|
 |
- Search
| J. Electrochem. Sci. Technol > Volume 12(1); 2021 > Article |
|
Abstract
This study reports for the first time the application of electrocoagulation (EC) from laboratory to pilot scales for the treatment of printing wastewater, a hazardous waste whose treatment and disposal are strictly regulated. The wastewater was taken from three real printing companies with strongly varying characteristics. The treatment process was performed in the laboratory for operational optimization and then applied in the pilot scale. The weight loss of the electrode and the generation of sludge at both scales were compared. The results show that the raw wastewater should be diluted before EC treatment if its COD is higher than about 10,000 mg/L. Pilot scale removal efficiencies of COD and color were slightly lower compared to those obtained from the laboratory scale. At pilot scale, the effluent CODs removal efficiency was 81.9 ŌĆō 88.9% (final concentration of 448 ŌĆō 992 mg/L) and color removal efficiency was 95.8 ŌĆō 98.6% (final level of 89 ŌĆō 202 Pt-Co) which proved the feasibility of EC treatment as an effective pre-treatment method for printing wastewater as well as other high colored and hard-biodegradable wastewaters.
Printing ink is a complex, multi-component compound, composed principally of dyes, pigments, resins, binders, solvents, and optical additives. Hence, wastewater generated from the printing process is highly colored, and contaminated with organic composes because of the afore-referred compounds. Most of the components in wastewater are synthetic organic polymers with strong stability, which bring on the BOD5/COD of the wastewater generally lower than 0.4. Moreover, printing ink wastewater contains plenty of substances inhibiting biological activity and heavy metal ions, making of difficulty to be microbial decomposed and transformed [1]. Due to the harmful effect on human health and the environment, as well as the aesthetical aspect, the printing ink wastewater cannot be directly discharged into the environment without any treatment. Practically, various methods have been used to treat this wastewater, including coagulation, adsorption, oxidation, biological treatment, electrolysis [1], electrocatalytic process [2], and electrochemical treatment [3]. Among these methods, electrocoagulation (EC) has been proposed as an effective method for treating various types of wastewater from textile, paper mill, bakerŌĆÖs yeast, restaurant, and chemical mechanical polishing [4,5], paint manufacture wastewater [6], and lithographic wastewater [7]. However, there is very rare report on the application of this technique for printing waste-water [8ŌĆō10]. Scaling up of EC was found for domestic greywater reuse [11] and almond industry wastewater treatment [12] but has not been conducted for printing wastewater.
To the best of our knowledge, it is the first time EC process was applied for printing wastewater treatment, from laboratory to pilot scale. Printing wastewater was collected from three printing companies, including fabric screen printing, packing printing with various branches, and paper packing printing. Firstly, experiments were performed on a laboratory scale to find the optimum condition from wastewater dilution level, input voltage, pH, electrode distance, mixing speed, and electrolyte concentration. The treatment was then scaled up to pilot scale with both batch and continuous modes, and operated for a long duration.
Wastewater samples were collected from a fabric screen printing wastewater source in Ho Chi Minh City (Vietnam) during November 2017 to March 2018 (denoted as S1); from a packing printing wastewater source in Long An Province (Vietnam) during April to June 2018 (denoted as S2, including three subsamples, denoted as S2-1, S2-2, and S2-3). Before that, wastewater samples from another paper packing factory in An Giang Province (Vietnam) were taken from March to June 2017 (denoted as S3).
Electrodes were made by aluminum (96.61%), with the components analyzed by X-ray fluorescence (XRF) elemental analysis as given in Table 1.
NaCl, acid, and base were of analytical grade (Xilong, China), purchased from Viet Hoang Long Company (Ho Chi Minh City, Vietnam). All the analyses of COD, BOD5, total nitrogen, true color, and TSS were done in Consultancy Center of O.S.H & Environmental Technology (Ho Chi Minh City, Vietnam). pH was measured using PHS-550 (Taiwan). Color was measured according to 2120 C spectrophotometric method [13] using a Photolab 6100 VIS spectrophotometer (Germany) operated at 455 nm. TSS was analyzed followed by 2540 D [13]. COD, BOD5, and total nitrogen were analyzed followed by 5220 C (Closed Reflux, Titrimetric Method) [13], TCVN 6001-1:2008, and TCVN 6638:2000, respectively. Weight of sludge and electrode was measured using a Sartorius TE412 portable scale balance.
