1. Introduction
Recent advances in room temperature ionic liquids (RTILs) or deep eutectic solvents (DESs) have created the possibility of replacing the traditional aqueous metal plating electrolytes, which are often toxic and raises concerns on its sustainability and environmental risks [1ŌĆō3]. DES-based baths offer some good engineering advantages for electroplating by having properties such as low vapor pressure, wide electrochemical window, good electrical conductivity, and high solubility for metal salts [4]. More importantly, they have emerged as a ŌĆ£greenerŌĆØ alternative since a majority of DESs are composed of biodegradable and less toxic compounds. There are four types of DES, classified according to chemical composition [5]: (i) Type 1 consists of quaternary ammonium salts (e.g. choline chloride, C5H14ClNO) with a metal chloride salt [e.g. zinc chloride, ZnCl2); (ii) Type 2 consists of quaternary ammonium salts with a hydrated metal chloride salt (e.g. aluminum chloride hexahydrate, AlCl3H12O6)); (iii) Type 3 has quaternary ammonium salts mixed with a hydrogen-bond donors (HBD) (e.g. carboxylic acid, amide or an alcohol); and (iv) Type 4 that has a hydrated metal chloride salts with a hydrogen bond donor. These mixtures are often used at eutectic concentrations, where the melting point is lowest [5].
A wide range of DESs have already been proven feasible as plating solutions to deposit a wide range of metals [6], including Cr [7ŌĆō9], Ni [10ŌĆō14], Zn [15], Sn [16], Cu [17], Ag [18], and Pd metal coatings and some Fe-based [19], Co/Cr [20], Ni/Cr [21], Ni/Co/ Sn, and Fe/Ni/Cr [22] alloy systems. Its inherently wide electrochemical window allows electrodeposition of elements such as Al, Ge, Si, and Ta, which are challenging to do in traditional aqueous plating techniques [23,24].
One common DES is ethaline, a Type 3 DES created by mixing choline chloride (ChCl) and ethylene glycol (EG) at a 1:2 molar ratio. Ethaline has already been employed for the successful electrodeposition of different metals including Ag, Cr, Ni, Zn, Cr-P, ZnŌĆōSn, and Cu composites, [7,25], and even for nanomaterials [23]. For example, Pereira et al. [15] observed thin Zn nanoplatelets growing perpendicular in relation to the electrode surface using an ethaline-based Zn plating bath. Abbott et al. [13] reported nanocrystalline Ni deposits created from ethaline-based Ni baths. These deposits exhibited lower surface roughness and higher hardness (100 HV harder) than deposits obtained from traditional aqueous-plated Ni coatings.
The first-generation ionic liquids have been known to be air and water-sensitive - which limits its applicability in electroplating - while DES have been known to be air-stable and water-tolerant [24]. Recently, more studies have investigated how water uptake influences the different properties of DESs.
The presence of hydrophilic groups in the choline chloride molecule is found to be responsible for the high hydrophilic behavior of most choline chloride-based DES [25ŌĆō29]. These DESs are also considered hygroscopic, substances that have the tendency to absorb moisture from the atmosphere if left exposed. For example, choline chloride-urea DES (aka reline) can absorb atmospheric moisture up to a water concentration of 40 wt% [30].
The contamination of DESs with water has a significant effect on its properties. In general, water in DESs affects its chemical structure [31], decreases its kinematic viscosity, increases its electrical conductivity, and notably narrows its electrochemical window (EW). Interestingly, some studies have intentionally added water to DES baths to enhance plating mechanics by taking advantage of the improved conductivity and reduced viscosity [9,32,33].
In contrast, the narrowing of the EW due to presence of water can have substantial consequences for electroplating applications. The electrochemical window defines the range of potential at which the solution is stable, and electroplating is most feasible. A high EW is desirable to allow plating of metals which has high oxidation potentials and require considerable energy for reduction. The typical EW of water is 1.2 V [24]. Du et al. [30] observed a decrease in the electrochemical window (vs. Ag) from 2.54 V for pure reline to 1.03 V when the water content was at 12 wt%. However, at 6 wt% water, there was no observed change in the EW of reline and improvements viscosity, conductivity, and current efficiency were noted. Nickel deposited under this system exhibited a more uniform and compact surface with numerous finer pyramid-like clusters compared to water-free reline [30]. Bobrova et al. reported that an increase in water content in the CrCl3-xH2O and choline chloride DES contributed to the early onset of hydrogen evolution reaction (HER) and led to a drop in the plating current efficiency. The presence of hydrogen gas is undesirable since it can induce hydrogen embrittlement in the substrate and coating [11]. Furthermore, absorbed water could result to decomposition of DES leading to undesirable side reactions [27].
Though the water-tolerance of DES and the ensuing consequence of water content on its chemical properties are well known, there are few studies that details the moisture uptake of DES at typical ambient conditions. From the aspect of electroplating applications, uncontrolled water uptake of the DES bath can possibly alter deposit properties [29]. This uncertainty in deposit quality can easily hinder the commercial adoption of DES as plating bath by the metals industry. Thus, investigation of DESŌĆÖs water uptake from the environment at different storage conditions is worth investigating and will have practical value to electroplaters. Moreover, techniques for the removal of absorbed water also needs consideration, especially if water degrades the relevant DES properties. Cammarata et al. [34] reported that simple heat treatment can indeed remove moisture in ILs. Li et al. [27] reported the use of N2 bubbling and vacuum drying for moisture removal. Nitrogen bubbling was found to remove water effectively from [HOEMIM]Otf ionic liquid as interpreted from the increase of EW to 5.77 V from 5.38 V (fresh sample, with initial water content). Vacuum drying at 60┬░C resulted to an EW of 5.50 V[24]. Du et al. [30] employed solution heating at 80┬░C to remove water from reline containing 40 wt% water, and reached a low of 1.5 wt% water content after 18 hours. These simple heat treatment strategies to remove absorbed water and return the water-saturated DES to its starting ŌĆ£dryŌĆØ state is of great importance to widen the application window of DESs.
