3D Hierarchical Flower-Like Cobalt Ferrite Nanoclusters-Decorated Cotton Carbon Fiber anode with Improved Lithium Storage Performance
Article information
Abstract
The inverse spinel Cobalt ferrite (CoFe2O4, CFO) is considered to be a promising alternative to commercial graphite anodes for lithium ion batteries (LIBs). However, the further development of CFO is limited by its unstable structure during battery cycling and low electrical conductivity. In an effort to address the challenge, we construct three-dimensional hierarchical flower-like CFO nanoclusters (CFO NCs)-decorated carbonized cotton carbon fiber (CFO NCs/CCF) composite. This structure is consisted of microfibers and nanoflower cluster composited of CFO nanoparticle, in which CCF can be used as a long-range conductive matrix, while flower-like CFO NCs can provide abundant active sites, large electrode/electrolyte interface, short lithium ion diffusion path, and alleviated structural stress. As anode materials in LIBs, the flower-like CFO NCs/CCF exhibits excellent electrochemical performance. After 100 cycles at a current density of 0.3 A g−1, the CFO NCs/CCF delivers a discharge/charge capacity of 1008/990 mAh g−1. Even at a high current density of 15 A g−1, it still maintains a charge/discharge capacity of 362/361 mAh g−1.
1. Introduction
In the past few decades, energy storage technology has been improved due to the demand of various forms of energy. Many innovative energy storage devices such as flow batteries [1–3], lithium ion batteries (LIBs) [4–6], and lithium-sulfur batteries [7] have been developed. Among them, LIBs have been widely utilized as power sources in practical fields due to their high operated voltage, long service life, high energy density and design flexibility, etc. [8]. However, the commercial LIBs are gradually unable to meet the requirement of practical applications. One of the important reasons is that the commercial anode material for LIBs (i.e. graphite) has lower capacity (372 mAh g−1). Therefore, carbonaceous materials, transition metal oxides (TMOs) [9], transition metal nitrides (TMNs) [10], transition metal disulfides (TMSs) [11] and transition metal phosphide (TMPs) [12] and many others have been explored to seek high capacity. In the last few years, ternary transition metal oxides (TTMOs) have attracted researchers’ attention, due to their elevated reversible capacity, improved structure stability and enhanced electronic conductivity compared with TMOs [13]. In addition, TTMOs also show significant advantages with respect to economic and environmental aspects. Nevertheless, TTMOs still exhibit unsatisfying rate performance and rapid capacity degradation during cycling resulted from low conductivity and serious volume change during Li+ de/intercalation.
To date, many studies have been focusing on nanosized active materials with rational designed morphologies, e.g. multi-shell hollow nanospheres [14], platelets [15], nanofibers [16], nano-octahedron [17], york-shell [18], nanoclusters [19], etc. and hybrid architectures, which are mainly composited with nanocarbons, i.e. carbon coated film [20], graphene [14], carbon nanotube [21], etc. These approaches can alleviate structural strain during Li+ de/intercalation and enhance the charge transfer kinetic [22]. For examples, Qi et al. obtained controllable multi-shell cobalt ferrite nanospheres by controlling the calcination conditions of the precursor, which has a capacity retention rate of 88.9 % (at 1 A g−1) and 86.9% (at 5 A g−1) after 500 cycles [14]. Zhang et al. synthesized a network of interconnected ferrite nanospheres with carbon nanotubes, which exhibited excellent cycling performance due to the stable ion/electron transfer pathway and alleviated volume change provided by the network structure [21]. Recently, three-dimensional (3D) micro-architectures have drawn many attentions, which typically are composed of low-dimensional building blocks (such as 2D nanosheets). 3D micro-architectures demonstrates diverse advantages, such as short Li+ diffusion distance, large contact areas with electrolytes, as well as abundant active sites [23–25]. In addition, 3D micro-architectures can prevent self-aggregations of active materials, maintain the contact area with the electrolyte, as well as alleviate the volume stress induced by the electrochemical reaction [26]. Therefore, it is likely to boost the electrochemical performance of TTMOs by constructing a hierarchical hybrid structure composed of 3D micro-architecture assembled by low-dimensional nanostructured TTMOs on a carbon matrix. Biomass-derived carbon appears to be a desirable matrix to support nanostructured TTMOs [27], due to low cost and abundance of biomass, as well as the facile synthesis processes. Among biomass, cotton fibers are abundant, sustainable and green biomass carbon source in nature and could act as a matrix and provide a long-range electron transport pathway [28], eventually increasing the utilization of the active material.
Herein, we prepared a 3D hierarchical structure, carbonized cotton fibers decorated with flower-like CoFe2O4 nanoclusters (CFO NCs/CCF), by facilely mixing metal salt precursors and carbonized cotton fibers by stirring and subsequent calcination. In the CFO NCs/CCF, the nanoflower-like CFO NCs anchored on the surface of carbon fibers are assembled by nano-petals composed of nanoparticles. The CFO NCs/CCF exhibits an excellent cycle and high rate performance. In term of cycling performance, the CFO NCs/CCF exhibits a specific discharge/charge capacity was 1008/990 mAh g−1 after the 100th cycle at a current density of 0.3 A g−1. In term of rate performance, it displays a discharge/charge specific capacity of 362/361 mAh g−1 at a current density of 15 A g−1. The good electrochemical performance is ascribed to the synergistic effect of CCF and CFO NCs in the unique 3D hierarchical structure. On one hand, CCF can serve as a long-range conductive matrix to alleviate the inherent low conductivity of ferrite. On the other hand, CFO NCs can not only provide abundant active sites, short diffusion pathway for Li+, large contact interface between electrolyte and active materials, but also alleviate structural stress stemmed from electrochemical reactions.
