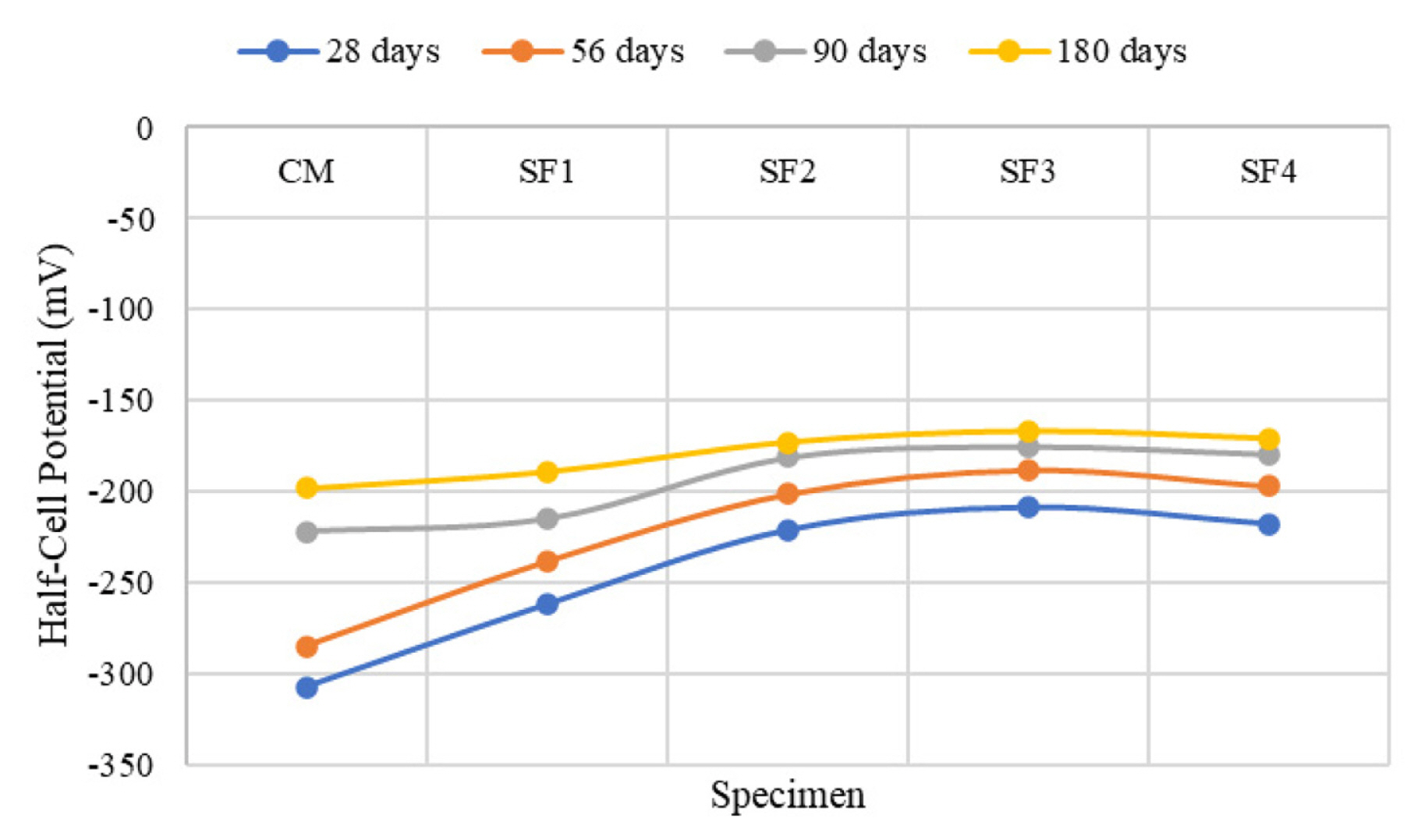