Based on the existing studies above, this study presents the room temperature moisture absorption behavior of ethaline (1:2 ChCl:EG) over a period of 14 days at two storage conditions. The ethalineŌĆÖs water content, viscosity, conductivity, and electrochemical window were monitored during this period. 1H NMR spectroscopy was used to assess the incorporation of water molecules into the chemical structure of ethaline. Lastly, solvent heating was employed as an attempt to remove the absorbed water and recover the initial properties of ethaline.
2. Methodology
2.1 Chemicals, Materials and Synthesis
Choline chloride (ChCl) crystals (Sigma Aldrich, 98%) were oven-dried at 70┬░C overnight [35] and ethylene glycol (EG) (Ajax Finechem, >95%) was heated at 70┬░C on a magnetic stirrer (EchoTherm HS10-2, USA) for 3 hours to remove initial water content of the reagents prior to synthesis and moisture uptake experiments.
Ethaline was prepared by mixing ChCl and EG at 1:2 molar ratio and heated to 70┬░C until a clear, homogeneous liquid had formed. The prepared ethaline was allowed to cool to room temperature.
2.2 Moisture uptake experiments
Twenty-five (25) mL aliquots were transferred into centrifuge tubes (7.07 cm2 exposed area) for assessment of moisture uptake. Half of the samples was capped and stored inside a glass container with silica desiccants at 18ŌĆō20% relative humidity (RH), as shown in Fig. 1(a). This condition simulates the case in an electroplating plant where the bath is stored in a controlled condition. The other half was left uncapped and exposed to ambient atmosphere (27┬░C) at 55ŌĆō60%RH, as shown in Fig. 1(b). This condition simulates typical electroplating plant operating conditions where the electroplating bath is left open and exposed to air.
2.2. Characterization of DES
For both storage conditions, the water content, viscosity, electrical conductivity, and electrochemical window of ethaline were monitored over a two-week period (14 days).
Water content (wt%) was determined using a volumetric Karl Fischer titrator (TitroLine 7500KF, SI Analytics, Germany). Sample solution was manually mixed to homogenize before titrating. Three drops or 0.08 ŌĆō 0.1 g of sample were added to the dry methanol solvent (HYDRANALŌäó, max 0.01 m% water) and titrated with the appropriate titrant (HYDRANALŌäó, 4.5ŌĆō5.5 mg/mL water) for moisture determination.
Kinematic viscosity was measured at room temperature using a rotational viscometer (VISCOŌäó-6800, Atago Co., Japan) employing A1S setting (composed of A1 spindle type and S-type beaker) at 150 rpm. Electrical conductivity was measured at room temperature using a conductivity meter (LAQUAact-EC120, Horiba, Japan). Measurements were done using a titanium-platinum black conductivity electrode (K = 1 cmŌłÆ1) (LAQUA 9382-10D, Horiba, Japan) as probe.
Cyclic voltammetry (CV) was performed to observe the changes in the electrochemical window of the DES at different storage conditions. CV measurements were performed at room temperature using a potentiostat (BioLogic SP-150, France) employing a three-electrode system composed of a glassy carbon electrode (area: 7.07 mm2) (ALS Co., 002012, Japan) as the working electrode, platinum electrode (area: 2.01 mm2) (ALS Co., 002013, Japan) as the counter electrode and silver wire (diameter: 1.5 mm) for the reference electrode. The electrodes were polished using 0.5 um alumina before the start of measurement. A scan rate of 10 mV/s in the scan range of ŌłÆ1.9 V to 1.4 V [27] was employed.
Incorporation of water into the structure of ethaline was observed using proton nuclear magnetic resonance (1H NMR) spectroscopy (Agilent 400 MHz NMR, USA) at 400 MHz. A 1:2 volume ratio of ethaline sample to deuterated water solvent (D2O) was employed in preparing 600 uL of the sample inside a 5-mm NMR tube (WG-1235-7, Wilmad Lab-Glass, USA). The 1H NMR spectra were recorded at 32 scans and an average acquisition time of 2 minutes.
2.3 Water removal experiment
Experiments were conducted to determine if the absorbed water from the environment may be removed from the DES. At the end of the 14th day of storage, the DES was heated at 100┬░C at different durations and allowed to cool to room temperature. The water content was then measured using the Karl Fischer titrator, while the electrochemical window was again assessed via cyclic voltammetry.
3. Results and Discussion
3.1 Moisture absorption of ethaline
The synthesized ethaline was initially tested for its water content. Results show that the as-prepared solution had 2.4 wt% water, even after thorough drying of precursor materials. This confirms hygroscopicity or the inherent tendency of ethaline to absorb water from the atmosphere.
