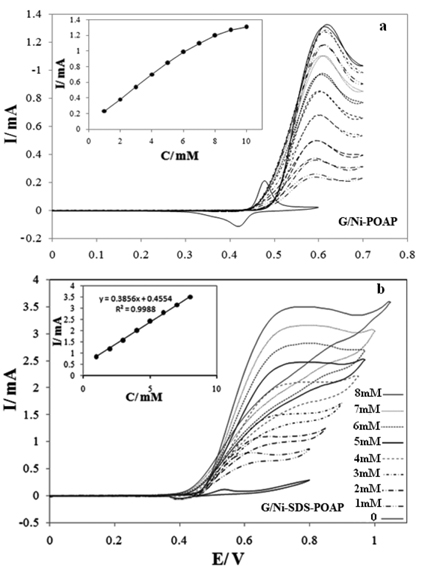
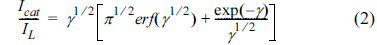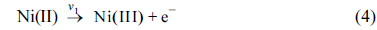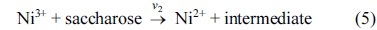[1] D.C. Johnson and W.R. LaCourse, Carbohydrate Analysis. Elsevier Science, Amsterdam, 1995.
[2] E. Katz, I. Willner and A.B. Kotlyar,
J. Electroanal. Chem.,
1999,
479, 64.

[3] S.B. Khoo, M.G.S. Yap, Y.L. Huang and S.X. Guo, Anal. Chim. Acta., 1997, 351, 1331.
[4] D.C. Johnson, S.G. Weber, A.M. Bond, R.M. Wightman, R.E. Shoup and I.S. Krull,
Anal. Chim. Acta,
1986,
180, 187.

[5] H.W. Lei, B.L. Wu, C.S. Cha and H. Kita,
J. Electroanal. Chem.,
1995,
382, 103.

[6] V.S. Bagotzky and Yu.B. Vassilyev,
Electrochim. Acta,
1964,
9, 1329.

[7] J.O’M. Bockris, B.J. Piersma and E. Gileadi,
Electrochim. Acta,
1964,
9, 1329.

[8] T. Ikeda and K. Kano,
Biophys. Acta,
2003,
1647, 121.

[9] R.A. Bullen, T.C. Arnot, J.B. Lakeman and F.C. Walsh,
Biosens. Bioelectron.,
2006,
21, 2015.

[10] C. Zhao, C. Shao, M. Li and K. Jiao, Talanta, 2007, 71, 1773.
[11] M. Morita, O. Niwa, S. Tou and N. Watanabe,
J. Chromatogr. A,
1999,
837, 17.

[12] M. Fleischmann, K. Korinek and D. Pletcher,
J. Electroanal. Chem.,
1971,
31, 39.

[13] A. Hilmi and J.H.T. Luong,
Anal. Chem.,
2000,
72, 4677.

[14] E. Skou,
Electrochim. Acta.,
1977,
22, 313.

[15] L.A. Colon, R. Dadoo and R.N. Zare,
Anal. Chem.,
1993,
65, 476.

[16] M. Fleischmann, K. Korinek and D. Pletcher, J. Chem. Soc. Perkin Trans., 1972, 2, 1396.
[17] P.F.F. Luo and T. Kuwana,
Anal. Chem.,
1994,
66, 2775.

[18] V. Singh, S. Mohan, G. Singh, P.C. Pandey and R. Prakash,
Sens. Actuators B,
2008,
132, 99.

[19] S. Mokrane, L. Makhloufi, H. Hammache and B. Saidani,
J. Solid State Electrochem.,
2001,
5, 339.

[20] D.N. Upadhyay and D.M. Kolb,
J. Electroanal. Chem.,
1993,
358, 317.

[21] J. Chen, C.O. Too, G.G. Wallace, G.F. Swiegers, B.W. Skelton and A.H. White, Electrochim. Acta, 2002, 47, 27.
[22] P.C. Pandey, S. Upadhyay, G. Singh, Rajiv Prakash, R.C. Srivastava and P.K. Seth,
Electroanalysis,
2000,
12, 517.