For EC treatment at the laboratory scale, wastewater was treated using a 1-L batch cell made by polyethylene. Two similar aluminum plate electrodes with an effective area of 91 cm2 were placed vertically in the cell and connected to a DC power supply (TES 6102, Taiwan). Samples with a volume of 15 mL were taken from electrocoagulation cell at certain operating times for true color and COD analyses. The wastewater was first checked for dilution requirement using tap water, based on the observation of changes in the color of treated wastewater after running the treatment for 30 min. Dilution factor (f) was the ratio between (volume of wastewater + volume of tap water) and volume of wastewater, varied from 1 to 9. If the color of the effluent was obviously faded, the effluent was then taken to measure COD and color. To find the optimum working condition, effects of voltage (5 ŌĆō 40 V) with the operation time from 0ŌĆō120 min, pH (5 ŌĆō 8), electrolyte (NaCl) concentration (0.05 ŌĆō 0.40 g/L), and inter-electrode distance (1 ŌĆō 5 cm) on COD and color removals were evaluated. The pH of the wastewater was adjusted by using 1N NaOH and 1N H2SO4 solutions. When the optimum operating condition was obtained, the removal efficiencies of COD, BOD5, true color, and TSS were measured at this condition.
A pilot was designed including an EC rectangular reactor (600 ├Ś 240 ├Ś 300 mm), connected to a sedimentation tank (200 ├Ś 200 ├Ś 400 mm), both made by acrylic resin. The filled volume of wastewater in the reactor was fixed at 24 L. The number of electrodes was changed from 2 to 8 with an effective area of 720 cm2 for each aluminum electrode. The pilot was run either in batch or continuous modes. The operating condition for each type of wastewater was the corresponding optimum condition for these wastewaters from laboratory results (30 V, 30 min, pH 6ŌĆō7, electrode distance of 2 cm, without mixing and adding of electrolyte). During the treatment, floating sludge was manually removed from the top of the tank. After treatment by EC process, the wastewater was settled for 30 min, and samples were taken to analyze COD, color, BOD, TSS, and total nitrogen.
The characteristics of three types of wastewater are given in Table 2 and the images of wastewater can be seen in the Supplementary data (Fig. A). In general, printing wastewater is generated in a low volume but very strong in the concentration of contaminants and varies strongly depending on the sources. The reason would be the different types and concentrations of inks used in each process as well as the original printing wastewater with/without a combination of domestic wastewater or/and cleaning wastewater. In addition, the range of variation in characteristics of S2 (n = 3) was stronger than those of S1 and S3, which attributes to the change in printed products from the company that generated S2 wastewater. During this study period, their products were changed three times, resulted in three subsamples, denoted as S2-1, S2-2, and S2-3.
At the beginning, the electro-coagulation treatment was designed to treat directly raw printing wastewater. This succeeded for wastewater S1 and S3 but did not work for wastewater S2. For raw S2 samples, applying EC treatment resulted in unstable electric currents with no visible change in color after 30 min of EC treatment. Also, very less sludge was generated and floated. Hence, different dilution factors were tested for these samples. Table 3 shows the changes in COD and color of the S2-1, S2-2, and S2-3 wastewater before and after treatment at different dilution factors.
For S2-1 and S2-3, applying EC treatment for samples diluted at the factors (f) of 2 and 3 still resulted in no visible change of color after 30 min of treatment. This is because of too high COD and color in raw wastewater (Table 2) as well as the diluted wastewater. When f was equal to or greater than 4, change in color of the treated wastewater was observed.