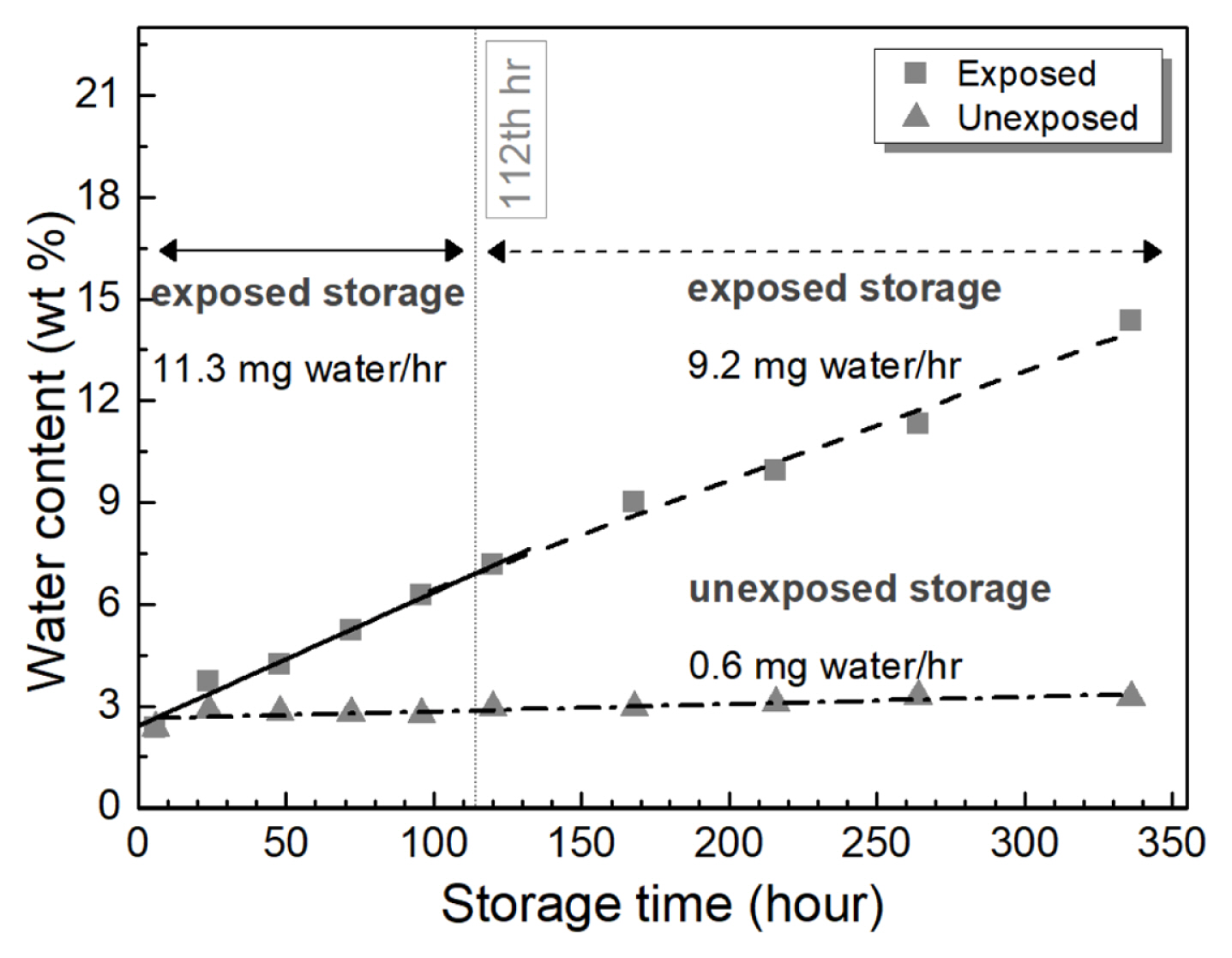
Fig. 2 compares the water uptake of ethaline at the different exposure conditions. For the ethaline exposed in air, the water content increased with time. Two regions of water absorption behavior were observed, with the transition point occurring at 112 hrs. Before 112 hrs, the linear rate of moisture absorption of ethaline was 11.3 mg hrŌłÆ1. After which, the rate of absorption slowed by about 20% to 9.2 mg hrŌłÆ1. After two weeks, the water content of ethaline in the exposed condition reached a maximum of 14.3 wt%, which is about 6 times higher than its initial value.
In contrast, the moisture absorption rate of the unexposed ethaline is significantly lower than its exposed counterpart. The absorption rate was essentially steady at only 0.6 mg hrŌłÆ1. At the end of two weeks, the moisture content was 3.3 wt%.
1H NMR spectroscopy was employed to determine which functional groups in ChCl-EG complex were responsible for moisture absorption. Measurements were taken at certain time points over a 14-day period. The relevant spectra are shown in Fig. 3, and the corresponding chemical shifts (ppm) and their associated hydrogen type are listed in Table 1. The peaks of the aliphatic hydrogens in ChCl and EG overlapped at 3.35ŌĆō3.40 ppm [37,38] while the rest are separated and can be identified: 3.05ŌĆō3.10 (s, 9 HŌĆÖs, ŌłÆCH3 of ChCl ), 3.50ŌĆō3.55 (s, 1H, ŌłÆOH of ChCl), 3.90ŌĆō3.95 (m, 2 HŌĆÖs, O-CH2- of ChCl), 4.60ŌĆō4.65 (s, 2 HŌĆÖs, water), and 5.555 (d, ŌłÆOH in EG) [38,39].
Significantly increasing peak intensities observed on chemical shifts (d) 3.05ŌĆō3.10, and (a) 3.50ŌĆō3.55 are interpreted to be the functional groups where water is likely to form hydrogen bonds and/or dipoledipole interactions. This result suggest that the presence of hydrophilic groups, ClŌłÆ, ŌłÆCH3, and ŌłÆOH [29,41], in the ChCl-EG complex promotes its moisture absorption property, consistent with other works [26,42,43].