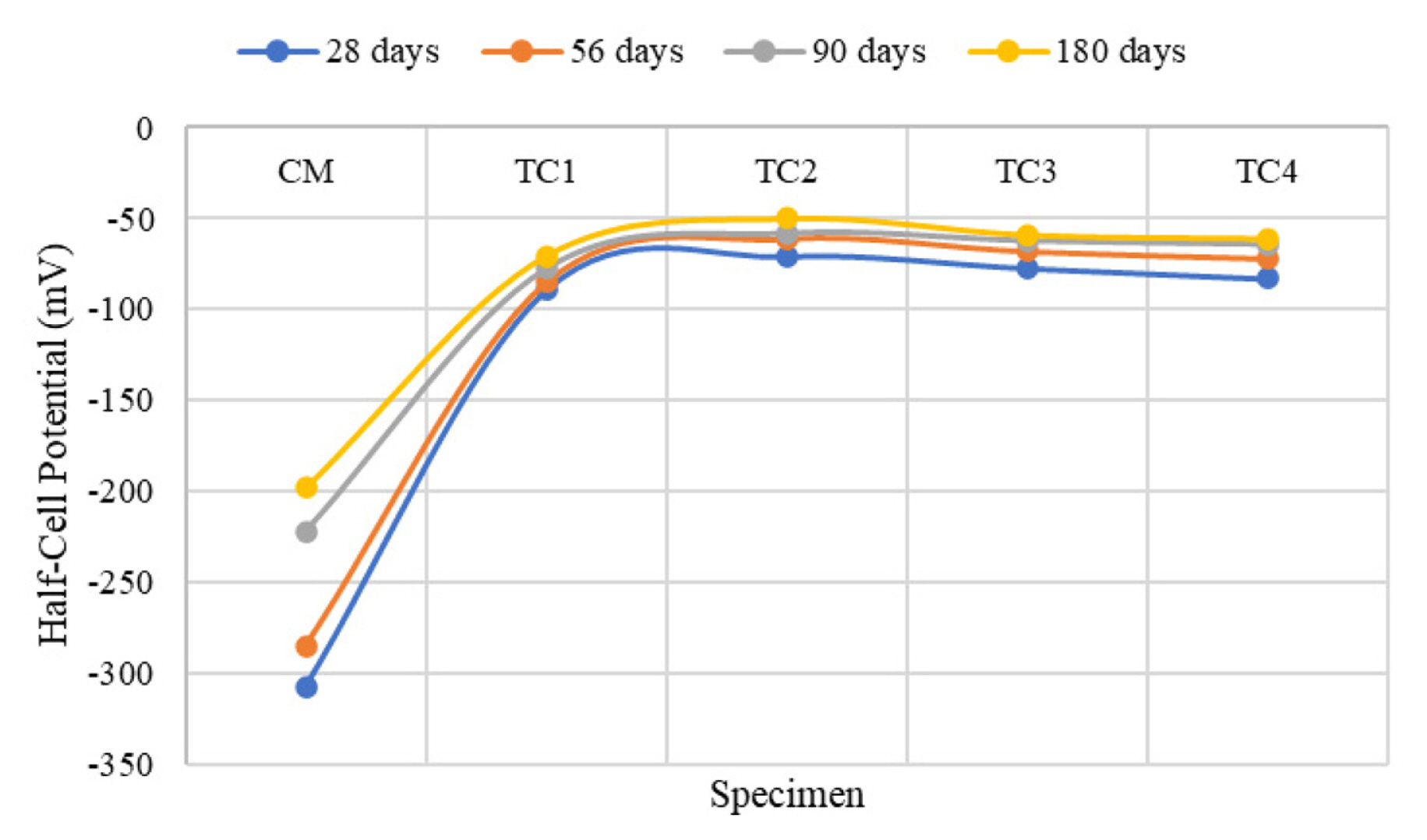
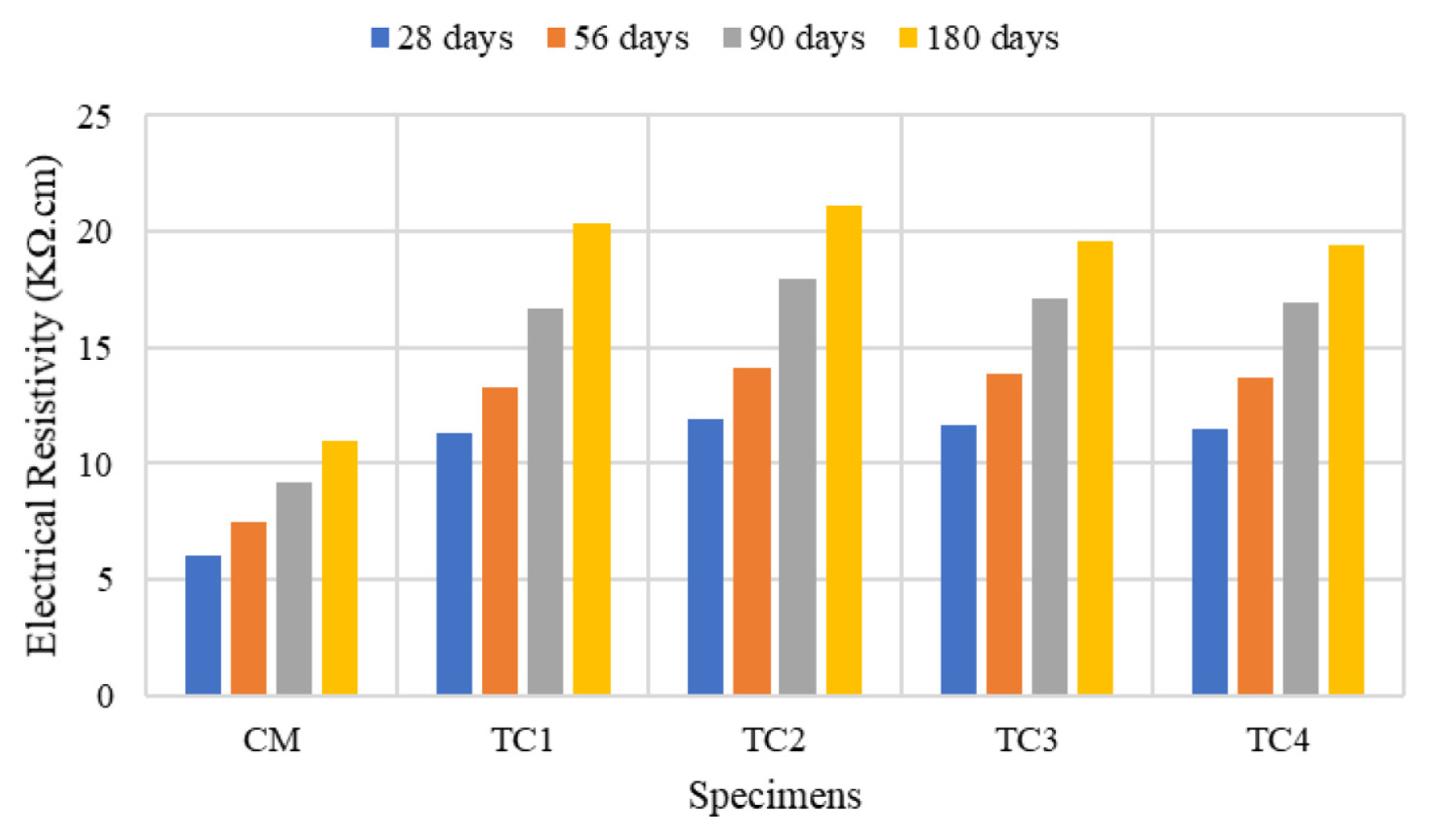
