Equivalent circuit models for fitted curves for both concentrations of NaCl (1.75% and 3.5%) at (a) pH-2, (b) pH=7 and (c) pH=12
In the neutral solution, the coating stability was found to improve with decrease in concentration of Cl− ions in the solution (Table 3) in addition to increase in solution resistance [54]. This can be explained by analyzing the constant phase element associated with the double layer formation which indicated that at the lower concentration of NaCl the oxide coating formed has higher resistance thus giving the neutral solution a comparatively much larger loop formation in the Nyquist plot analysis as shown in Fig. 9 (a& b)[39,55].
In the alkaline environment, the EIS results are in accordance with the data obtained in polarization tests. Initially, a passive film is formed on the surface as a result of the interaction with the electrolyte solutions. However, preferential dissolution occurs due to the localized nature of attack of OH− ions in the electrolyte solution when there is decrease in NaCl concentration to 1.75% from 3.5% (Fig. 8). Actually, there is observed a near 10 times decrease in resistance of the passive film of the alloy, but the double layer relatively stabilizes with the decrease in flux of hydroxyl ions on the surface. The constant phase elements (CPE) indicate lowered resistance to ion transfer and collapse of capacitive nature of the double layer and the coating on the surface. Furthermore, the porous oxide or mud cake structure formed in the cyclic polarization test exhibits much higher resistance of magnitude (10 folds). This accompanied by constant phase elements of Al and Zn as major constituents of the alloy [18] which indicates the facilitation of ion transport to the surface in localized regions thereby accelerating the pitting phenomena [42].
Comparatively speaking, the inductive loop in the acidic environment exists because of the minimum polarization resistance observed for the samples treated in the acidic environment (Fig. 10c). It is determined from the Nyquist plot analysis that a high frequency coating loop with very low resistance is formed on the surface. The underlying value of n is slightly less than one in case of the constant phase element is due to capacitive nature of the coating formed albeit with pitting tendency destabilizing it. In addition, the resorption characteristics of the Al3+ ions on the surface are evaluated by the presence of the low frequency inductor element.
The aspect of strong ions discharge and resorption is not imminent in the neutral and the alkaline solution owing to the greater polarization resistance at the surface. This is owing to the release and adsorption of Al3+ ions on the surface of the alloy. In the acidic conditions, the passive layer formation is less favored here due to low stability of passive layer until higher anodization potentials favor oxide formation instead of the Cl− based species on the surface. Furthermore, we see that the number of constant phase elements act linearly to the corrosion behavior of the alloy in the environments (Fig. 8 (b) & (d)). This corresponds from the fact that in pH= 2 there is only one Constant Phase element (Yo1) whereas two for pH=12 and three for pH=7 tested environments. So the phase elements, particularly in the case of pH=12, have highly resistive nature with an>0.5 but the high concentration of OH− ions in the alkaline environment allows for stable oxide layer to grow which provide resistance which is offset by the capacitive nature of the porous coating (R1) and that free flow of oxide ions occurs to the underlying matrix thus offering lower charge transfer resistance (R2) as compared to the pH=7 environment.
Additionally, the trend of the results obtained lies in parallel to the results obtained with potentiodynamic polarization. The charge transfer (R1), passive layer (R2) and polarization resistance (Rp) showed highest value for samples analyzed under neutral conditions for both, 3.5% NaCl and 1.75% NaCl concentration. Furthermore, the resistance values obtained from model simulation showed they were considerably less than that under neutral conditions. However, a slightly keen observation showed that R1, R2 and Rp was comparatively lower for tests under acidic environment than alkaline conditions. An underlying reason for this is that EIS determines surface conditions under varying potential. As alkaline solution lead to strong passive layer formation whereas acidic condition did not favor the formation of continuous layer, the circuit model for acidic environment showed parallel connections for R1 and R2 elements signifying weak interface resistance and that they overlap. Therefore, the EIS showed slightly higher values of resistance for alkaline conditions as opposed to acidic conditions
3.6 Surface Examination via Microscopy
Surface examination of the tested samples showed consistent corrosion product and pitting formation. Fig. 11 which shows the micrographs of the specimens after the immersion tests which represents the 3.5% NaCl (Fig. 11 (a, b &c)) and in 1.75% NaCl (Fig. 11 (d, e &f)), the formation of pits and oxide corrosion products occurs. In addition, fine corrosion morphology around the precipitates and that the severity of the pit formation increases with shift in pH from neutral values [10,36].