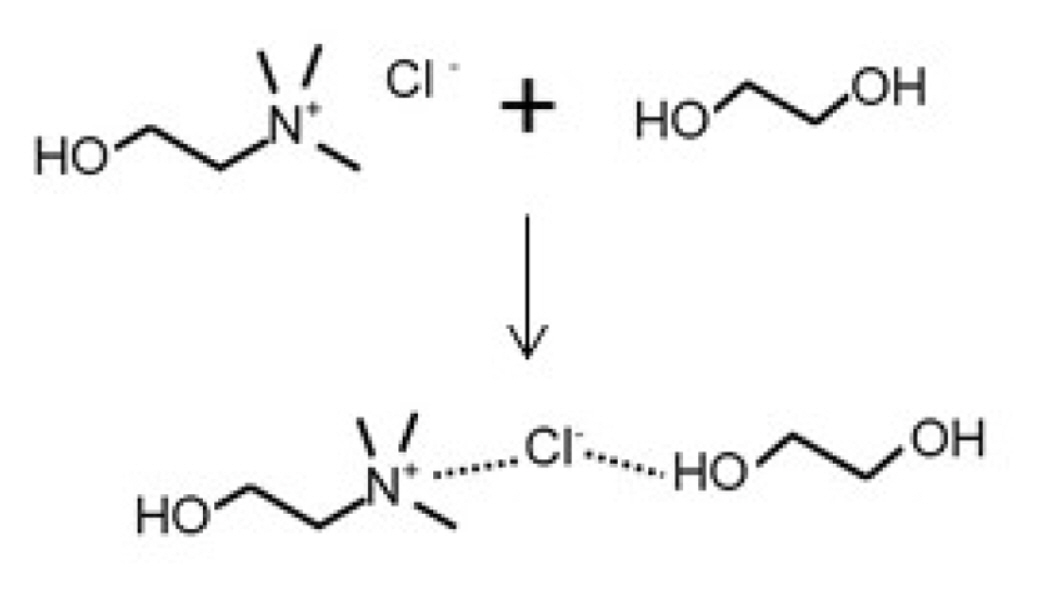
The complexation mechanism of ChCl and EG is shown in Fig. 4. Hydrogen bonding is established between the chloride anion and the acidic proton on the ŌłÆOH groups of EG forming a complex anion represented by [EG]ClŌłÆ. Ionic interaction dominates between the choline (Ch+), and [EG]ClŌłÆ anion forming a ChCl-EG complex [36]. In this molecule, the two ŌłÆOH groups and the central ClŌłÆ ion are clearly present [26,43]. Chen et al. [26] confirmed that an increase in hydroxyl ions in the molecule enhances a DESŌĆÖ water absorption ability. Aside from the number of hydroxyl groups, they observed that other factors such as methyl groups (e.g. ChCl:methylurea vs ChCl:urea) and chain length can also influence water absorption.
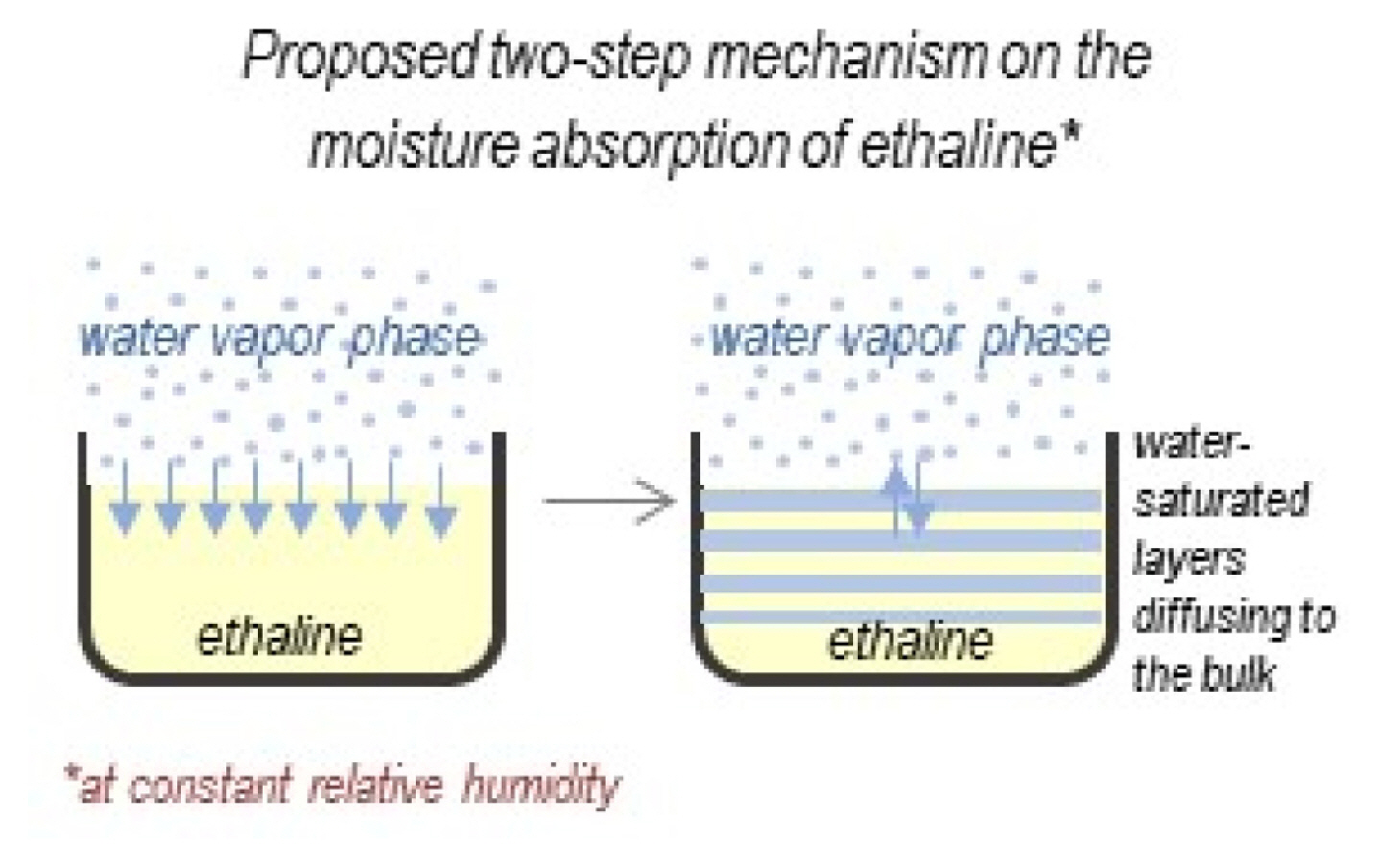
Cao et al. [28] proposed a two-step process mechanism on the moisture absorption of DESs: (1) water vapor molecules from the atmosphere are attracted and absorbed on the surface of the DES, and, after saturation, (2) water moves from the surface to the bulk DES via diffusion as shown in Fig. 5. They observed that a quick shake test reveals the viscosity variation from various levels of the solvent from the surface. This is interpreted as the diffusion of water-saturated layers down to the bulk solution.