[23] R.M. Banik, Mayank, R. Prakash and S.N. Upadhyay,
Sens. Actuators B,
2008,
131, 295.

[24] M.G. Mahjani, A. Ehsani and M. Jafarian,
Synth. Met.,
2010,
160, 1252.

[25] A. Ehsani, M. G. Mahjani, M. Jafarian and A. Naeemy,
Electrochimica. Acta.,
2012,
71, 128.

[26] A.R. Feizbakhsh, A. Naeemy, A. Ehsani, A. Aghasi and I. Danaee,
J. Chin. Chem. Soc.,
2012,
59, 1086.

[27] A. Ehsani, M.G. Mahjani and M. Jafarian,
Synth. Met.,
2011,
161, 1760.

[28] A. Ehsani, M. G. Mahjani, M. Jafarian and A. Naeemy,
Progress in organic coating,
2010,
69, 510.

[29] S. Radhakrishnan, A. Adhikari and DK. Awasti,
Chem. Phys. Lett.,
2001,
341, 518.

[30] S. Berchmans, H. Gomathi and GP. Rao,
J. Electroanal. Chem.,
1995,
394, 267.

[31] R. Ojani, J.-B. Raoof and S. Fathi,
Electrochim. Acta,
2009,
54, 2190.

[32] F.D. Eramo, J.M. Marioli, A.A. Arevalo and L.E. Sereno,
Electroanalysis,
1999,
11, 481.

[33] A.J. Bard and L.R Faulkner, Electrochemical Methods. Wiley, New York, 2001.
[34] A. Ehsani, MG. Mahjani and M. Jafarian,
Synth. Met.,
2012,
162, 199.

[35] A. Ehsani,
Prog. Org. Coat,
2015,
78, 133–139.

[36] A. Ehsani, MG. Mahjani, M. Bordbar and S. Adeli,
J. Electroanal. Chem.,
2013,
710, 29.

[37] A. Ehsani, M.G. Mahjani and M. Jafarian, J. Chem., 2011, 35, 1.
[38] A. Ehsani, M.G. Mahjani, S. Adeli and S. Moradkhani,
Prog. Org. Coat.,
2014,
77, 1674.

[39] A. Ehsani, MG. Mahjani, R. Moshrefi and H. Mostaanzadeh, J. Shabani Shayeh, RSC. Adv., 2014, 4, 2231.
[40] A. Ehsani, M. Nasrollahzadeh, MG. Mahjani, R. Moshrefi and H. Mostaanzadeh,
Ind. Eng. Chem.,
2014,
20, 4363.

[41] A. Ehsani, M.G. Mahjani, M. Nasseri and M. Jafarian,
Methods Mater.,
2014,
61, 146.

[42] A. Ehsani, B. Jaleh and M. Nasrollahzadeh,
J. Power Sources.,
2014,
257, 300.

[43] A. Ehsani, S. Adeli, F. Babaei, H. Mostaanzadeh and M. Nasrollahzadeh,
J. Electroanal. Chem.,
2014,
713, 91.

[44] S. Naghdi, B. Jaleh and A. Ehsani,
Bulletin of the Chemical Society of Japan,
2015,
88, 722.

[45] A. Ehsani, R. Moshrefi and M. Ahmadi,
Electrochemical Science and Technology,
2015,
6(
1), 7.

[46] A. Ehsani, F. Babaei and H. Mostaanzadeh, J. of the Brazilian Chem. Soc., 2015, 26, 331.
[47] A. Ehsani, M G. Mahjani, F. Babaei and H. Mostaanzadeh,
RSC Advances,
2015,
5, 30394.

[48] A. Ehsani, A. Vaziri-Rad, F. Babaei and H. Mohammad Shiri,
Electrochim. Acta,
2015,
159, 140.