The results from Table 3 also suggest that EC treatment seemed to work obviously for the wastewater with COD about 10000 mg/L. No more dilution was required for the wastewater with COD lower than this recommended value. It is noted that in the case of S2-2, we selected the dilution factor f = 3 instead of f = 2 since we desired COD in EC treated wastewater lower than 1000 mg/L to facilitate the further treatment step of the effluents. Our approach in selecting dilution differed from previous studies using EC treatment without dilution for paint manufacturing wastewater (COD = 19700 mg/L [6]) and lithographic wastewater (TOC = 18400 mg/L [7]). The treated wastewater from these studies still contained very high in COD (about 1182 mg/L) and in TOC (3825 mg/L), which could be still hard for selecting the next step of treatment, especially for biological treatment. For the real industrial scale, we suggested designing the system with the recycled line of treated wastewater for the dilution instead of using tap water as in this study.
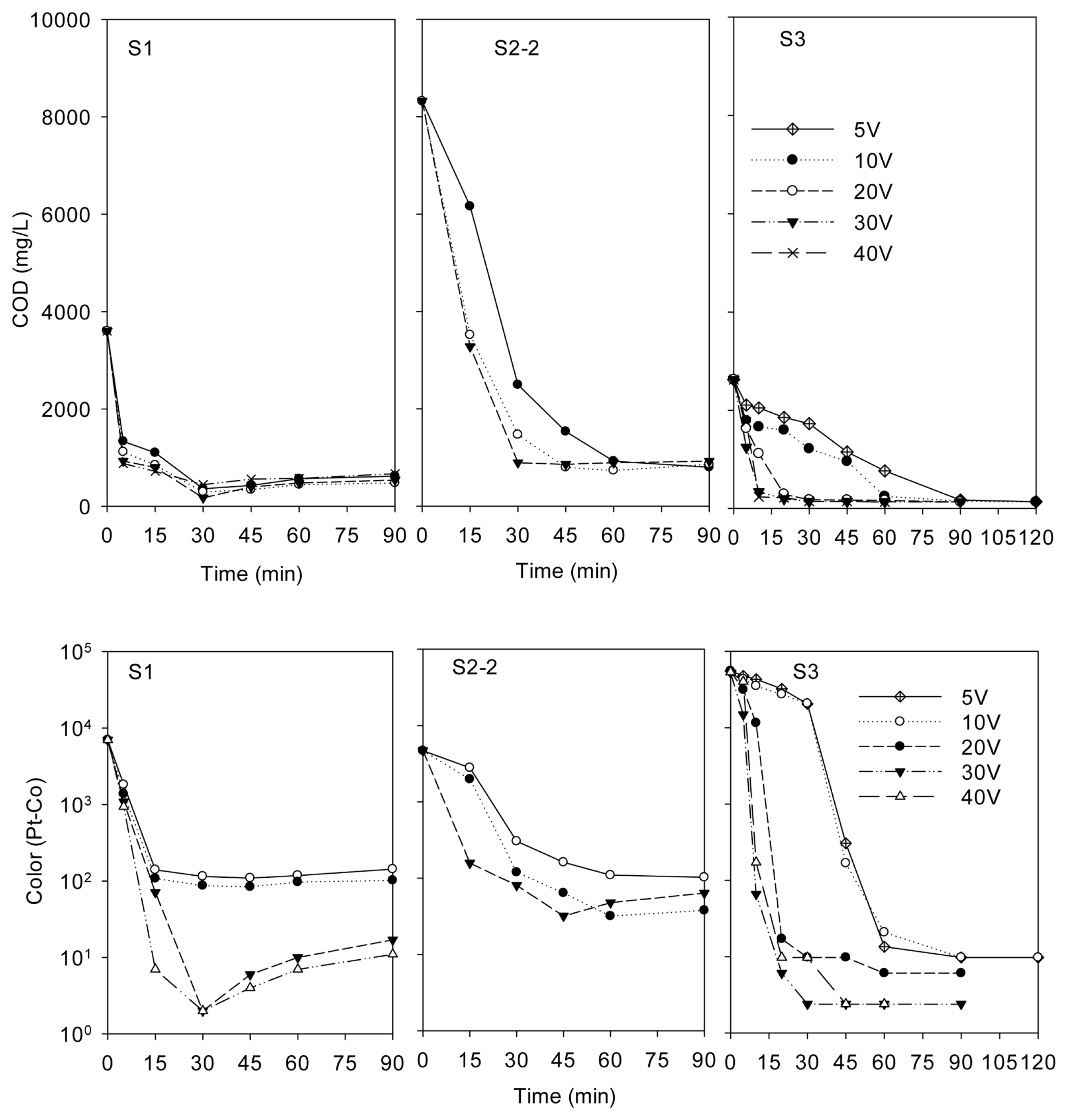
The effect of applied voltage on COD and color in the effluents is plotted in Fig. 1 and the corresponding current densities are given in Table 4. Images of feed wastewater and effluents can be seen in the Supplementary data (Fig. B). It is noted that we enlarged the applied voltage to 40 V for the treatment of S1 and S3, but to 30 V for S2-2. As can be seen from Fig. C in the Supplementary data, the wastewater temperature increased by the increase of both voltage and treatment time. The temperature increased slowly for S1 with the maximum value of 41┬░C at 40 V and 90 min, but significantly for S2-2, reached 95┬░C at 30 V and 90 min. Hence, for safety as well as saving energy we did not operate the treatment of S2-2 at the voltage of 40 V or higher.
The increase in applied voltage (from 5 to 30 V), corresponding to the current density increase (Table 4), resulted in better elimination of both COD and color in the wastewater. It is explained that current density increased led to the increase of coagulants concentration, and therefore enhancing the coagulation process. Current density increased also led to a higher number of bubbles generated, which promotes the flotation process. However, COD and color removals at 30 V differed insignificantly compared to that at 40 V.
It is further noted that for each voltage, increasing treatment time at the beginning of the period led to a significant increasing in COD and color removal. However, operating the experiment for longer time mostly unchanged or even slightly increased COD and color in the effluents. Hence, for three types of printing wastewater, the optimum voltage and time for COD removal were 30 V and 30 min, which were mostly concomitant with the suitable voltage and time for color removal.
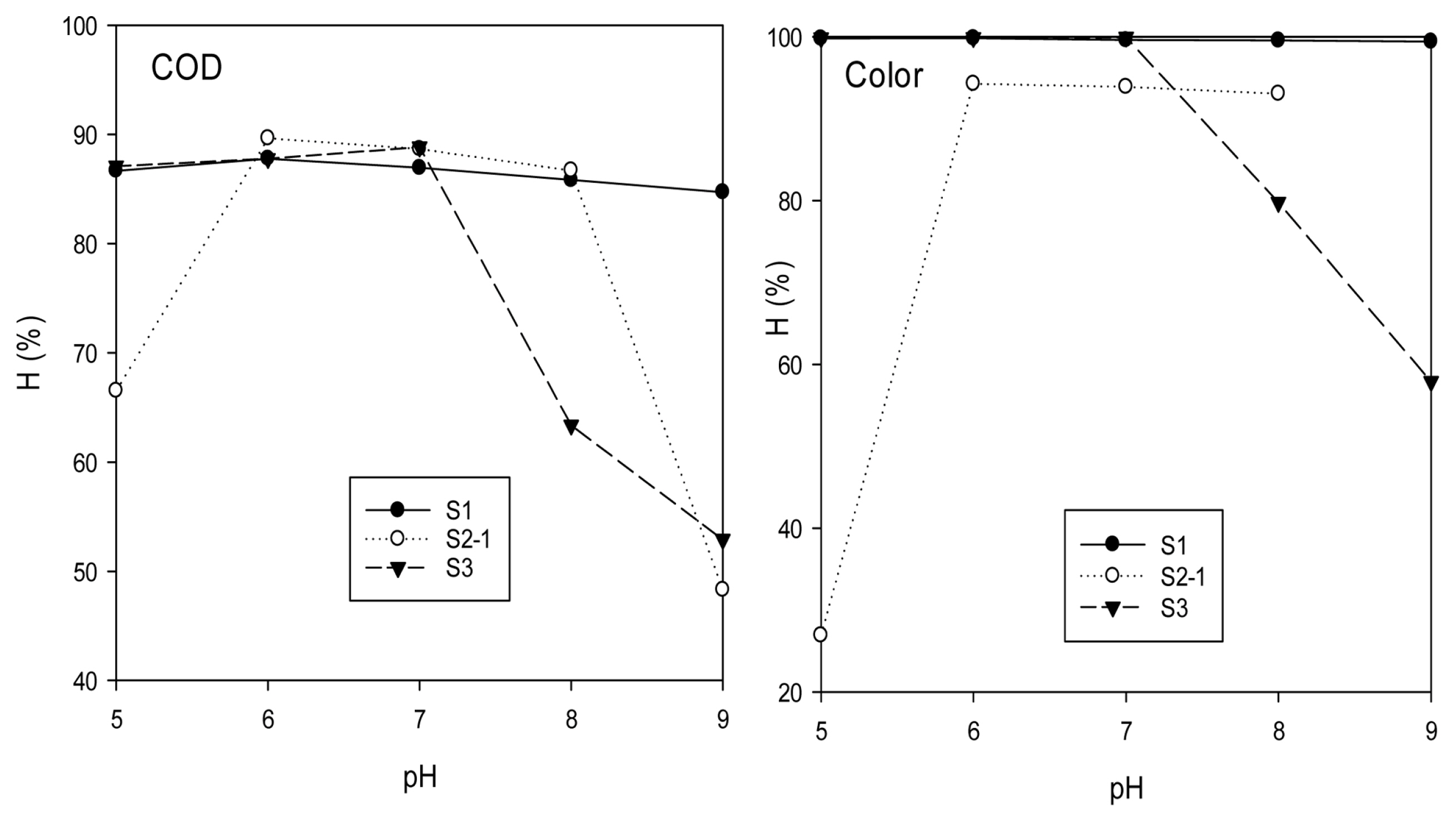
Variation in pH affects solution conductivity, electrodesŌĆÖ dissolution, forms of aluminum species, and zeta potential of colloidal particles [5]. In this study, the effect of pH from 5 ŌĆō 9 was examined and the results are shown in Fig. 2. For S1, changing pH from 5 ŌĆō 9 varied slightly the removal efficiency of COD from 84.7 to 87.8%, and color from 99.4 to 99.9%, suggesting the optimum pH of 6. For S2-1 and S3, increasing pH affected strongly on COD and color removal efficiency, which firstly increased and then reduced. Maximum COD and color removal efficiency was 89.7 and 96.3%, respectively, for S2-1 at pH 6, and 89.3 and 99.9%, respectively, for S3 at pH 7. For all three types of wastewater, COD removal efficiencies in this study at optimum pH were clearly higher compared to those provided by electrocoagulation treatment in Papadopoulos et al. [10] (72.0ŌĆō85.8%). Also, the results suggested that pH ~ 6 ŌĆō 7 was the optimum range for the treatment of COD and color in printing wastewater, possibly because of the dependence of the forms of aluminum species on pH. It is well known that Al3+ ions generated from anode during electrocoagulation combine with water and hydroxyl ions to form monomeric, polymeric, amorphous and less soluble species of Al [14]. Since the solubility of aluminum hydroxide Al(OH)3 precipitate is minimum at pH of 6.0 [15], large amounts of these precipitates were formed leading to the rapid adsorption of soluble organic compounds and trapping of colloidal particles [16]. However, due to aluminum amphoteric character, Al(OH)3 solubility increases as the wastewater becomes either more acidic or alkaline leading to the increase of Al soluble forms (e.g. Al3+, Al(OH)2+ or Al(OH)4ŌłÆ), resulted in the decreases of COD and color removal efficiency. Our approach in the adjustment of pH to optimum value differed from Adamovic et al. [8] where the EC treatment was directly performed at initial pH (11 ŌĆō 12) for printing wastewater without the evaluation of pH effect. Optimum pH found in this study is similar to the values finding from Bensadok et al. [14] for COD and turbidity removal in cutting oil emulsions and close to the optimum pH (5.7) found by Valero et al. [12] for TOC removal in almond industry wastewater. However, these pH values differed from Su├Īrez-Escobar et al. [7] who found optimum pH 8.23 for lithographic wastewater, and from Akyol [6] with optimum pH from 4 ŌĆō 8 for paint manufacturing wastewater. Accordingly, no pH adjustment was required for S1 and S3 whose initial pH values were in the range of 6 ŌĆō 7, but slight acidification was needed for S2 with initial pH about 8 ŌĆō 9.