Fig. 2 suggests a lowering of the moisture absorption rate upon prolong exposure providing an insight on the water saturation capacity of DESs. Du et al. [30] noted that a hygroscopic ChCl:urea mixture absorbed a wt% moisture (~40 wt%) that was equivalent to the relative humidity (~40% RH) of the test environment. This suggests that it is likely that the rate of moisture uptake of the ethaline will slow down beyond the 14-day period and will absorb a maximum of 18 wt% water similar to the test roomŌĆÖs RH.
The negligible change in the water content of the ethaline stored in the controlled environment gives a simple procedure on how to control or limit the hygroscopicity of the DES. This procedure may be considered by future electroplating plants that will deal with hygroscopic ILs.
3.2 Effect of water on the physical properties of ethaline
The kinematic viscosity and electrical conductivity of the ethaline at the different storage conditions were also measured within a 14-day period, and results are shown in Fig. 6. The freshly synthesized ethaline was initially assessed and exhibited a viscosity of 39.5 mPa-s and electrical conductivity of 8.8 mS┬ĘcmŌłÆ1. Published values for the viscosity and conductivity of ethaline are 36ŌĆō37 mPa┬Ęs [5,44] and 7.61 mS┬ĘcmŌłÆ1 [5], respectively. It is likely that the deviation observed here from other published values can be attributed to the difference in the water content of ethaline due to difference in fabrication and purification routes. Thus, this observation suggests the importance of including a report on the water content of DESs for every production.
For the ethaline exposed in air (Fig. 6(a)), results show a notable decrease in viscosity, with two linear decline rates defined at the same 112 hrs transition point. The rate of viscosity decline was initially 0.07 mPa┬Ęs hrŌłÆ1, and decreased to 0.03 mPa┬Ęs hrŌłÆ1 after 112 hours. After 14 days, the viscosity of ethaline was lower by about 38%, from 39.5 mPa┬Ęs to 24.6 mPa┬Ęs. In contrast, the unexposed ethaline exhibited negligible change in viscosity.
Fig. 6(b) indicate that the electrical conductivity of the ethaline exposed to the atmosphere steadily increased over the 14-day period. Again, two regions of conductivity enhancement were observed. Initially, the conductivity increased at a rate of 18.5 ╬╝ScmŌłÆ1 hrŌłÆ1. After 112 hours, the rate slowed to 13.6 ╬╝ScmŌłÆ1 per hour. At the end of 2 weeks, the electrical conductivity has increased by about 37% (i.e. from of 13.9 mScmŌłÆ1 to 8.8 mScmŌłÆ1). For the unexposed ethaline, there were no significant changes in conductivity observed.
The properties that dictate the mass and charge transport efficiency of an electrolyte are its kinematic viscosity and electrical conductivity. Du et al. [30] was also able to confirm the relationship between electrical conductivity (s) and fluidity (h) in DESs using the equation:
Clearly, there is a strong correlation between the observed changes in viscosity and conductivity to the water uptake of ethaline during the test period, as seen in Fig. 2 and Fig. 6. The same bi-modal behaviour was observed, coinciding at a similar transition point (112 hrs).
This relationship between the viscosity and conductivity of the DES and water content may be explained by the hole theory model [8,41]. According to this theory, DESs contain void spaces arising from thermally generated fluctuations in local density, which have random size and position and are constantly moving. An ion can only move if it is adjacent to a hole of equal or larger size. Therefore, an ionŌĆÖs mobility, and consequently the DESŌĆÖ viscosity and conductivity, depends on having these appropriately-sized voids or spaces. Bobrova et al. [8] proved that increasing water content of DES resulted to a significant increase in the average void radius, which then promotes the mobility of ions in the system. DŌĆÖAgostino et al. [45] also reported on the diffusivity of the components of ethaline as influenced by water content. In pure ethaline, the EG molecule moves faster since the relatively large size of the ChCl molecule reduces its mobility. In water-bearing ethaline, both the diffusivity of EG and ChCl is increased, indicating the strong influence of water on the inter-molecular interactions of the ethalineŌĆÖs components.