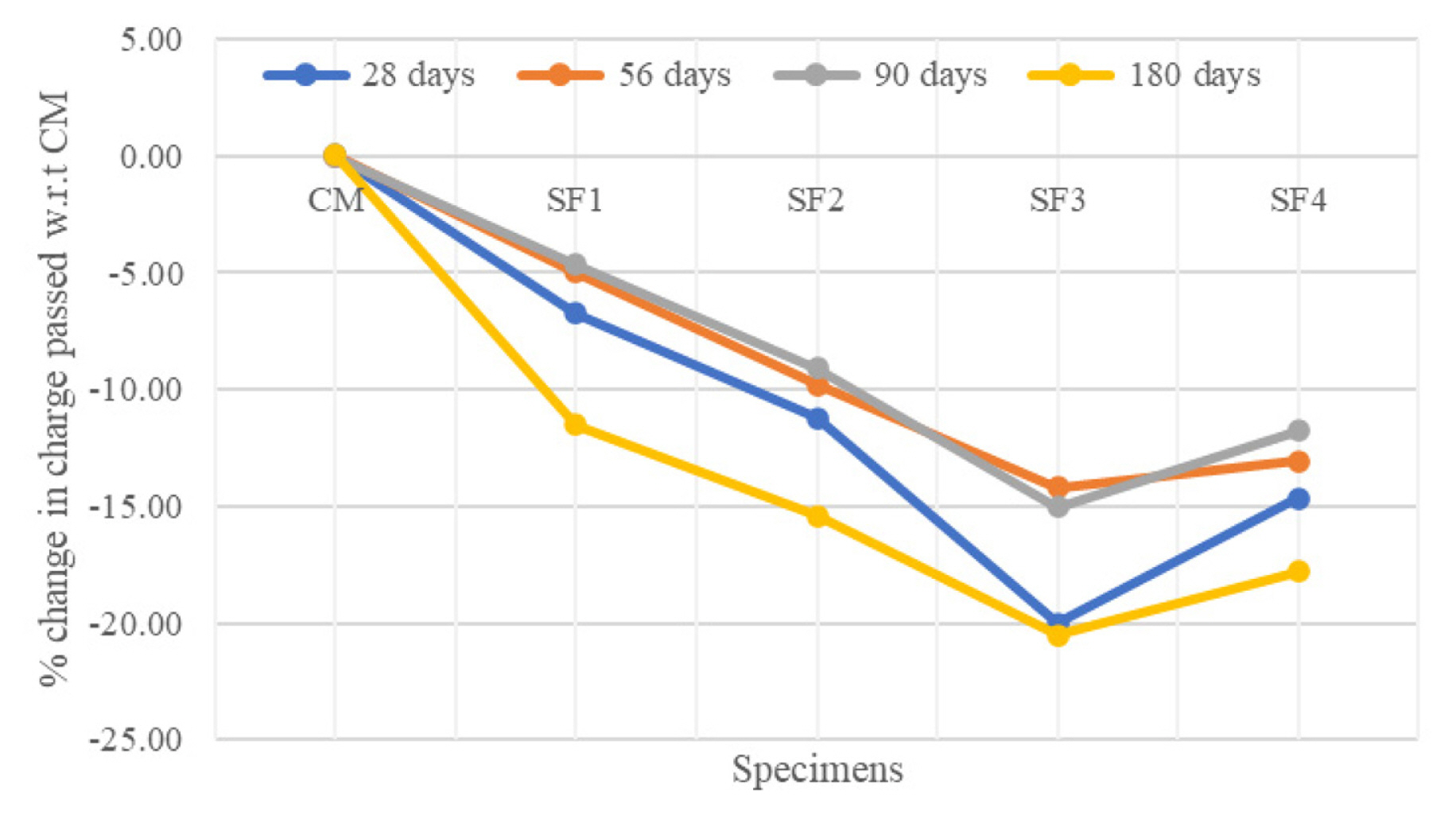