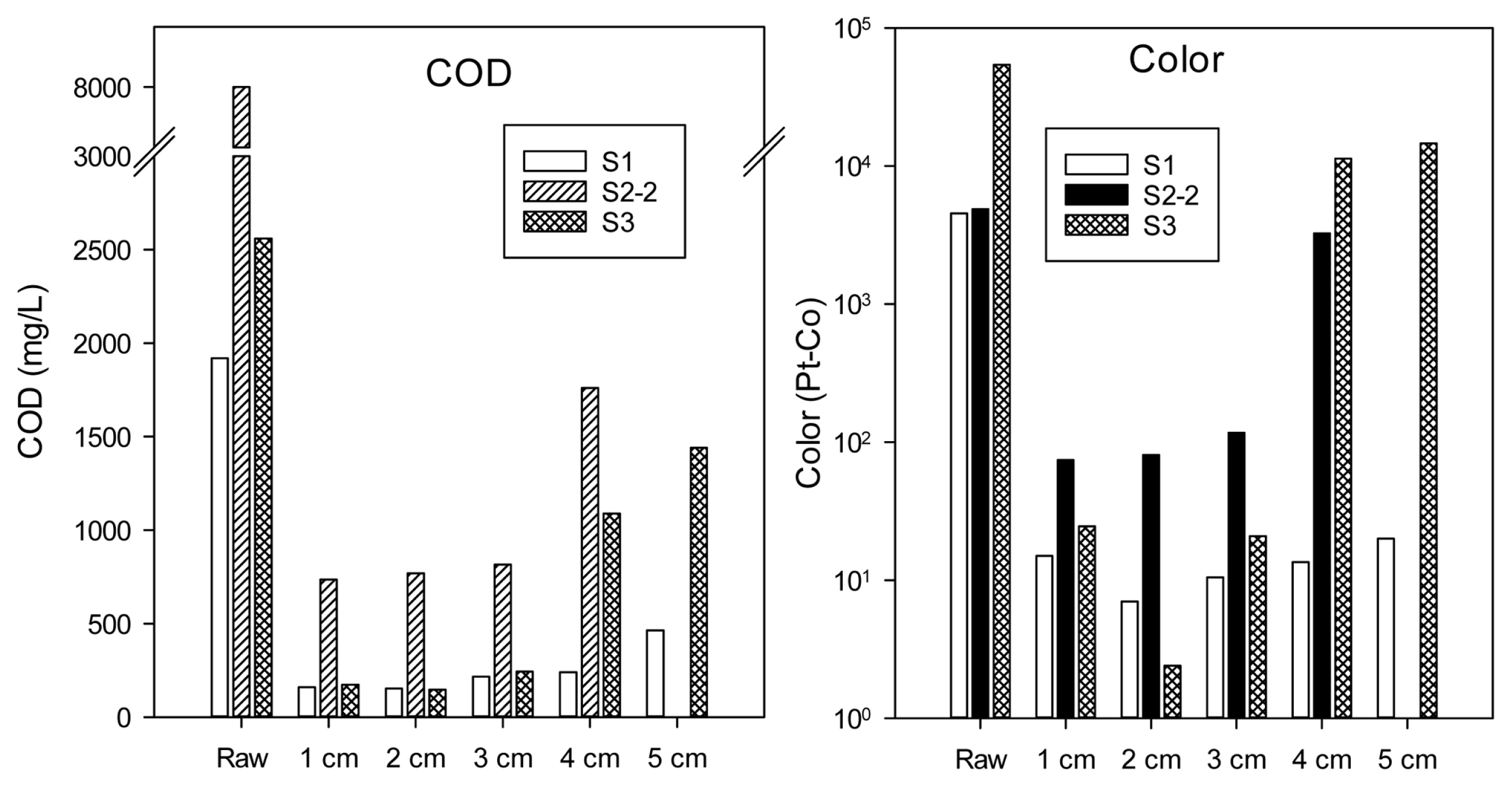
The distance between the two electrodes affects the electrocoagulation process as the electrostatic field depends on the distance of the electrodes [17]. Optimum electrode distance in the EC treatment varied from different studies, e.g. 0.1 cm [18], 0.5 ŌĆō 1.0 cm [8], or 2.8 cm [19]. In this study, the effect of electrode distance (1 ŌĆō 5 cm) on COD and color removal was tested. As can be seen from Fig. 3, electrode distance of 1 and 2 cm provided similar COD in effluents and the best color removal was found for the distance of 2 cm. Further broadening electrodes gap (>2 cm) resulted in lower removal efficiencies of both COD and color which would be due to: (i) the decrease of current density at a fixed voltage, e.g. current densities were 263.74, 219.78, 142.86, and 87.91 corresponding to the distance of 1, 2, 3, and 4 cm for S2-2. Similarly, current densities were 43.96, 32.97, 21.98, 16.48, and 10.99, corresponding to the distance of 1, 2, 3, 4, and 5 cm for S1. Current density reduction with the increase of electrode distance was also observed by Phan et al. [20], and further mentioned by Thella et al. [21] as a consequence of resistance increase; (ii) the decrease in the electrostatic attraction that caused a slower movement of the generated ions [17] leading to the lower removal efficiency of pollutants since Fekete et al. [22] found that the dissolved Al from anode being active as a coagulant for seconds only after its formation; and (iii) the diminution of mass transfer in the reactor [5].
On the other hand, a too narrow distance between two electrodes is also not recommended because metal hydroxides generated get degraded by collision with each other [23]. Thella et al. [21] also preferred not to apply too short distances to avoid short-circuiting and Sahu et al. [5] mentioned the frequent floc deposits induced by the low level of turbulence between the electrodes. Hence, we selected a distance of 2 cm instead of 1 cm for the next experiments to facilitate the floating of sludge, its harvesting and the convenience in cleaning electrodes.
Adding salt into the EC system can increase the conductivity of wastewater, reduce adverse effect of other anions (HCO3ŌłÆ, SO42ŌłÆ), decrease in power consumption, and provide additional disinfection [24]. In this study, electrolyte concentration in S1 and S3 was varied by adding different NaCl dosages, i.e. 0, 0.05, 0.1, 0.2, 0.3, and 0.4 g/L. For S1, the effect of adding salt had an unclear impact on COD removal efficiency, ranged from 84 ŌĆō 88%. In a previous study, Valero et al. [12] also found the removal efficiency of TOC was unchanged (i.e. 80%) between adding or non-adding of NaCl. We further observed a slight decrease in COD removal efficiencies for the treatment of S3 with the increase of salt concentration added, within 91 ŌĆō 95%. Hence, the addition of salt may not be necessary for printing wastewater, possibly due to the sufficient ion existing in the raw wastewater.
Many studies apply mixing at different speeds, e.g. 200 rpm [25], 250 rpm [14], 450 rpm [8], and 900 rpm [7] to make the reaction solution homogeneous. In this study, mixing at different speeds (i.e., 0, 100, 200, 300, and 400 rpm) was applied for EC treatment of S3. Interestingly, mixing led to a slight reduction in COD removal compared to no mixing, and provided no effect on color removal. The result from this study is in agreement with Phan et al. [20] who achieved the highest COD and color removal efficiency in coffee wastewater without the addition of agitation. However, it differed with the results from several studies where the optimum speed was 300 rpm for cadmium removal [26], 150 rpm for arsenic removal [27], COD removal in poultry slaughterhouse wastewater [28], and boron removal [29]. In our study, the increase of COD in the effluent when the EC treatment applied stirring would be due to the breaking of flocs, and some floating sludge returning back to the solution under stirring. It is further noted that bubbles generated at two electrodes already assisted the mixing in the solution. Since the effect of stirring speeds has not been tested for wastewater S1 and S2, further investigation is needed for this factor with a broader type of printing wastewater. For the next experiments, we decided not to apply stirring in the electrocoagulation reactor.