3 .3 Effect of water on the electrochemical window of ethaline
The electrochemical window (EW) plays a crucial role in using DESs in the electrodeposition of metals and alloys. EW is a good indicator of the bathŌĆÖs electrochemical stability. The EW is defined as the electrochemical potential range over which the electrolyte is neither reduced nor oxidized at an electrode, and is determined from the equation [24]:
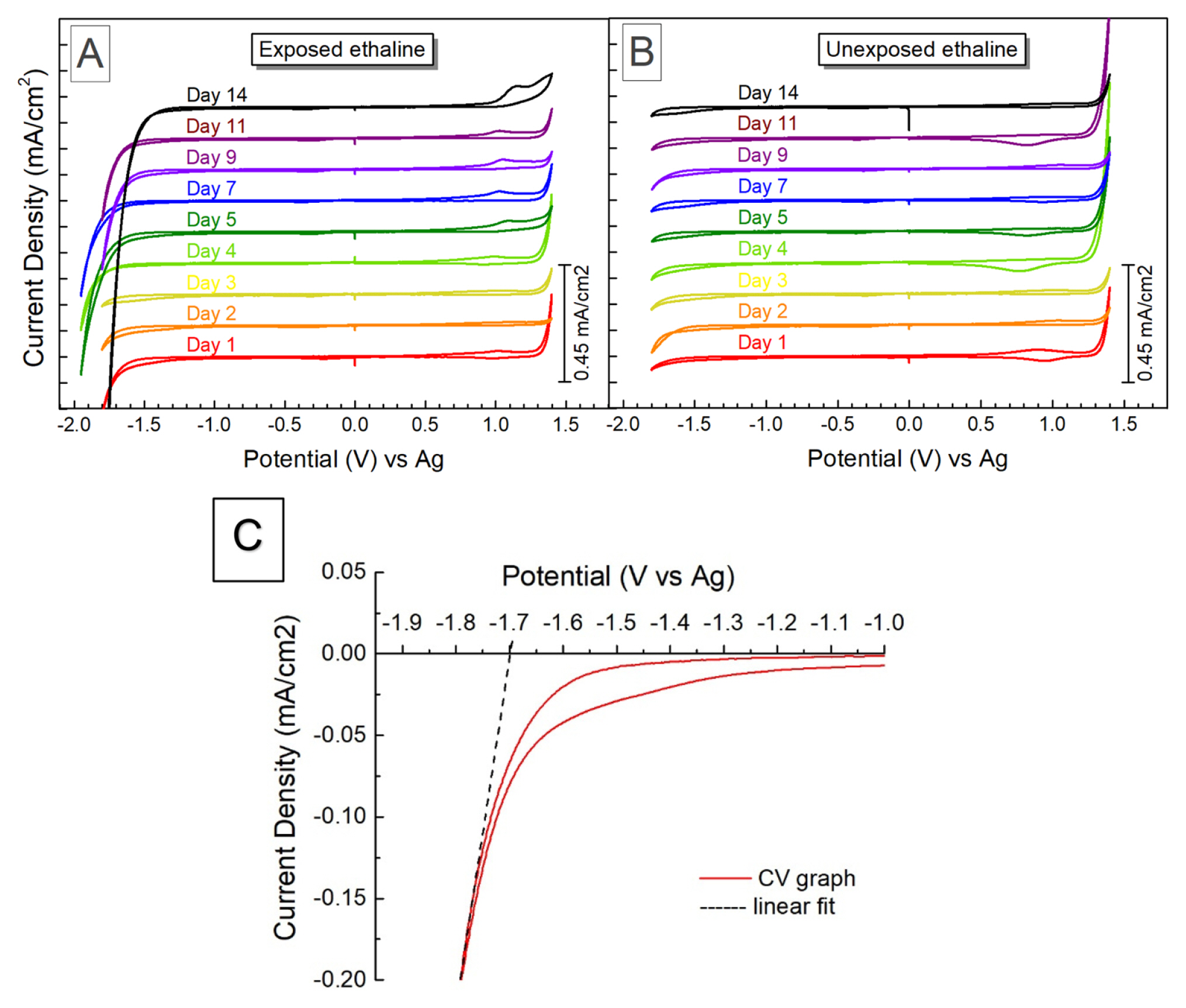
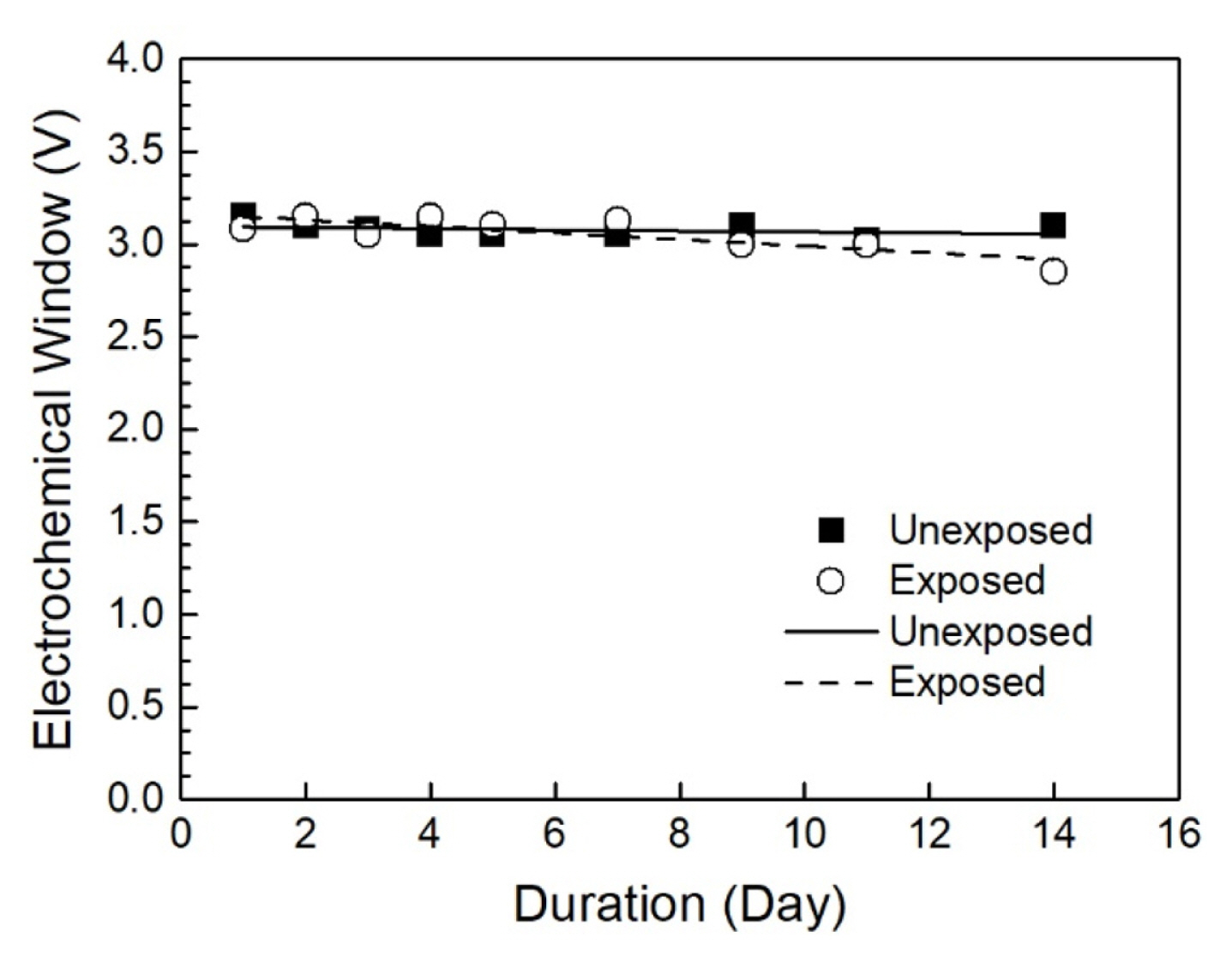
where Ered is defined as the onset of the reduction potential (cathodic limit), and Eox is defined as the onset of the oxidation potential (anodic limit). The cathodic and anodic potential limits were obtained via linear fit method recommended elsewhere. The potential limit for either cathodic or anodic reactions is defined as the intersection of the linear fit of the currentŌĆōvoltage relationship after the onset of electrochemical reduction of the electrolyte at the potential axis positioned at 0 mA/cm2 (Fig. 7c) [46,47]. The obtained values for the cathodic and anodic potentials are summarized in Table 2.