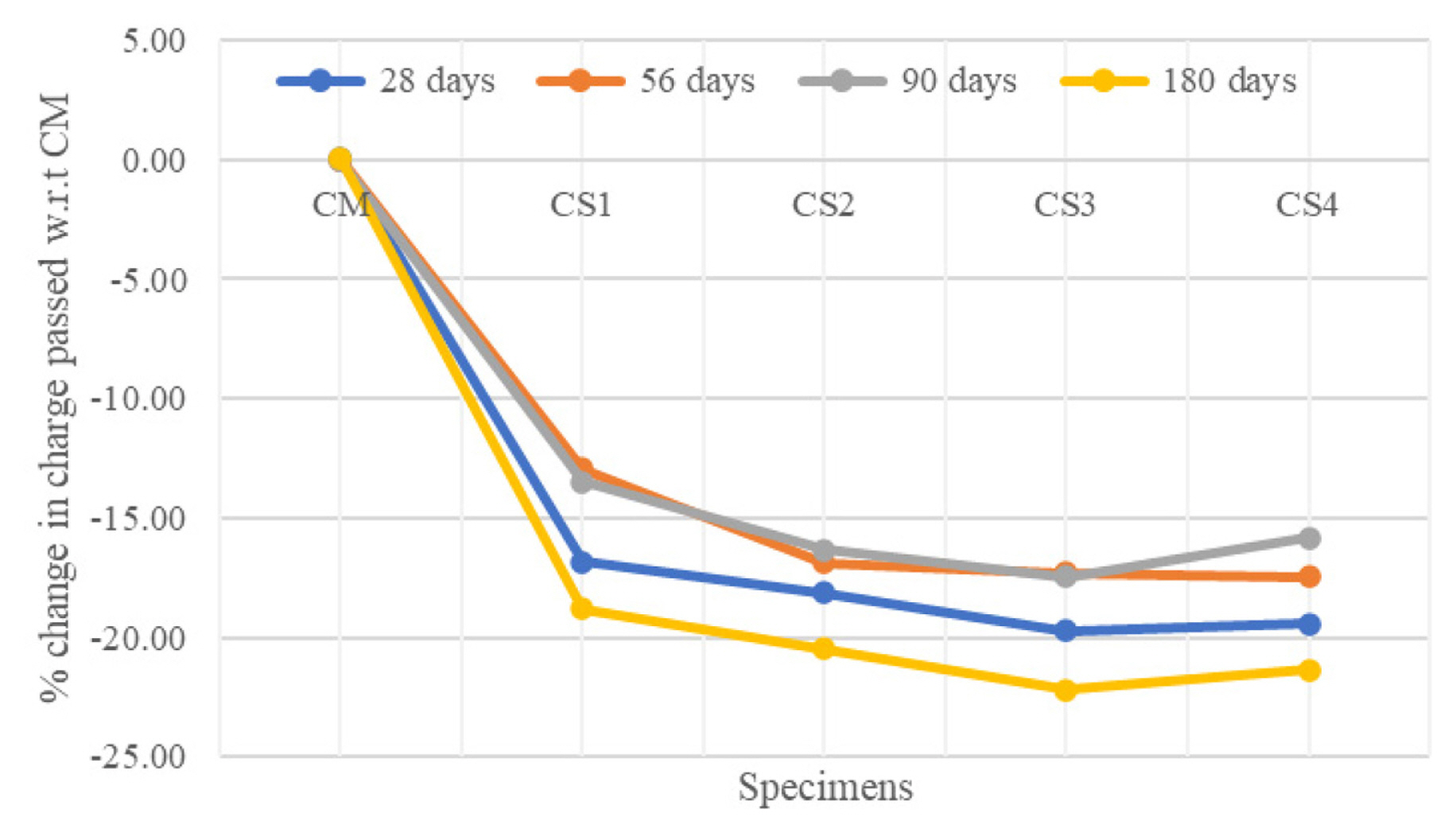