The results from laboratory experiments provided us the feasibility of EC for the actual treatment of printing wastewater. Optimum conditions in terms of wastewater dilution, applied voltage, pH, electrode distance, mixing, and electrolyte concentration were finalized for each type of wastewater and were applied for pilot scale to treat S1 and S2.
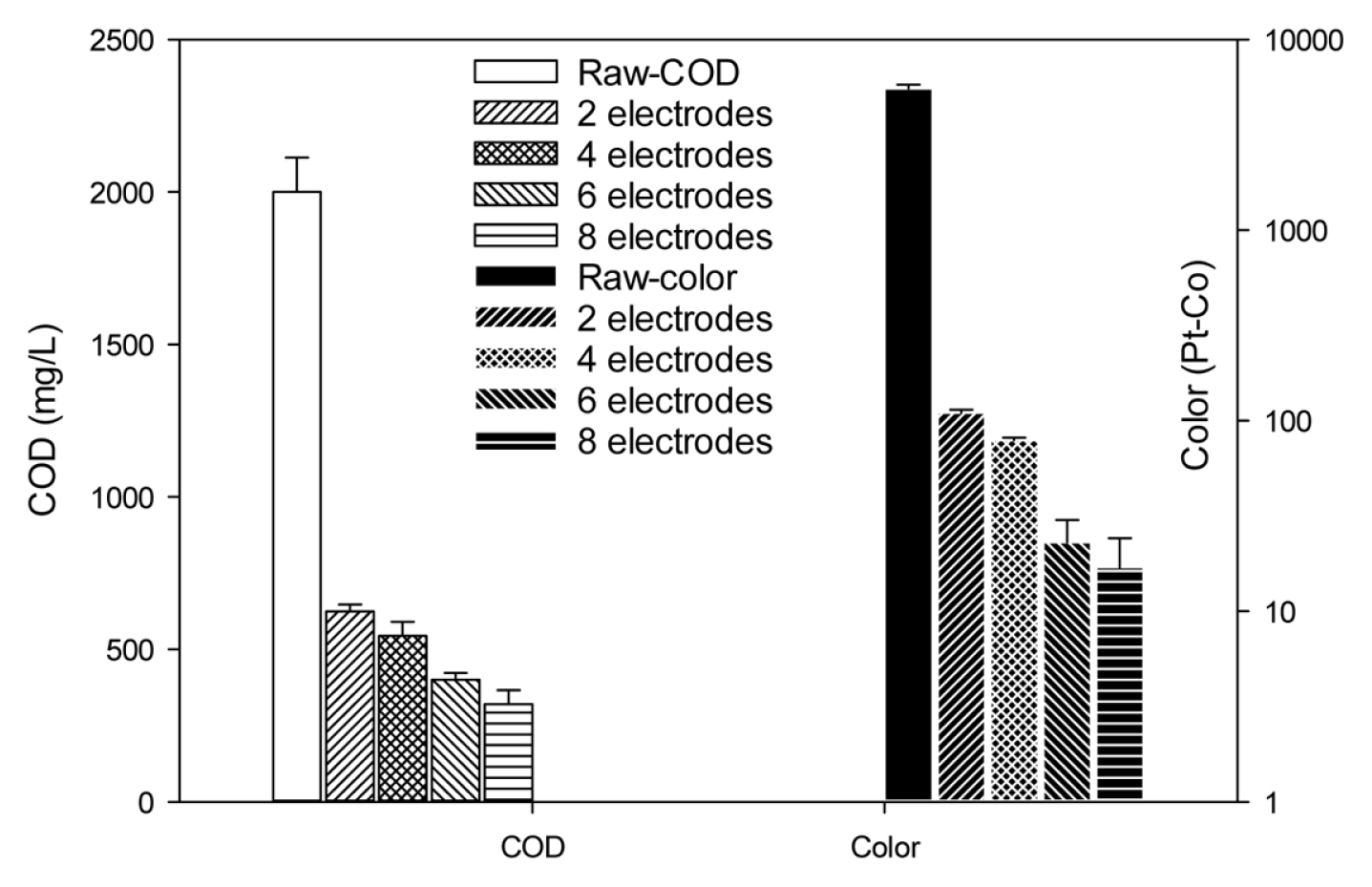
According to the result from laboratory scale, ratio between electrodes area/wastewater volume (S/V) was 2├Ś91 (cm2)/1 (L) = 18.2 (m2/m3). For the pilot scale, we tended to keep the value close to this ratio, by applying 6 electrodes with monopolar arrangement (Fig. D in the Supplementary data), resulted in the ratio of [(6├Ś720) (cm)2/240 (L) = 18.0 (m2/m3). This ratio was a few higher than the value selected by Khaled et al. [26] (i.e. 13.6 m2/m3). In addition, the effect of the number of electrodes from 2 to 8 on COD and color removal was investigated for S1. The current intensity was also kept at optimum condition found from laboratory scale, i.e. 33 A/m2 for individual unit.
As can be seen from Fig. 4, the increase in the number of electrode improved the removal of both COD and color because of the enlarging active surface [26]. This trend was similar as the findings from Akbal and Camc─▒ [30] for electrode number increasing from 2 to 6 in the removal of Cu, Cr, and Ni, and from Ahmadian et al. [31] with electrode number from 4 to 8 to treat slaughterhouse wastewater. In term of S/V ratio, our results reinforced that increase in S/V ratio by increase of electrode number led to the increase in removal efficiency, which is consistent with the trend found by Khaled et al. [26] where much better removal efficiency of cadmium was achieved with larger S/V ratio. It is noted that this study changed the total electrical current applied for the system to keep the current density of each unit at 33 A/m2 as the optimum value found from the laboratory experiment. Hence, when the number of electrode increased, the total current required increased, resulted in the increase in power consumption. Since the improvement of removal efficiency was more obvious when the number of electrodes increased from 4 to 6 (i.e. 7.2% for COD and 1.0% for color) compared from 6 to 8 (i.e. 4.0% for COD and 0.1% for color), 6 electrodes were selected as appropriate number.
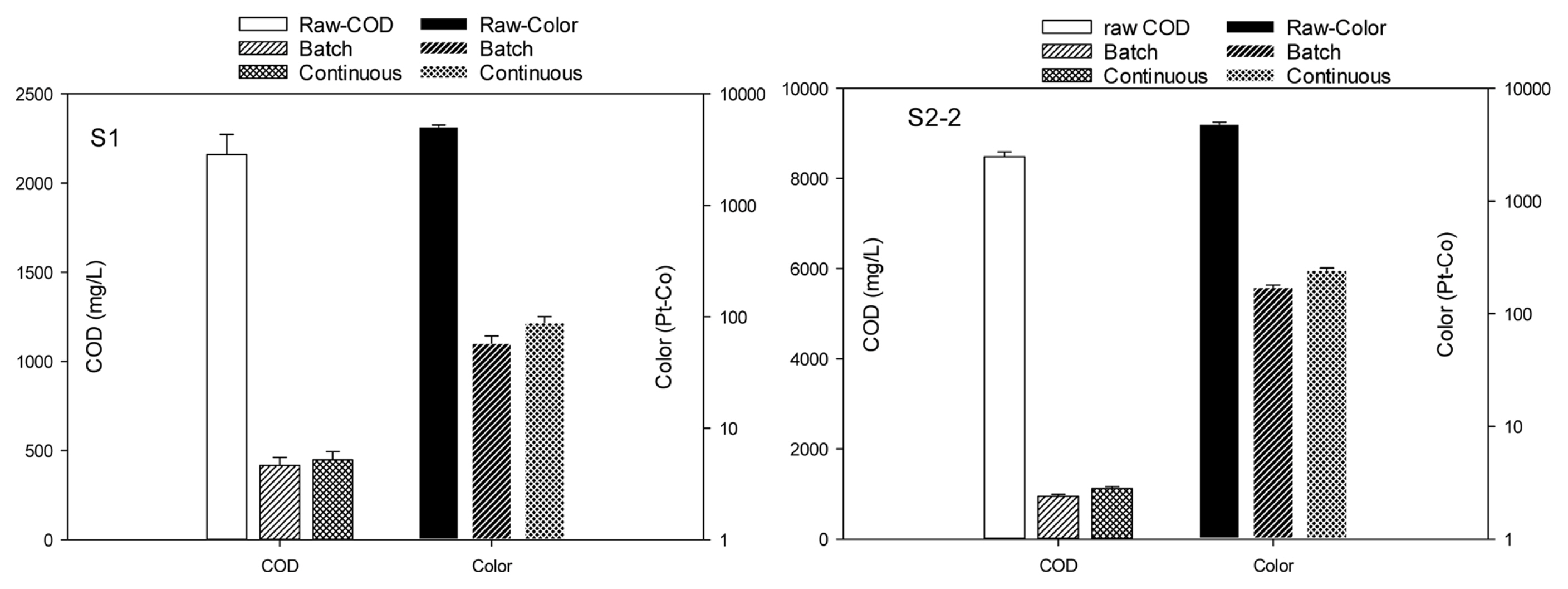
It is obvious that performing the treatment in the laboratory scale where most of the conditions are well controlled will result in better removal efficiency as compared to the treatment on the pilot scale. As demonstrated in Fig. 5, removal efficiencies at the pilot scale were 81.9 % COD and 98.6% color for S1 and 88.9% COD and 95.8% color for S2-2. These efficiencies were lower than those achieved from laboratory scale but the difference in efficiency was small, i.e. 2.6% for COD and 0.9% for color of S1, and 1.4% for COD and 1.5% for color of S2-2. The images of feed wastewater and effluents after lab and pilot-scale treatment can be found in Fig. E in the Supplementary data.
Selection of batch or continuous operation modes depends on the discharging regime of the source as well as the volume of the printing wastewater. While the batch mode is suitable for the wastewater discharged intermittently in low volume, the continuous mode is preferred for the case of wastewater discharged continuously with a larger volume. In this study, we examined the performance of both operation modes using S1 and S2-2 samples, and the results are given in Fig. 6. Obviously, compared to continue mode operation, batch mode provided a slightly better COD and color removal efficiency. The difference in efficiency between batch and continue modes were 1.5% COD and 0.62% color for S1, and 2.07% COD and 1.52% color for S2.
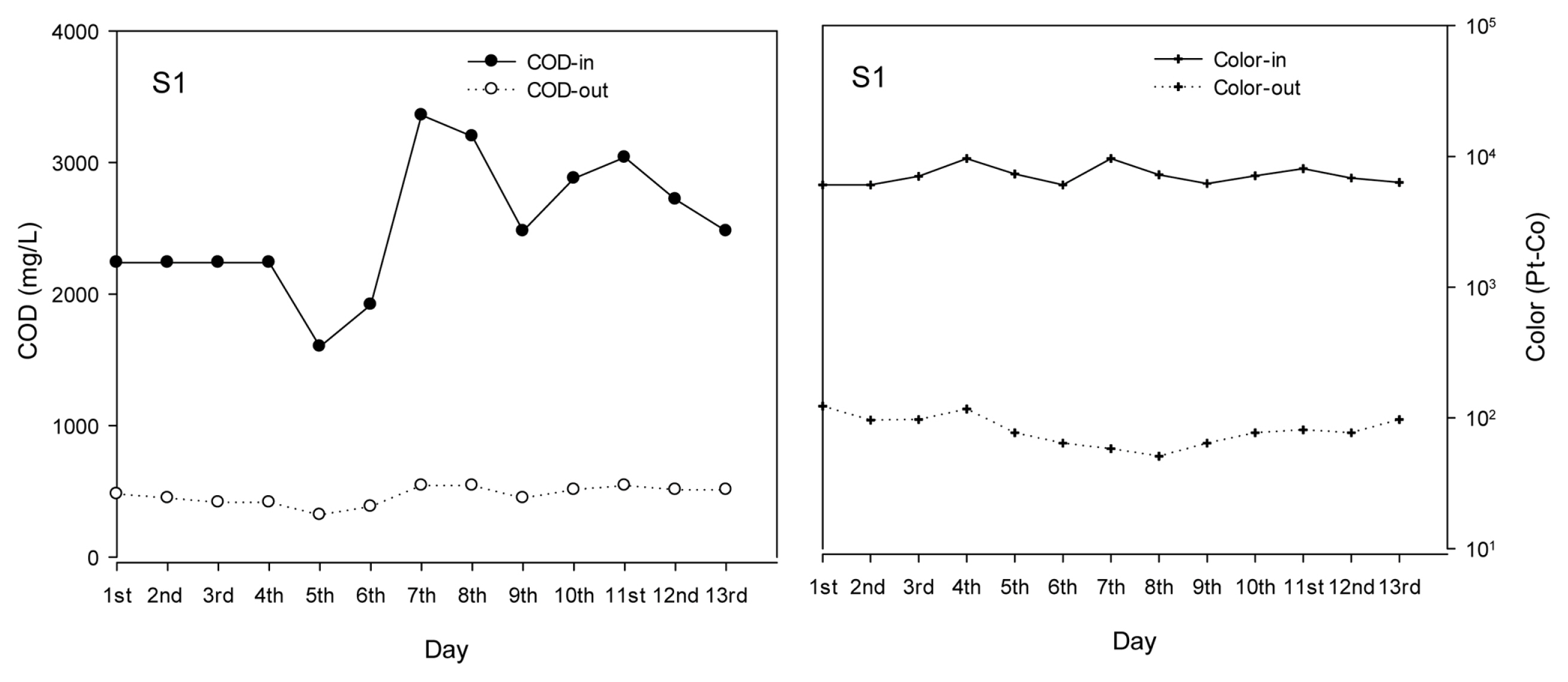
To test the stability of the wastewater treatment system in the pilot scale, experiments were conducted using continuous mode with 8 h/day for 13 days using in-situ S1 wastewater. As can be seen from Fig. 7, though COD in the raw wastewater varied strongly during 13 days, from 1600 to 3360 mg/L, COD in the effluent was quite stable in the range of 302 ŌĆō 544 mg/L. Similarly, the color in raw wastewater was from 6060 to 9619 Pt-Co, resulted in color of 51 ŌĆō 123 Pt-Co after EC treatment. This relatively stable outlet concentration of COD and color despite the high variation in the inlet suggests EC as a very potential technology for the treatment of printing wastewater in practical application.