Fig. 7 presents the cyclic voltammogram measurements of the ethaline stored at different conditions over the 14-day test period, while Fig. 8 is the corresponding plot of the derived EW as a function of storage time. The as-prepared ethaline reveals a broad electrochemical window of 3.15 V. The cathodic limit potentials were determined by the reduction of Ch+ to triethylamine at about ŌłÆ1.8 V (vs Ag), according to the reaction [48]:
and the anodic limit potential from the oxidation of ClŌłÆ to Cl2 gas at about 1.35 V (vs Ag), [48]:
For the ethaline exposed to the environment, a narrowing of the EW was observed over time. The specimen exhibited a 7.5% drop in EW, from 3.08 V to 2.85 V by the end of Week 2, as seen in Fig. 8. In contrast, the unexposed ethaline had minimal changes in EW, from 3.15 to 3.10 V. Again, this change in EW coincides with the DESŌĆÖ water uptake during the test period. Water can reduce the EW of the DES via the occurrence of electrolytic decomposition reactions, such as the hydrogen evolution reaction [30,49].
3.4 Water removal via solution heating
Considering the hygroscopic nature of the DES and the ensuing effects of water absorption on the DESŌĆÖ properties and performance, it is important to adopt simple water removal strategies that could restore the solventŌĆÖs original properties.
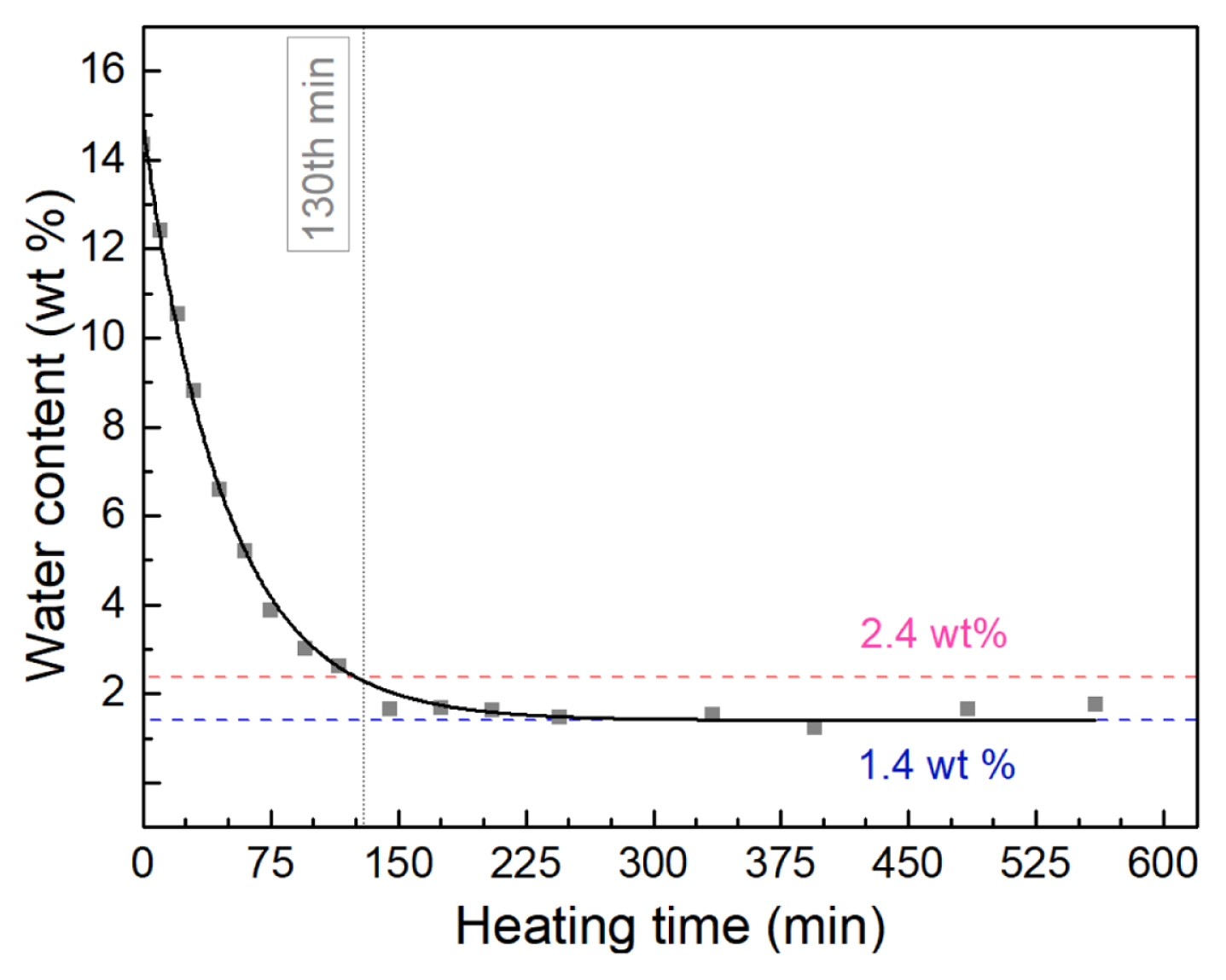
Fig. 9 shows the measured water content in the ethaline that was exposed to air for 14 days after heating at different durations. Evidently, solvent heating at 100┬░C was successful in removing the absorbed water in ethaline. The starting water content value of 2.4 wt% was obtained after a heating time of about 2 hours, as determined graphically in Fig. 9. Further heating caused the water content in the DES to continue to drop and eventually level. The onset of the water content plateau of 1.4 wt% started after about 3 hours of heating and was maintained up to 9 hours of heating.