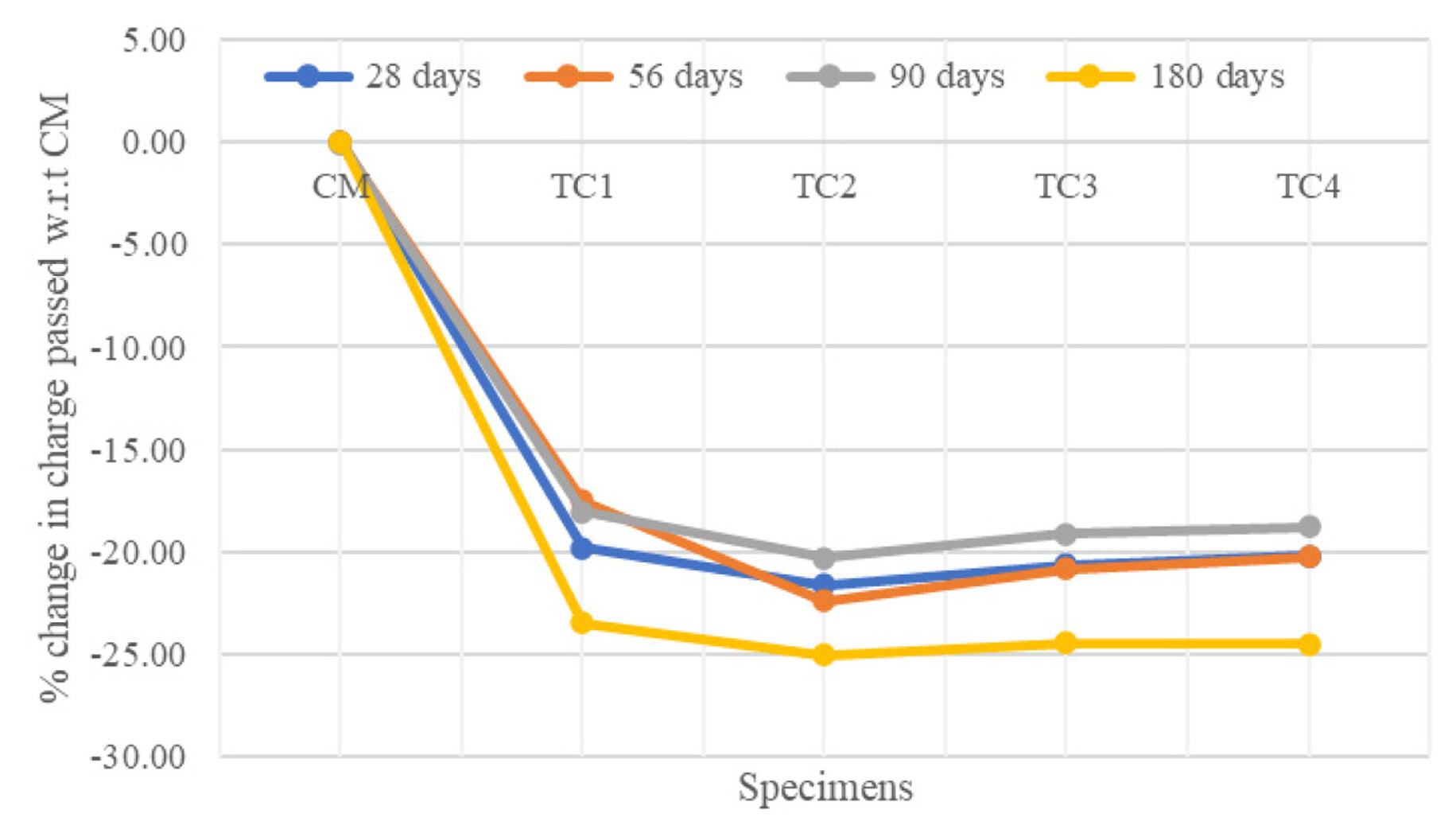
[1] A. Leemann, P. Nygaard, J. Kaufmann and R. Loser,
Cem Concr Compos,
2015,
62, 33ŌĆō43.