2. Experimental approaches
2.1 Material preparations
All chemical reagents were analytical purity and not further purified.
2.1.1 Preparation of cotton carbon fiber (CCF)
2 g of absorbent cotton fibers (Jiangxi Huazhong Textile Co., Ltd., China) was ultrasonically washed in deionized water and absolute ethanol for 1 hour, and then dried in a vacuum oven at 80 °C for 12 hours. The washed absorbent cotton fibers were carbonized in an argon-containing vertical tube furnace at 800°C for 2 hours and the resulted product is designated as CCF.
2.1.2 Preparation of pure CoFe2O4 (p-CFO) and 3D hierarchical flower-like CoFe2O4 nanoclusters/CCF (CFO NCs/CCF) composite
100 mg of CCF was dispersed into a methanol ((Aladdin Industrial Corporation, China) solution containing 0.5 mmol of Co(NO3)2×6H2O (Aladdin Industrial Corporation, China) and 1 mmol of Fe(NO3)39H2O (Aladdin Industrial Corporation, China) and stirred at 30°C for 12 hours. The remaining liquid was then evaporated in a vacuum oven at 30°C to obtain a precursor, which subsequently was calcined at 550°C for 2 hours in an argon-filled vertical tube furnace to obtain CFO NCs/CCF. The synthesis of p-CFO was consistent with the above process except that no CCF powder was added.
2.2 Structural characterization
The crystal structure of as-prepared material was obtained by the powder X-ray diffraction (XRD, D/max-2400, Rigaku) with Cu Ka radiation (l = 1.5418 Å) at 40 kV and 100 mA. Morphology observation was performed on scanning electron microscope (SEM, SIGMA HD, ZEISS) with an energy dispersive X-Ray spectrometer (EDS Inca X-Max, Oxford Instruments). Microstructure was analyzed by transmission electron microscope (TEM, JEM-1200EX, JEOL). The Raman spectra of the sample were obtained on a microscopic confocal Raman spectrometer (LabRAM HR Evolution, HORIBA Jobin Yvon). The surface area and porosity of the samples were obtained from the nitrogen adsorption/desorption isotherm using an automatic specific surface and porosity analyzer (MICROMERITICS ASAP 2460).
2.3 Electrochemical characterizations
Electrochemical performance measurements were carried out using CR2032 coin-type cells. The active material (CCF, p-CFO, or CFO NCs/CCF), the conductive agent (Acetylene black), and the binder (Polyvinylideneuf fluoride, PVDF) were placed in an agate mortar with a mass ratio of 7: 2: 1, and ground with a solvent of N-methylpyrrolidone (NMP) to form a homogeneous slurry. The slurry was coated on a copper foil, and dried in a vacuum oven at 80°C for 12 hours. The coated copper foil was cut with a slicer to obtain working electrode discs (about 12 mm in diameter, with active material mass loading of 0.6 mg). The assembly of the half-cell was carried out in an argon-filled glove box (H2O and O2 are less than 0.1 ppm). 1 M LiPF6 in a mixture of ethylene carbonate (EC) and diethyl carbonate (DEC) (1:1 vol %) was used as the electrolyte. Lithium foil was used as counter electrode, and Celgard 2400 microporous monolayer Polypropylene (PP) film with a thickness of 25 mm was used as the separator. Galvanostatic charge/discharge test was performed on a LAND CT2001A cycler. Cyclic voltammetry (CV) curves at various scan rates and electrochemical impedance spectra (EIS) in a frequency ranging from 100 kHz to 0.01 Hz were recorded on an electrochemical workstation (CHI 660e, Chenhua, Shanghai).
