|
 |
- Search
| J. Electrochem. Sci. Technol > Volume 13(4); 2022 > Article |
|
Abstract
Solar energy harvesting is practiced by various nations for the purpose of energy security and environment preservation in order to reduce overdependence on oil. Converting solar energy into electrical energy through Photovoltaic (PV) module can take place either in on-grid or off-grid applications. In recent time Lithium battery is exhibiting its presence in on-grid applications but its role in off-grid application is rarely discussed in the literature. The preliminary capacity and PeukertŌĆÖs study indicated that the battery quality is good and can be subjected for life cycle test. The capacity of the battery was 10.82 Ah at 1 A discharge current and the slope of 1.0117 in the PeukertŌĆÖs study indicated the reaction is very fast and independent on rate of discharge. In this study Lithium Iron Phosphate battery (LFP) after initial characterization was subjected to life cycle test which is specific to solar off-grid application as defined in IEC standard. The battery has delivered just 6 endurance units at room temperature before its capacity reached 75% of rated value. The low life of LFP battery in off-grid application is discussed based on State of Charge (SOC) operating window. The battery was operated both in high and low SOCŌĆÖs in off-grid application and both are detrimental to life of lithium battery. High SOC operation resulted in cell-to-cell variation and low SOC operation resulted in lithium plating on negative electrode. It is suggested that to make it more suitable for off-grid applications the battery by default has to be overdesigned by nearly 40% of its rated capacity.
Across the globe a paradigm shift is happening with respect to energy generation and consumption. The use of renewable energy has been identified as an unavoidable mitigation action to tackle global warming [1]. Energy sustainability, green and clean energy to mitigate climate change are the buzzword among policy makers. The energy mix is shifting from non-renewable (oil, coal and coke) to renewable such as solar and wind. Solar and wind are the renewable energy sources available in plenty across the globe in various proportions. After converting this energy in to electrical energy using PV panel and when the output is fed into nationŌĆÖs main electric grid, overload and instability may result and it is undesirable. Secondary rechargeable batteries play a critical role and it acts like a cushion. It stores energy from renewable sources such as wind and solar and release/absorb energy to/from electrical main grid depending on the main grid requirements [2].
In recent past, lithium battery is also trying to play its role in this system. Lithium system due to its higher energy and power density and long cycle life at cell level is drawing more attention [3]. The most widely employed material for the cathodes among lithium batteries is lithium cobalt oxide (LCO) [4,5] or LiCoO2, however, it has disadvantages such as accelerated aging at high currents [6] and low thermal stability when operating in temperatures between 100 and 150┬░C [7]. When Co is partially substituted with Ni, it reduces the cationic disorder [8] but thermally disturbed. The lithium nickel oxide cell (LNO) or LiNiO2 has the same crystal structure but it is more thermally unstable [6].
The widely used lithium nickel cobalt aluminum oxide, also causes fast capacity decrease rates [6] at elevated temperatures of operation. In the lithium manganese oxide (LMO) cells, the change in structure during the lithium-ion extraction has a negative impact on cycle life, and Mn tends to dissolve into the electrolyte when the battery is not cycled [9]. The lithium-nickel-manganese-cobalt-oxide (NMC) cells also show appropriate cycle stability at 50┬░C, although one of the significant issues of NMC is the unique voltage profile, which does not present the expected flat region found in cells such as LiFePO4 (LFP). Further NMC cells are highly priced and the instability of the voltage makes it an unfavorable candidate. Lithium iron phosphate batteries are considered as one of the more mature and stable Li-ion technologies [4], showing an excellent thermal stability and cycle life, good power capability, and it is regarded as the safest lithium-ion type concerning thermal runaway risk.
The grid applications are broadly classified in to (a) on-grid and (b) off-grid. Thus, in on-grid applications the battery is acting as an instantaneous power source/sink for most of the time. Occasionally it acts as an energy source to stabilize the electric gridŌĆÖs performance when randomly major amount of electrical energy from renewable energy sources are fed into or extracted from it. In solar off-grid PV application, the integrated system (solar PV panel, inverter, controller and battery) should supply energy even for 3 to 15 days without any solar irradiation. Though there are plenty of studies available in the literature on lead-acid battery hardly limited study was available in the literature regarding application of lithium-ion battery in solar PV-off grid applications [10]. LFP cells from different manufacturers were taken and tested for off grid solar PV applications and the capacity of the cells was compared and found that nominal capacity and uniformity of the cells differ significantly across and even within manufacturers. In addition, the lithium-based batteries were subjected to life cycle at medium SOC (40 to 70% SOC) in most of the times and very few times at low SOC (10 to 40%) and at high SOC (75 to 100%). IEC-61427 demanded a battery to be subjected to 10 to 40% SOC for 33% of the times and 75 to 100% SOC for 66% of times to simulate real life conditions.