[2] JHM. Visser, Heron, 2012, 57(3), 231ŌĆō247.
[3] VG. Papadakis,
Cem Concr Res,
2000,
30(
2), 291ŌĆō299.

[4] L. Qin and X. Gao,
Fuel,
2019,
245, 1ŌĆō12.

[5] BS. Dhanya, S. Rathnarajan, M. Santhanam, RG. Pillai and R. Gettu, Indian Concr J, 2019, 93(4), 10ŌĆō21.
[6] S. Rukzon and P. Chindaprasirt,
Indoor Built Environ,
2009,
18(
4), 313ŌĆō318.

[7] O. Cizer, K. Van Balen, D. Van Gemert and J. Elsen, In: Sustain Constr Mater Technol - Int Conf Sustain Constr Mater Technol; 2007; pp 611ŌĆō621.
[8] L. Senff, D. Hotza, WL. Repette, VM. Ferreira and JA. Labrincha,
Adv Appl Ceram,
2010,
109(
2), 104ŌĆō110.

[9] LG. Li, ZH. Huang, J. Zhu, AKH. Kwan and HY. Chen,
Constr Build Mater,
2017,
140, 229ŌĆō238.

[10] . Atta-ur-Rehman, A. Qudoos, SH. Jakhrani, HG. Kim and J-S. Ryou, Int J Concr Struct Mater, 2019, 13(1), 35.
[11] LG. Li, J. Zhu, ZH. Huang, AKH. Kwan and LJ. Li,
Constr Build Mater,
2017,
140, 229ŌĆō238.

[12] A. Agarwal, S. Bhusnur and T. Shanmuga Priya,
Mater Today Proc,
2019,
22, 1433ŌĆō1442.

[13] R. Garg, M. Bansal and Y. Aggarwal, IJERT, 2016, 5(04), 16ŌĆō19.
[14] R. Garg, R. Garg, M. Bansal and Y. Aggarwal,
Mater Today Proc,
2020,
43, 769ŌĆō777.

[15] R. Garg and R. Garg,
Mater Today Proc,
2021,
43, 778ŌĆō783.

[16] D. Han and H. Kim,
J Asian Archit Build Eng,
2020,
19(
4), 305ŌĆō314.

[17] L. Sadowski, Sci World J, 2013, 2013.
[18] L. Wang, D. Zheng, S. Zhang, H. Cui and D. Li,
Nanomaterials,
2016,
6(
12), 241.

[19] MESI. Saraya,
Constr Build Mater,
2014,
72, 104ŌĆō112.

[20] C. Hu, Y. Han, Y. Gao, Y. Zhang and Z. Li,
Mater Charact,
2014,
95, 129ŌĆō139.

[21] I. Ismail, SA. Bernal, JL. Provis, R. San Nicolas, DG. Brice, AR. Kilcullen, S. Hamdan and JSJ. Van Deventer,
Constr Build Mater,
2013,
48, 1187ŌĆō1201.

[22] C. Zhuang and Y. Chen,
Nanotechnol Rev,
2020,
8(
1), 562ŌĆō572.


[23] R. Madandoust, E. Mohseni, SY. Mousavi and M. Namnevis,
Constr Build Mater,
2015,
86, 44ŌĆō50.

[24] E. Mohseni, W. Tang and S. Wang,
Molecules,
2019,
24(
7), 1360ŌĆō1376.

[25] T. Gupta, S. Chaudhary and RK. Sharma,
Constr Build Mater,
2014,
73, 562ŌĆō574.

[26] T. Gupta, S. Siddique, RK. Sharma and S. Chaudhary, Struct Concr, 2020, 1ŌĆō13.
[27] TA. Tawfik, MA. El-Yamani, S. Abd El-Aleem, A. Serag Gabr and GM. Abd El-Hafez,
Asian J Civ Eng,
2019,
20(
1), 135ŌĆō147.


[28] X. Zhang, X. Du, X. Zhao, Z. Zhou and X. Cheng,
J Mater Civ Eng,
2019,
31(
6), 04019073ŌĆō9.

[29] JC. Arenas-Piedrahita, P. Montes-Garc├Ła, JM. Mendoza-Rangel, HZ. L├│pez Calvo, PL. Valdez-Tamez and J. Martinez-Reyes,
Constr Build Mater,
2016,
105, 69ŌĆō81.

[30] MM. Khotbehsara, E. Mohseni, MA. Yazdi, P. Sarker and MM. Ranjbar,
Constr Build Mater,
2015,
94, 758ŌĆō766.

[31] E. Mohseni, BM. Miyandehi, J. Yang and MA. Yazdi,
Constr Build Mater,
2015,
84, 331ŌĆō340.

[32] R. Garg, M. Bansal and Y. Aggarwal,
Int J Electrochem Sci,
2016,
11(
5), 3697ŌĆō3713.


[33] R. Puente-Ornelas, LC. Guerrero, GF-S. Miguel, EA. Rodr├Łguez, A. Trujillo-├ülvarez, HE. Rivas-Lozano and HM. Delgadillo-Guerra,
Int J Electrochem Sci,
2016,
11, 277ŌĆō290.

[34] S. Haruehansapong, T. Pulngern and S. Chucheepsakul,
Constr Build Mater,
2014,
50, 471ŌĆō477.