3. Results and Discussion
3.1 Materials synthesis and characterization
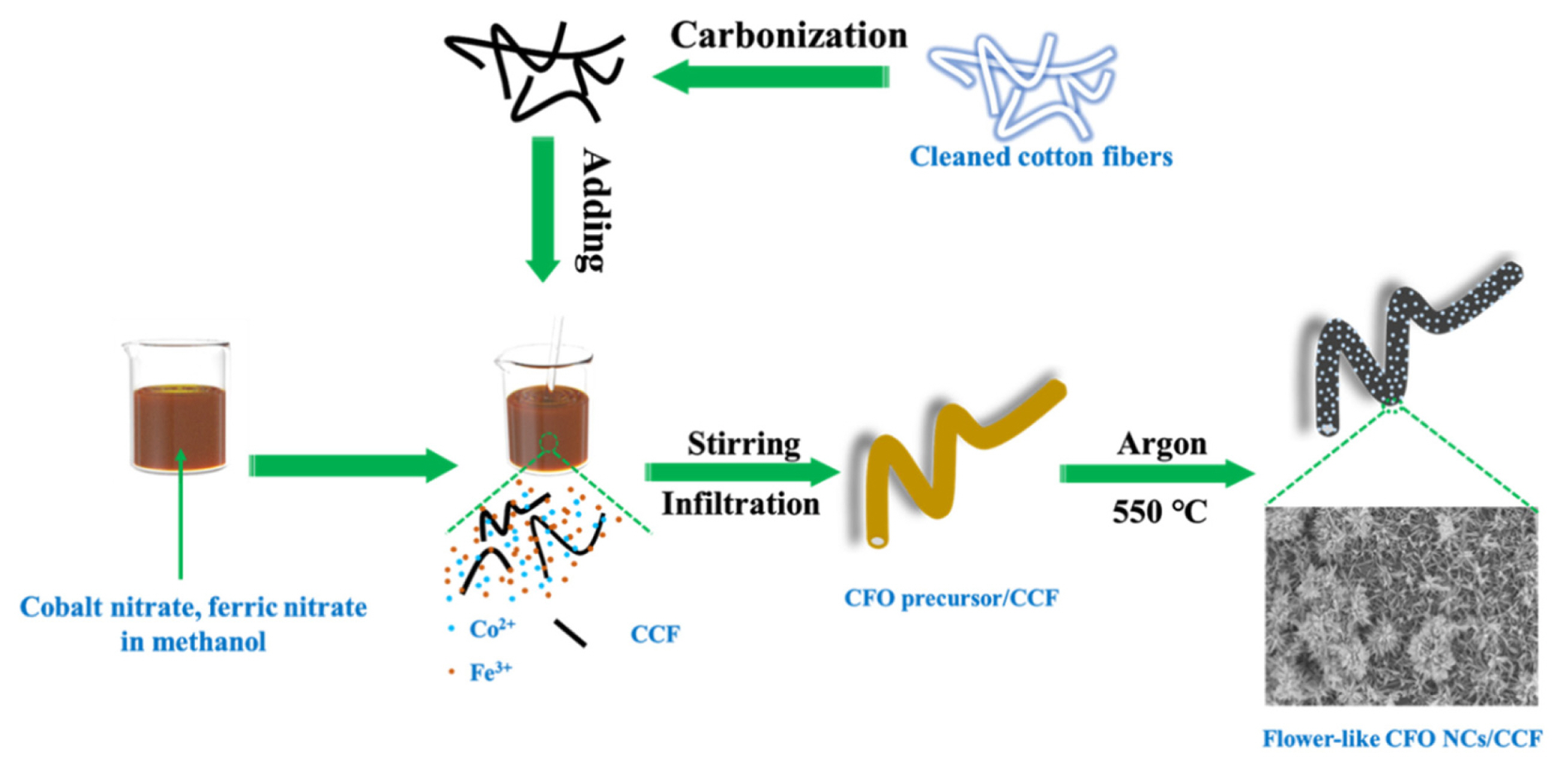
Fig. 1 is the schematic diagram of the facile synthesis process of CFO NCs/CCF. First, the absorbent cotton fibers are carbonized into CCF by heat-treatment in Argon at 800°C for two hours. The high temperature ensures complete carbonization as well as high integrity of the cotton fibers, which ensures a high electrode integrity and fast electron pathways [29]. To prepare CFO NCs/CCF, CCF is dispersed in a solution of cobalt nitrate and ferric nitrate in methanol by stirring in a water bath at a temperature of 30°C. The low surface tension of methanol, together with the capillary effect render enable CCF to be saturated with the CFO precursor solution. After agitation for some time, the residual liquid was completely evaporated in an oven to obtain a CFO precursor/CCF composite. At last, the composite was calcined in argon to obtain CFO NCs/CCF, during which, nitrates are decomposed and converted to CFO NCs.
Fig. S1 shows the XRD patterns of the CCF, p-CFO and CFO NCs/CCF. The broad peaks at 22° in the diffraction patterns of CCF and CFO NCs/CCF are the characteristic peak of disordered carbon materials [30]. The peaks at 30.00°, 35.34°, 42.80, 53.33°, 56.81°, 62.47°, and 73.96° n the diffraction patterns of the p-CFO and CFO NCs/CCF correspond to (220), (311), (400), (422), (511), (440), and (533) crystal planes of CoFe2O4 (PDF# 22-1086, Fd-3m), respectively [31]. The above results indicate the successful synthesis of CFO on CCF. The Raman spectrum of CFO NCs/CCF was recorded and the result is shown in Fig. S2, and the inset is a partial amplification with a wavenumber of 150–750 cm−1. The weak peaks in 150–750 cm−1 correspond to the Raman activity modes of CoFe2O4, where the peaks of 308 cm−1 and 472 cm−1, namely Eg and T2g, represents the motion of O ions, while the peak at 684 cm−1, namely A1g, represents the octahedral and tetrahedral sites ions [32]. The D, G and D+G bands are characteristic bands of carbon materials. The D band at 1357 cm−1 corresponds to the disordered and defective regions of the sp3-bonded carbon and G band at 1601 cm−1 G band corresponds to in-plane stretching of ordered sp2-bonded graphitic crystallites [23]. The Raman further confirms the successful synthesis of CFO NCs/CCF, in agreement with XRD patterns.
To investigate morphologies of synthesized samples, SEM observation was conducted and the images of CCF and CFO NCs/CCF are shown in Fig. 2. The SEM images of CCF show fibers with a diameter of approximately 10 μm and a length of tens of micros (see Fig. 2a). Some fibers have a hollow structure (see Fig. 2b). Flower-like CFO NCs are successfully loaded on CCF, shown in Fig. 2c–e. The flower like CFO NCs are assembled from many nanorods with a diameter of tens of nanometers and a length of 100–300 nanometers (see Fig. 2e). Element mappings of Co, Fe, and O as shown in Fig. 2f–i indicate that flower-like CFO NCs grow uniformly on the surface of CCF. In this unique hierarchical architecture, CCF could provide long-range electron transport pathway, and CFO NCs can provide abundant active sites for Li+ storage and large contact interface with electrolyte, as well as alleviate structural stress during Li+ intercalation.