Micrographs of the corroded surfaces after immersion tests in 3.5% NaCl solution at (a) pH=2, (b) pH=7, (c) pH=12 and in 1.75% NaCl at (d) pH=2, (e) pH=7 and (f) pH=12.
Additionally, immersion tests show large degree of localized pits around the periphery of the precipitates in the matrix, indicating that the presence of these intermetallic phases have contributed to the aggravation of pitting behavior in the alloy and that deep circular pits were found to be observed in the polarized and immersion tested samples [56,57]. Furthermore, from the potentiodynamic polarization scans, it was observed that the immersion test provide differentiation in terms of the pitting and corroded surface (Fig. 12). The results show that the variation in pH provides change in the degree of pitting occurrence in the solution though variation in pitting morphology was not observed. Furthermore, potentiodynamic polarization tested samples show effective degree of polarization however excess degree of corrosion can be seen in the alkaline solution with the high volume of corrosion product formed (Fig. 12(a) and (b)).

Micrographs of the samples after the potentiodynamic polarization tests in 3.5% NaCl solution at (a) pH=2, (b) pH=7, (c) pH=12 and in1.75% NaCl solution at (d) pH=7
The excess degree of pitting occurrence that was seen in cyclic polarization results confirmed the susceptibility of the Al-7075 T-6 alloy to the higher pitting behavior influenced by the presence of the intermetallic phases (precipitates). These phases acted as cathodic sites thus causing selective dissolution of the matrix phase around the periphery of the second phase particles (Fig. 13 (c)). This preferential dissolution precedes severe inter-granular corrosion owing to the presence of precipitates at the grain boundaries.

Micrographs of the corroded surfaces after the cyclic polarization tests in 3.5% NaCl Solution at (a) pH=2, pH=7, (c) pH=12 and in1.75% NaCl solution at (d) pH=7
A major difference can be observed in the morphology of the corrosion product in the immersion sample and polarized one. The corrosion product morphology is more crystalline in nature than the one in immersion which is defined as hemispherical corrosion [58]. The corrosion product is crystallographic in nature owing to the polarization which results in the reactive species being produced in unique planar orientation which is possessed by the grain(s) of that region (Fig. 14). This is because the electrical conductivity is intrinsically a function of planar orientation so certain grains provide greater conductivity to current. Furthermore, the optical microscopy shows for the cyclic polarization that the neutral and alkaline samples tested in 3.5% NaCl solution have a thicker layer of corrosion product and recovery reminiscent of the mud cake structure which showed recovery of the corroded surface [59]. The polycrystalline orientation of the alloy allows for selective or concentrated regions of preferable corrosion. A conclusive observation from microscopy reveals that optical microscopy technique only accounts for showing visible intergranular corrosion as a result of low magnification and depth of focus associated with optical microscopes [28,36]. Furthermore, we can conclude here that the degree of pitting and corrosion of sample transcends across grains so many regions of intergranular corrosion are overlapped with aggravated pitting or trans-granular corrosion [11,31,39]. What makes them further indistinguishable is the uneven corrosion product formation due to destructive interference of reflected light from the sample.
4. Conclusions
Al-7075-T6 alloy was investigated via electrochemical testing to determine its corrosion resistance by immersion corrosion testing and electrochemical corrosion testing. The composition of the solutions as altered by the variation of pH, effected the electrochemical behavior of this alloy. The high degree of OH− species in the alkaline solution leads to larger degree of pitting in the alloy, as evaluated by cyclic polarization, which is facilitated by the presence of AlCuFe type precipitates that act as cathodic sites in the matrix. The onset of the corrosion is accelerated with the higher concentration of the oxidizing species as seen from the larger hysteresis loop. This generates corrosion product around the periphery of the precipitates and the least weight loss occurred in the neutral conditions which is reflective of the degree of the oxidizing species that attack the periphery of the precipitates. The cyclic polarization reflects on the ability for re-passivation of the alloy which is achieved on a higher potential than the corrosion potential in alkaline condition as compared to the neutral and acidic environments. The morphology of the tested solutions indicated that the onset of corrosion is facilitated by passivation breakdown that aggressively enhances the morphological degradation towards inter-granular corrosion. This was confirmed by the EIS plots indicating capacitive element for the alkaline solution, inductance for the acidic solution and the adherent coating formation in the neutral solution.