Fig. 10 shows that the EW of the water-laden DES increased after the applied heat treatment from 2.85 V (EW of Day 14 ethaline sample) to 3.0 V. This value is about 95% of the EW of the freshly prepared ethaline of 3.15 V. This indicates that heating did not only remove water but also allowed the successful recovery of the DES properties.
However, prolonged heating of up to 9 hours had an unwanted downside as the ChCl recrystallized upon cooling down to room temperature. The recrystallization of ChCl is a strong indication that the heated ethaline has strayed from its eutectic concentration (i.e. 1:2 molar ratio of ChCl:EG). Rusdi et al. [50] reported that increasing water concentration in an EG-water mixture increases the evaporation rate and decreases the activation energy for the vaporization of the binary system. Hence, it is likely that the combination of water and prolonged heating caused the evaporation of some of the EG and induced the precipitation of ChCl.
The above results confirm the hygroscopicity of ethaline, and the property changes associated with moisture absorption of the DES. As mentioned, the ability of water to positively influence viscosity and conductivity of DES has already been used to improve electroplating results [9,32,33]. However, uncontrolled water uptake can lead to unwanted consequences. For example, Protsenko et al. [9] noted that beyond a critical amount of water content, the current efficiency of a ChCl-based chromium plating bath significantly dropped.
Clearly, when used in practical electroplating applications, the handling of ethaline should be an important consideration. Procedures should aim to minimize or control the water intake of the DES to obtain predictable solvent performance. Cao et al. [28] was able to identify other factors affecting hygroscopicity of DES. Temperature promotes diffusion in DES, thereby enhancing moisture absorption. This is important to monitor since the synthesis and electrodeposition steps are done at elevated temperatures for an extended period. Relative humidity, which describes the percentage of water vapor in the air, also has a direct relationship with the DESŌĆÖ water absorption [30]. Thus, humid environments will cause moisture absorption rates to increase. Electroplating operations, especially in the tropical regions, can undergo drastic humidity fluctuations all year round. Therefore, seasonal handling and storage protocols for DESs should be developed. A large, exposed surface area of the DESs will promote hygroscopicity and the faster incorporation of moisture into its bulk system; therefore, capped and narrow-opening mixing, electroplating and storage tanks are suggested for use in this situation.
Water removal strategies must also be integrated in this operation as this will greatly affect the recyclability and cost of the DESs. However, the vaporization mechanism of these DESs must be understood completely as to avoid the evaporation of other components, such as observed in the current study where ChCl recrystallized due to EG evaporation. These deviations from the eutectic composition will subsequently cause alterations in the properties and performance of the solvent.
4. Conclusions
The hygroscopicity of synthesized ethaline was investigated under two storage conditions, one exposed to the atmosphere and another unexposed or sealed, over a two-week period. When exposed to air, ethaline displayed notable hygroscopicity. Initially the rate of water uptake was high. At about the 4-day mark, the rate of uptake slowed by about 20%. Minimal water uptake was observed in the sealed or unexposed ethaline. 1H NMR spectroscopy revealed that ethalineŌĆÖs considerable hygroscopicity may be traced to the presence of hydrophilic groups, ClŌłÆ and ŌłÆOH, in the ethaline molecule.
With the increase in water content, the ethalineŌĆÖs viscosity decreased while the conductivity increased. This is likely due to waterŌĆÖs ability to increase molecule separation distance and expand the hole volume in ethaline, as well as its effects on the ChCl-EG molecular interactions. Furthermore, the EW of the exposed ethaline was reduced as a consequence of water uptake. This decrease in EW was due to the occurrence of water decomposition reactions such as the hydrogen evolution reaction.
Solution heating at 100┬░C was able to remove 80% of the absorbed water within two hours, and the EW was restored to near original values.
Hygroscopicity has been proven to affect the properties and possible electroplating performance of ethaline, and this likely applies to most of the DES that are hydrophilic by nature. Hence, appropriate handling, storage, and water removal strategies should be developed to acquire predictable performance and enhance the DESŌĆÖ viability in commercial applications.
5. Declarations
5.1 Funding
The authors would like to acknowledge the Philippine Council for Industry, Energy, and Emerging Technology Research and Development (PCIEERD) of the Department of Science and Technology (DOST) funded project entitled ŌĆ£Establishing an Environment-Friendly Chromium Plating Process using New Generation Ionic liquidsŌĆØ and the Science Education Institute ŌĆō DOST.