SEM images of CCF (a, b), flower-like CFO NCs/CCF composite (c–e) and Fe, Co. and O elements mapping of flower-like CFO NCs/CCF composite (f–i).
TEM characterization was conducted to further observe the microstructure of the synthesized samples. The TEM images of CCF and CFO NCs/CCF are shown in Fig. 3, illustrating that the CoFe2O4 clusters were consisted of nanoparticles with diameter of ~20 nm and successfully loaded on the CCF surface. The HRTEM image of the CFO NCs is shown in Fig. 3f, and the inset is the corresponding selected area electron diffraction (SAED) pattern. In HRTEM image, the lattice fringes with the interplanar spacing of 2.63 Å and 3.13 Å belong to the (311) and (220) crystal planes of CFO, respectively. The SAED pattern has five diffraction rings, corresponding to the (220), (311), (400), (422) and (511) planes of the crystalline CFO phase. The N2 adsorption/desorption isotherm curves are shown in Fig. S3, in order to study the porosity of CFO NCs/CCF. The Brunauer–Emmett–Teller (BET) specific surface area of CFO NCs/CCF is calculated to be 181 m2/g. And pore size is concentrated at 11 nm. The large specific surface area and mesoporous nature of CFO NCs/CCF could facilitate the penetration of the electrolyte.
3.2 Electrochemical characterization
To evaluate the electrochemical performance of the CFO NCs/CCF, CV measurement was conducted and the results are shown in Fig. 4. According to previous reports [14, 33, 34], the lithiation/delithiation behavior of CoFe2O4 in LIBs can be described by the following equations:
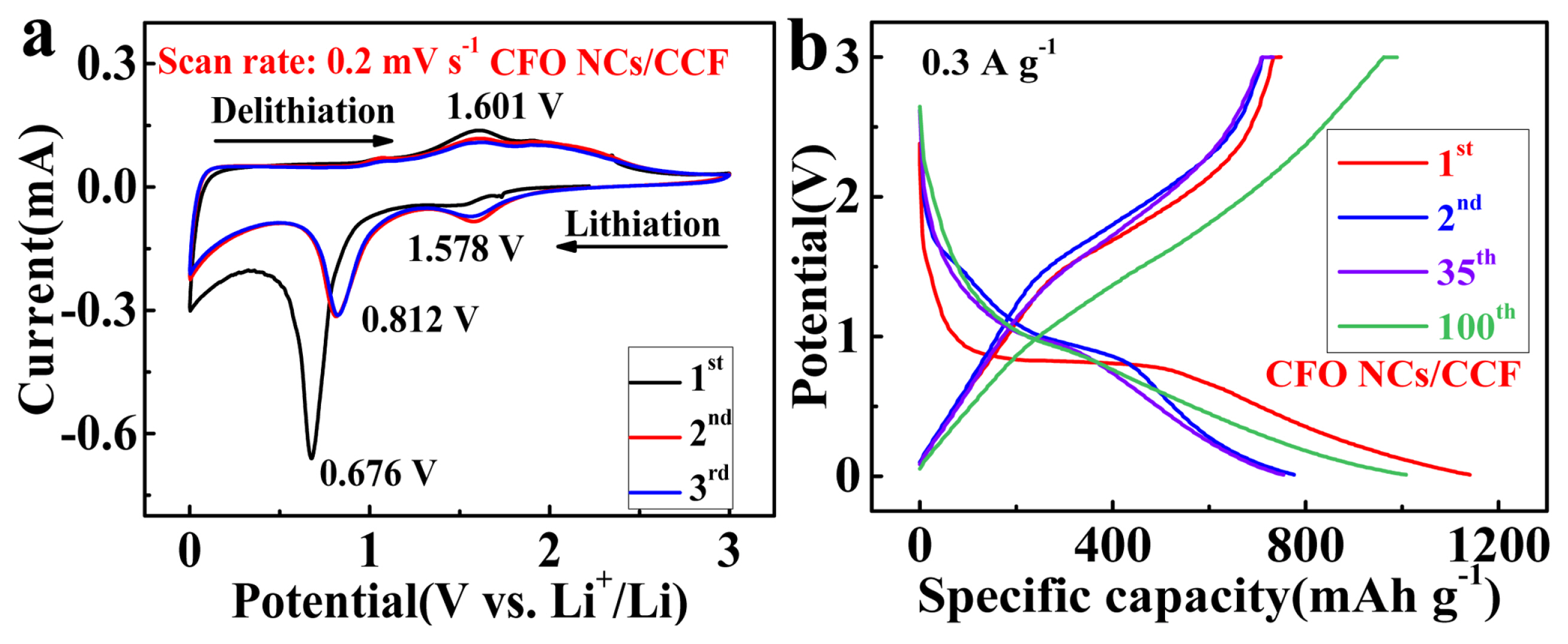
CV curves in the first three cycles at a sweep rate of 0.2 mV s−1 (a), galvanostatic charge and discharge curves in the 1st, 2nd, 35th and 100th cycle at o.3 A g−1 (b) of flower-like CFO NCs/CCF composite.