Table 5 shows the characteristics of treated wastewater (S1, S2-3) at the pilot scale and the standard for industrial wastewater discharging. Obviously, the values of all parameters were significantly reduced. Though pH, TSS, total nitrogen met the standard, COD and color (for S2-3) were still higher than the allowable values. The remaining COD, color, and BOD in treated wastewater possibly existed in the dissolved forms which could not be effectively removed by electro-coagulation. Similarly, previous studies also using electrocoagulation for paint manufacturing wastewater [6] and lithographic wastewater [7], printing ink wastewater [10], resulted in the treated wastewater still high in COD (1182 mg/L), TOC (3825 mg/L), and COD (Ōēź2000 mg/L), respectively. Hence, the electrocoagulation treatment is suitable as an effective pre-treatment for printing wastewater, and an advanced treatment process is needed to remove the remaining contaminants.
The weight loss of anode and cathode electrodes was measured for both laboratory and pilot scale from the treatment for S1 wastewater after running the operation for 30 min. As can be seen from Table 6, in pilot scale the weight loss of each either anode or cathode electrodes differed slightly from the others. Total weight loss from both anode and cathode electrodes was 0.098 kg/m3 which was a few lower than the weight loss given by Valero et al. [12] for almond industry wastewater in pilot scale. For S2-3, weight loss of all electrodes in the pilot scale was slightly higher compared to that in the lab scale. It is further noted that the treatment of S2-3 consumed a higher amount of aluminum than that of S1 due to the higher electrical current applied for S2-3 (Table 4).
Sludge generated was collected for five minutes and dried at 105┬░C before weight measurement. The dried sludge generated at the pilot scale for S1 was 6.55 kg/m3 while this value was higher (i.e. 8.23 kg/m3) at the laboratory scale. These both values are in the range found by Akyol [6] in paint wastewater treatment at the laboratory scale (approximately 5 ŌĆō 16 kg/m3). For S2-2 treatment, the amount of dried sludge collected for the pilot scale was 157.95 kg/m3 which is significantly higher than those from S1 treatment because of significantly higher COD in S2-2 feed water (Table 2 and Table 3).
For the first time, electrocoagulation (EC) was applied to treat real printing wastewater both in laboratory and pilot scales, proving the feasibility of this technique as an effective pre-treatment method for these strong COD and high color wastewaters to decrease the COD to less than 1000 mg/L and facilitate the further treatment step of the effluents. The selection of this technology for printing wastewater was based on the fact that this wastewater is generally generated in small volumes but with high COD concentration and strong color. EC treatment was firstly run in a laboratory scale to find the optimum operating condition for each type of wastewater. While the optimum condition of applied voltage (30 V), pH (6ŌĆō7), electrode distance (2 cm) was similar for three types of printing wastewater, their optimum current density was significantly differed, from 33 to 198 A/m2. The results of this study also recommended conducting electrocoagulation treatment without mixing and adding electrolyte. EC treatment at the pilot scale was then applied at both batch and continuous operations, and for a long duration. Removal efficiencies for the pilot scale were 81.9 ŌĆō 88.9% for COD and 95.8 ŌĆō 98.6% for color which were slightly lower compared to laboratory scale (87.8 ŌĆō 89.7 % for COD and 96.3 ŌĆō 99.9% for color). The weight loss of the electrode and the sludge generated were also considered for both lab and pilot scales. Running the system for 13 days showed stable concentrations of COD and color in the effluent though these parameters in feed wastewater highly varied.
Supplementary Information
Supporting Information is available at https://doi.org/10.33961/jecst.2019.00444
Acknowledgement
This work has been sponsored and funded by Ho Chi Minh City University of Food Industry under Contract No.146/H─É-DCT. We thank the students Huynh Minh Duc and Nguyen Thi Hong Phan for their kindly help in sampling and analysis.
Fig.┬Ā5
Comparison between laboratory scale and pilot scale on COD and color removal from S1 and S2-2 (n = 3).