During the 1st cathodic process, a sharp peak appears at 0.676 V, corresponding to the conversion of CoFe2O4 to metallic Co0 and Fe0 (see Eq. 1) and formation of solid-electrolyte interphase (SEI) layer. In the subsequent 1st anodic process, a broad peak appears around 1.601 V, corresponding to the conversion of metallic Co0 and Fe0 to CoO and Fe2O3 (see Eqs. 2 and 3). During the cathodic process of the 2nd and 3rd cycles, two peaks with respect to the conversion of Fe2O3 and CoO to metallic Fe0 and Co0 occurred at 0.812 V and 1.578 V, respectively, while the positions of the peaks in anodic process is consistent with the 1st cycle (see Equations 2 and 3). Notably, CoFe2O4 is re-arranged after the 1st lithiation, CoO and Fe2O3 cannot be restored to the original spinel architecture, which is similar to other ferrites (e.g. zinc ferrite [35], nickel ferrite [16], manganese ferrite [36], etc.). Therefore, the lithiation/delithiation except for the first cycle was achieved by the mutual reversible conversion between Co0/Fe0/Li2O and CoO/Fe2O3 (see Eq. 4). The CV curves of the 2nd and 3rd cycles tend to overlapping, suggesting an excellent reversibility of the electrochemical reaction.
Fig. 4b shows discharge-charge curves of flower-like CFO NCs/CCF composite for the 1st, 2nd, 35th, and 100th cycle at 0.3 A g−1 with a potential window of 0.01 to 3 V. The apparent loss of capacity in the first cycle is attributed to the formation of the SEI film and irreversible electrochemical conversion reaction [17, 37, 38] Besides, the specific discharge/charge capacity of the 2nd, 35th and 100th cycle are 775.6/729.7, 754.1/732.4 and 1008.1/990.2 mAh g−1, respectively.
Fig. 5a shows the cycling performance of the three samples at 0.3 A g−1. The slight decrease in the reversible specific capacity of the CFO NCs/CCF is assigned to the lithiation-induced mechanical attenuation and unstable SEI film in the first few cycles [17]. After the 100th cycle, the reversible discharge/charge specific capacity of the CFO NCs/CCF is maintained at 1008.2/990.1 mAh g−1. In contrast, the CCF electrode exhibits stable but low capacity with cycling. The p-CFO electrode exhibits a rapid capacity decay with cycling. The increasing of capacity with cycling for nano-architectured transition metal oxide anodes has also been reported in previous studies [39–41]. Such an increase in capacity may be caused by following reasons: firstly, active material particles are gradually pulverized under electrochemical grinding effect, and forming a reversible growth/decomposition polymeric gel-like film (PGF) on newly formed surfaces to provide additional lithium storage capacity alleged pseudo-capacitive behavior [15,41,42]. Furthermore, electrolyte penetrates into the porous structure of the solid electrode with cycling and lead to electrochemically activate more active materials [8,43]. In addition, CCF acts as a conductive and buffer matrix to improve the cycling stability of the electrode.
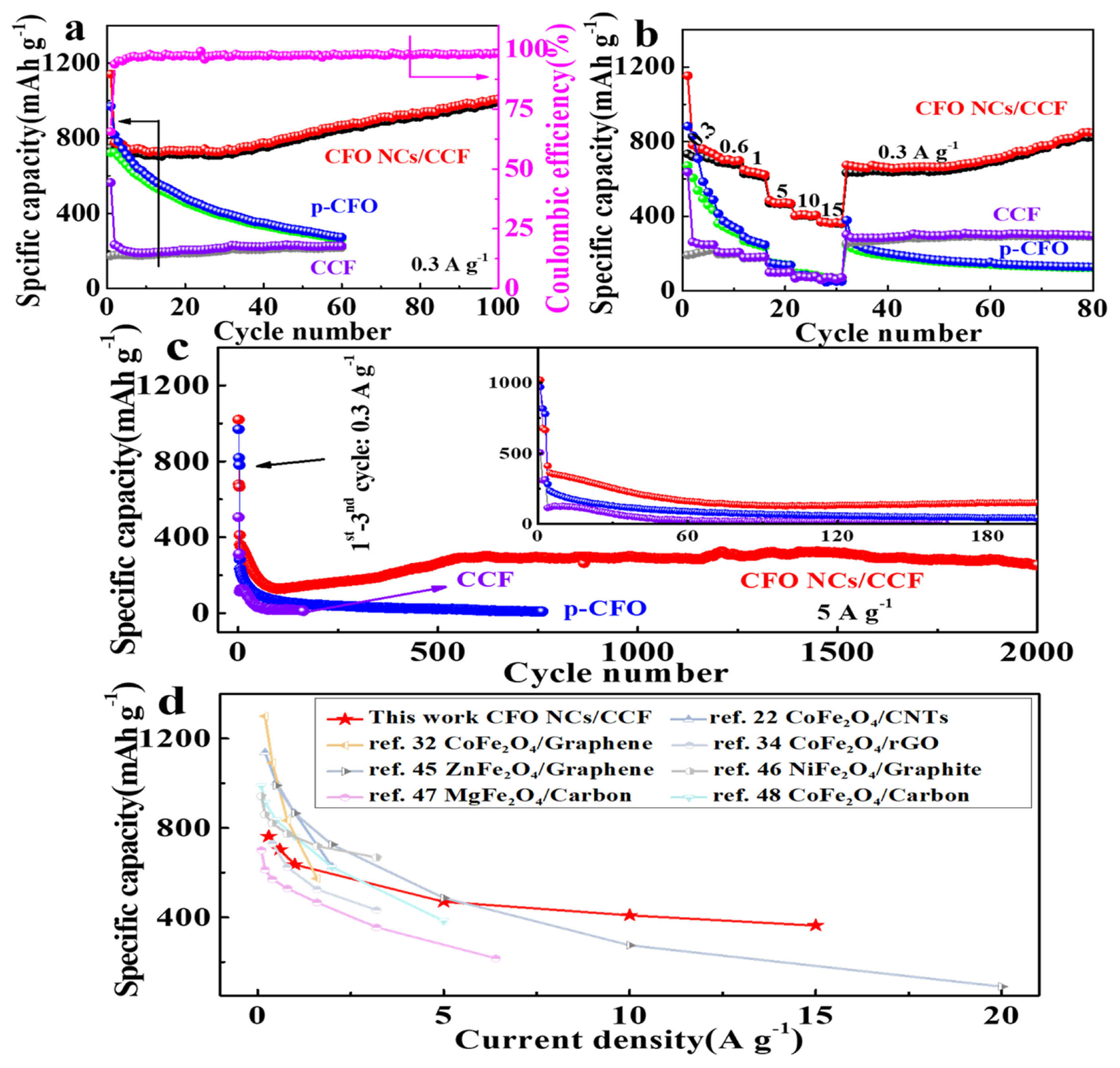
Cyclic performance at 0.3 A g−1 (a), rate performance (b), Cyclic performance at 5 A g−1 (c) of CCF, p-CFO and CFO NCs/CCF, and comparison of the rate performance of CFO NCs/CCF with previous cobalt or other ferrite-based anodes (d).
The rate performance of the three samples is shown in Fig. 5b. The CFO NCs/CCF has superior performance over CCF and p-CFO. Reversible discharge/charge capacities of 762.8/716.6 (3rd) and 703.7/682.6 (9th), 636/621.2 (14th), 470.5/464.9 (19th), 408.6/404.8 (24th), 362.7/361.0 (29th), 777.6/763.2 (70th) mAh g−1 correspond to the current densities of 0.3, 0.6, 1, 5, 10, 15, 0.3 A g−1 respectively. It is interesting to note that when the current density is reduced from 15 A g−1 to 0.3 Ag−1, the specific capacity does not completely return to the initial value, but the specific capacity increases with cycling. This may be due to the formation of PGF as described above and accompanied by the activation process, and this phenomenon is similar to previous report [41].
To further test stability performance of CCF, p-CFO and CFO NCs/CCF, long cycle performance at a current density of 5 A g−1 was carried out and the obtained data is shown in Fig. 5c (only for discharge specific capacity). In the first 80 cycles, the specific capacity of the CFO NCs/CCF fades severely. This might be caused by some CFO NCs that are detached from CCF or pulverized resulted from the high current density. The discharge specific capacity of CFO NCs/CCF subsequently shows an increase until it reaches 290 mAh g−1 after 550 cycle, and maintains at 255.8 mAh g−1 after 2000 cycles. For p-CFO and CCF, their specific discharge capacities have been attenuated to 9.7 (761st) and 11.2 (163rd) mAh g−1 respectively. These results suggest that, the long-term cycling performance of CFO NCs/CCF is far superior to p-CFO and CCF. Fig. 5d shows the compassion of the rate performance of CFO NCs/CCF and other similar materials reported previously [21,31,33,44–47], suggesting that the CFO NCs/CCF has an excellent capacity retention and a satisfactory high rate performance.