Table┬Ā1
Components of aluminum electrodes.
Table┬Ā2
Characteristics of raw wastewater from different sources.
Table┬Ā3
Effect of dilution factor (f) on COD and color in the effluent of S2-1, S2-2, S2-3.
Table┬Ā4
Applied voltages and current densities in EC treatment of printing wastewater.
| Voltage (V) | i (A/m2) | ||
|---|---|---|---|
|
|
|||
| S1 | S2-2 | S3 | |
| 5 | - | - | 24 |
| 10 | 16 | 99 | 36 |
| 20 | 22 | 165 | 83 |
| 30 | 33 | 198 | 131 |
| 40 | 44 | - | 143 |
Table┬Ā5
Summary of effluentsŌĆÖ characteristics after EC treatment by pilot scale.
| Parameter | S1 | S2-3 | QCVN 40:2011/BTNMT, column B |
|---|---|---|---|
| pH | 6.5 ŌĆō 7.5 | 7.5 ŌĆō 8.5 | 5.5 ŌĆō 9 |
| COD (mg/L) | 448 | 992 | 150 |
| Color (mg/L) | 89 | 202 | 150 |
| TSS (mg/L) | 40 | 36 | 100 |
| Total N (mg/L) | 7 | 40,6 | 40 |
Table┬Ā6
Weight loss of electrodes (S1) (n=2).
| Electrode | Weight loss (kg/m3) | ||||
|---|---|---|---|---|---|
|
|
|||||
| S1 | S2-3 | ||||
|
|
|||||
| Lab-scale* | Pilot-scale** | Lab-scale* | Pilot-scale** | ||
| Anode | 1st | - | 0.027 | - | |
| 2nd | 0.030 | - | |||
| 3rd | 0.027 | - | |||
| Total (Anode) | - | 0.084 | 0.52 | 0.43 | |
|
|
|||||
| Cathode | 1st | - | 0.005 | - | |
| 2nd | 0.004 | - | |||
| 3rd | 0.005 | - | |||
| Total (cathode) | - | 0.014 | 0.03 | 0.18 | |
| All electrodes | - | 0.098 | 0.55 | 0.61 | |
References
[1] L. Ding, Y. Chen and J. Fan, Journal of Environmental Chemistry and Ecotoxicology, 2011, 3(10), 272ŌĆō276.
[2] C-H. Tung, S-Y. Shen, J-H. Chang, Y-M. Hsu and Y-C. Lai, Sep Purif Technol, 2013, 117, 131ŌĆō136.