Some previous reports indicate that this unexceptional rate performance and cycle stability may be related to the capacitive behavior of nanomaterials with high surface area [22,48]. To further quantitatively examine the contribution of capacitive behavior to charge storage during the lithiation/de-lithiation process, CV of CFO NCs/CCF were performed at 0.2, 0.6, 1, 1.4, and 2 mV s−1, respectively (see Fig. 6a). In general, the relationship between the peak current (i) and the scan rate (v) can be described as the following equations [49]:
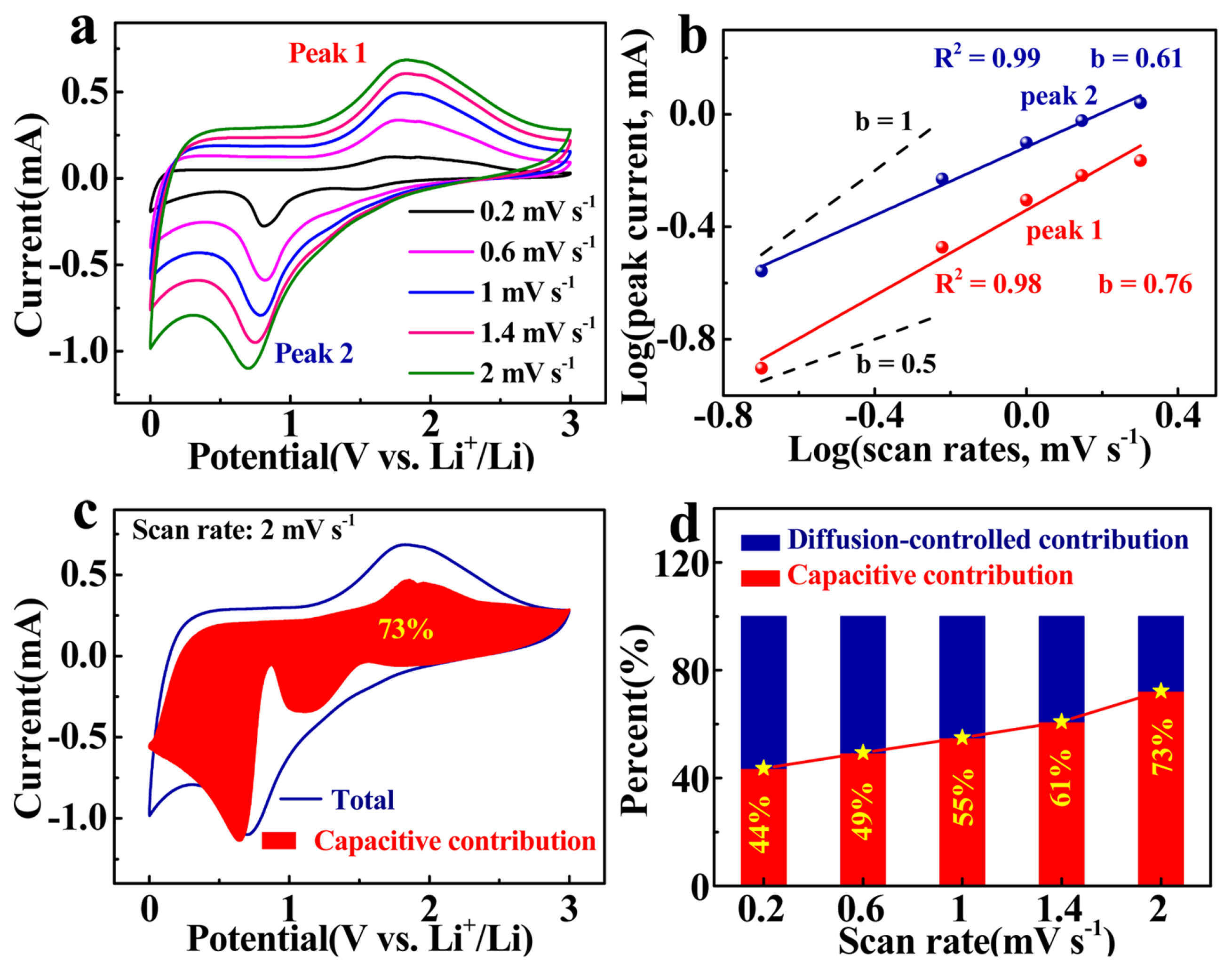
CV curves of CFO NCs/CCF at various scan rates (a), the relationship between peak current and sweep rate (b), capacitive current of CFO NCs/CCF at a scan rate of 2 mV s−1 (c), and the contribution percentages of the capacitive and diffusion-controlled processes to charge storage of CFO NCs/CCF at various scan rates.
Where, a and b are adjustable parameters, b ranges from 0.5 to 1. When b is close to 1, it indicates that the capacitive behavior dominates. Capacity comes primarily from the process of diffusion-controlled when the b is close to 0.5. As shown in Fig. 6b, the values of b at anodic and cathodic peaks are 0.76 and 0.61, respectively, demonstrating that the capacity of CFO NCs/CCF is contributed from both the capacitive behavior and diffusion processes [49]. The total current in the CV scan is the sum of the capacitive current (k1v) and the diffusion-controlled current (k1v0.5), described by the following equations [50]:
The calculated results are shown in Fig. 6d. As the scan rate increases, the percentage of capacity from capacitive behavior increases. When the scan rate is 2 mV s−1, the capacity contributed from capacitive behavior is 73%, which is shown as the red area in Fig. 6c (Others are in Fig. S4). The flower-like CFO NCs are composed of small nanoparticles of with high surface area, leading to a high capacitive induced capacity.
Electrochemical impedance spectroscopy (EIS) of CCF, p-CFO and CFO NCs/CCF was executed in the frequency range from 0.01 Hz to 100 kHz (see Fig. 7). In Fig. 7, a typical Nyquist plot consists of a semicircle in the high frequency region and a straight line in the low frequency region. The insert illustration is the equivalent circuit model. Where Rs is the intercept of the semicircle on the real axis representing the internal resistance of the battery, Rct is the semicircular diameter representing the charge transfer impedance with double layer capacitance CPEct, and Zw is the Warburg impedance of the ion diffusing into the interior of the bulk materials [12, 51]. Clearly, the CFO NCs/CCF has a lower charge transfer impedance. The corresponding exchange current densities (i0) of CFO NCs/CCF, CCF, and p-CFO are1.91×10−2, 9.7×10−3, 9.31×10−3 mA cm−2, calculated by the following equation:
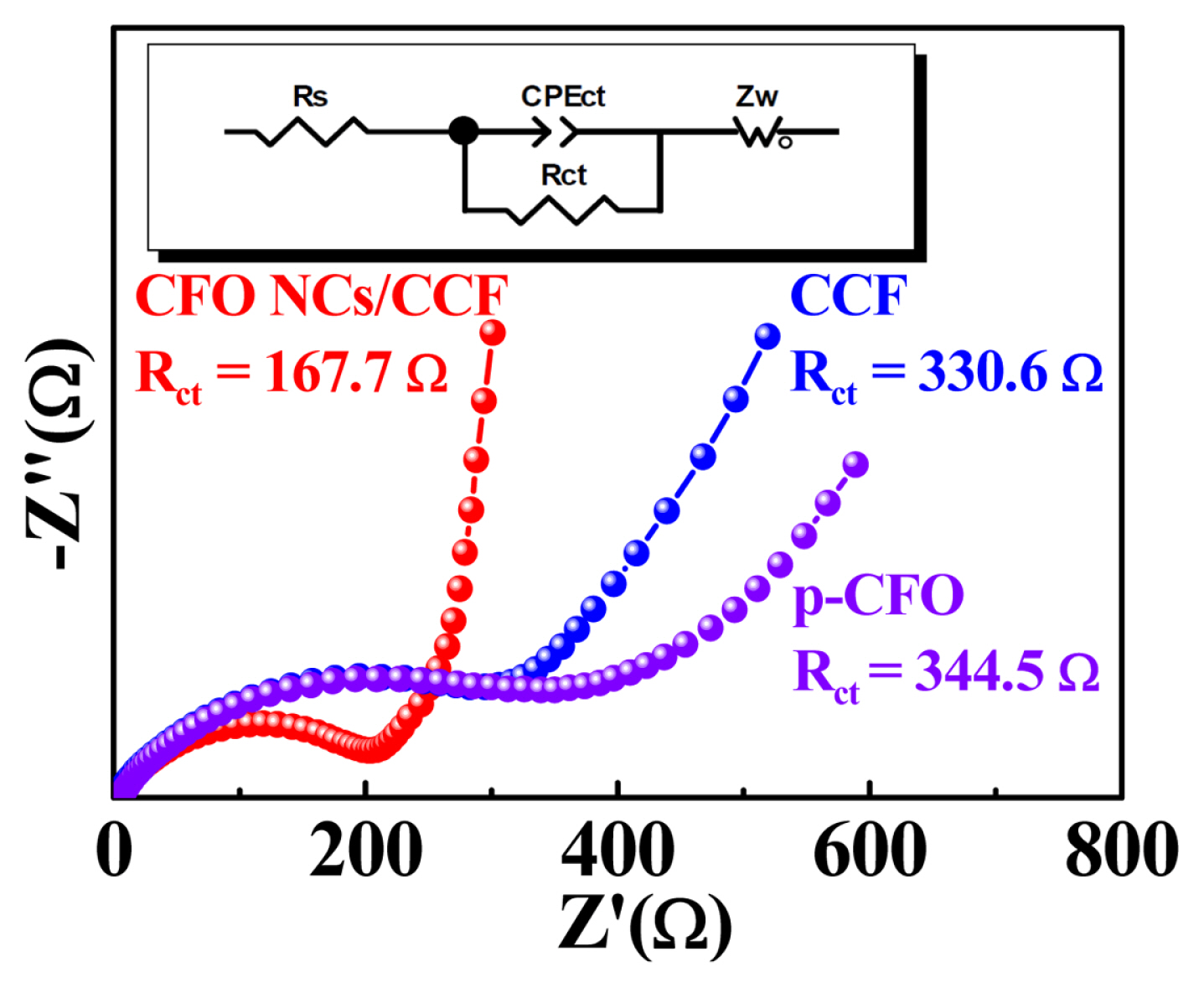
Nyquist plots of p-CFO, CCF, and flower-like CFO NCs/CCF composite before cycles (the inset is equivalent circuit model).
In the above Eq. 9, R is the gas constant, T is the absolute temperature (take 298 K), n is the number of electrons transferred in the redox reaction, F is the Faraday constant, and Rct is the charge transfer resistance. This result further confirms CFO NCs/CCF has improved electrode kinetics [8]. Compared to CCF and p-CFO, the higher slope of the CFO NCs/CCF in the low frequency region also means a faster Li+ diffusion rate [52]. In summary, explanations for the excellent electrochemical performance of 3D hierarchical flower-like CFO NCs/CCF composite might be as follows: (1) CCF acts as a long-range conductive matrix, providing an efficient electron transport channel and improving electrode reaction kinetics; (2) Flower-like CFO NCs composed of nanoparticles of about 20 nm is able to reduce the diffusion path of Li+ and alleviate volume stress during lithiation, and expand contact area with the electrolyte and (3) the high surface area of CFO NCs/CCF contribute the total capacity of the active materials by surface capacitive effect.
4. Conclusions
In this work, we have successfully grown flower-like CFO NCs onto the surface of carbon fibers by a facile and scalable method. When CFO NCs/CCF is used as the anode of LIBs, it exhibits excellent rate performance and long cycle performance. CFO NCs/CCF can deliver a capacity of 362.7 mAh g−1 at a current density of 15 A g−1 and maintain a capacity of 1008.2 mAh g−1 after 100 cycles at 0.3 Ag−1. Such satisfactory electrochemical performance is primarily due to the synergistic effect of CFO NCs and CCF in this 3D hierarchical architecture. CCF can provide an effective electron transfer channel as a long-range conductive matrix, while flower-like CFO NCs can increase the electrolyte contact area, reduce the volume stress and ion diffusion path. This work provides a facile, low cost, and scalable approach for preparing this special architecture of 3D ferrite nanoflowers to decorate biomass-derived carbon materials as anode in lithium storage.
Acknowledgement
This work was supported by the National Natural Science Foundation of China (Grant No. 52064035), and the Natural Science Foundation of Gansu Province (Grant No. 20JR10RA166).
Supporting Information
Supporting Information is available at https://doi.org/10.33961/jecst.2020.01648
Notes
Conflicts of interest
There are no conflicts to declare.