[3] Z. Liu, Y. Chen, S. Hu, J. Qiu, Y. Meng and L. Li, Chemical Engineering Transactions, 2016, 55, 85ŌĆō90.

[4] V. Kuokkanen, T. Kuokkanen, J. Ramo and U. Lassi, Green and Sustainable Chemistry, 2013, 3(2), 89ŌĆō121.

[5] O. Sahu, B. Mazumdar and PK. Chaudhari, Environmental Science and Pollution Research, 2014, 21(4), 2397ŌĆō2413.


[7] A. Su├Īrez-Escobar, A. Pataquiva-Mateus and A. L├│pez-Vasquez, Catal Today, 2016, 266, 120ŌĆō125.

[8] S. Adamovic, M. Prica, B. Dalmacija, S. Rapajic, D. Novakovic, Z. Pavlovic and S. Maletic, Arabian Journal of Chemistry, 2016, 9(1), 152ŌĆō162.

[9] NT. Thuy, CT. Bao, DX. Son, UD. Bao, DV. Thanh and NN. Huy, Vietnam Journal of Science and Technology, 2017, 55(4C), 192.

[10] KP. Papadopoulos, R. Argyriou, CN. Economou, N. Charalampous, S. Dailianis, TI. Tatoulis, AG. Tekerlekopoulou and DV. Vayenas, J Environ Manage, 2019, 237, 442ŌĆō448.

[12] D. Valero, JM. Ortiz, V. Garc├Ła, E. Exp├│sito, V. Montiel and A. Aldaz, Chemosphere, 2011, 84(9), 1290ŌĆō1295.

[13] APHA-AWWA-WEF. Standard methods for the examination of water and wastewater. 21st ed. Washington, D. C, 2005.
[14] K. Bensadok, S. Benammar, F. Lapicque and G. Nezzal, Journal of Hazardous Materials, 2008, 152(1), 423ŌĆō430.

[15] . Metcalf and . Eddy, Wastewater engineering : treatment and reuse. McGraw-Hill, Boston, 2003.
[16] M. Kobya, H. Hiz, E. Senturk, C. Aydiner and E. Demirbas, Desalination, 2006, 190(1ŌĆō3), 201ŌĆō211.

[19] E. Mohora, S. Ron─Źevi─ć, B. Dalmacija, J. Agbaba, M. Watson, E. Karlovi─ć and M. Dalmacija, J Hazard Mater, 2012, 235, 257ŌĆō264.

[20] HQH. Phan, NX. Hoan, NN. Huy, NDD. Duc, NTN. Anh, NT. Que and NT. Thuy, Journal of Environment and Sustainability, 2019, 3(3), 127ŌĆō212.
[21] K. Thella, B. Verma, VC. Srivastava and KK. Srivastava, Journal of Environmental Science and Health, Part A, 2008, 43(5), 554ŌĆō562.

[22] ├ē. Fekete, B. Lengyel, T. Cserfalvi and T. Pajkossy, J Solid State Electrochem, 2016, 20(11), 3107ŌĆō3114.


[23] E. Bazrafshan, L. Mohammadi, A. Ansari-Moghaddam and AH. Mahvi, J Environ Health Sci Eng, 2015, 13(1), 74.
[25] QH. Nguyen, T. Watari, T. Yamaguchi, Y. Takimoto, K. Niihara, JP. Wiff and T. Nakayama, International Journal of Electrochemical Science, 2020, 15, 39ŌĆō51.


[26] B. Khaled, B. Wided, H. B├®chir, E. Elimame, L. Mouna and T. Zied, Arabian Journal of Chemistry, 2019, 12(8), 1848ŌĆō1859.

[27] BZ. Can, R. Boncukcuoglu, AE. Yilmaz and BA. Fil, J Environ Health Sci Eng, 2014, 12(1), 95.
[28] S. Bayar, Y. ┼×Y─▒ld─▒z, AE. Y─▒lmaz and ┼×. ─░rdemez, Desalination, 2011, 280(1ŌĆō3), 103ŌĆō107.