In solar off-grid application, the cell/battery to be tested between 75 to 100% SOC which represents summer time and 10 to 40% SOC which represents winter time. Hardly any published literature is available in solar-off grid test regime for lithium battery and this has propelled the present authors to study performance of LFP battery in the above regime. In the present work after initial characterization LFP battery by the preliminary capacity and PeukertŌĆÖs studies, it was subjected to life cycle test which is specific to solar off-grid application as defined in IEC standard.
12.8 V/10 Ah (rated) LFP/C (Korean made) battery with 4S/2P (meaning four cells in series and two in parallel) configuration was taken for the study. The battery details are: Dimensions (l├Śm├Śh): 10.3├Ś1.4├Ś3.0ŌĆ│ (262├Ś36├Ś77 mm), Weight: 1.29 kg, Cell Type: cylindrical, Energy: 128 Wh, Resistance: Ōēż90 m╬® @ 50% SOC, Recommended Charge Current: 0.5ŌĆō5 A, Storage Temperature: ŌłÆ5 to 35┬║C. The battery was having inbuilt Battery Management System (BMS). The battery was charged at 0.2 C rate (2.0 A) till the voltage reaches 14.6 V (3.65 V/cell) and maintained at this voltage till the current reaches 0.05 A. After 30 min rest, capacity of the battery was measured by discharging the battery at 0.1 C rate (1 A) till the voltage becomes 12 V (3 V/cell). Then the battery was subjected to PeukertŌĆÖs study. The PeukertŌĆÖs protocol is as follows. Step 1: Fully charge the battery at 2.0 A (0.2 C rate) till the voltage reaches 14.6 V (3.65 V/cell) and maintain at 14.6 V till the current becomes 0.05 A; Step 2: Keep the battery in rest for 2 hours; Step 3: Discharge the battery at 1 A current till the battery voltage becomes 12 V (3 V/cell); Step 4: Repeat the steps 1 to 3 with discharge currents 2, 4, 6, 8 and 10 A in Step 2. After PeukertŌĆÖs study, the battery was subjected to life cycle test as per IEC-61427 (2013) - Part-1 for off-grid solar photovoltaic application.
The life cycle test protocol consisted of two phases namely Phase-A and Phase-B. Phase-A was cycling at low SOC between 10 to 40% SOC. Step 1: Fully charge the battery at 2 A current (0.2 C rate) till the voltage becomes 14.6 V and maintain at 14.6 V till the current becomes 0.05 A. Step 2: Discharge the current at 1 A for 9 hours so that SOC of battery is 10% of rated capacity. Step 3: Charge the battery at 1.03 A current for 3 hours and discharge at 1 A current for 3 hours. Step 4: Continue step 3 for 49 cycles. After Phase-A The battery was subjected to Phase-B cycling. Phase-B was cycling at high SOC between 70 to 100% SOC. Step 5: Discharge the battery at 1.25 A for 2 hours. Step 6: Charge the battery at 1 A current till the voltage become 14.6 V. Step 7: Repeat step 5 and 6 for 99 times. After Phase-B, the battery was subjected to capacity test. Step 8: Discharge the battery at 1 A current till the voltage becomes 12 V. With this one endurance unit was over. Step 9: Repeat this endurance unit (step 1 and 8) till the capacity of the battery becomes 70% of initial capacity value instead of 80% of Initial capacity as stated in IEC.
After life cycle test, the battery was dismantled and individual cellŌĆÖs capacity and IR were measured. Cell capacity was measured by charging the cell at 0.5 A current till the voltage becomes 3.65 V. After 2 h rest, battery was discharged at 0.5 A till the voltage become 3.0 V and once again charged at 0.5 A till the voltage becomes 3.65 V.
IEC 61427-1 (2013) - Secondary cells and batteries for renewable energy storage ŌĆō Photovoltaic off-grid applications [11] define life cycle test protocol. Here the batteries are tested both at low SOC (between 10 to 40%) for 49 cycles and as well as at high SOC (between 75 to 100%) for 99 cycles. The low and high SOC life cycle test constitutes one endurance unit. After every one endurance unit the health of the battery is monitored by measuring the capacity. This procedure is continued till the measured capacity after endurance unit becomes 80% of rated capacity. The test is conducted at 40 C to fasten the process and one unit at 40 C corresponds to one-year life in the field. The procedure is technology agnostics and for lead-acid the reference capacity is C-10 and for Ni/MH and Lithium based reference capacity is C-1.
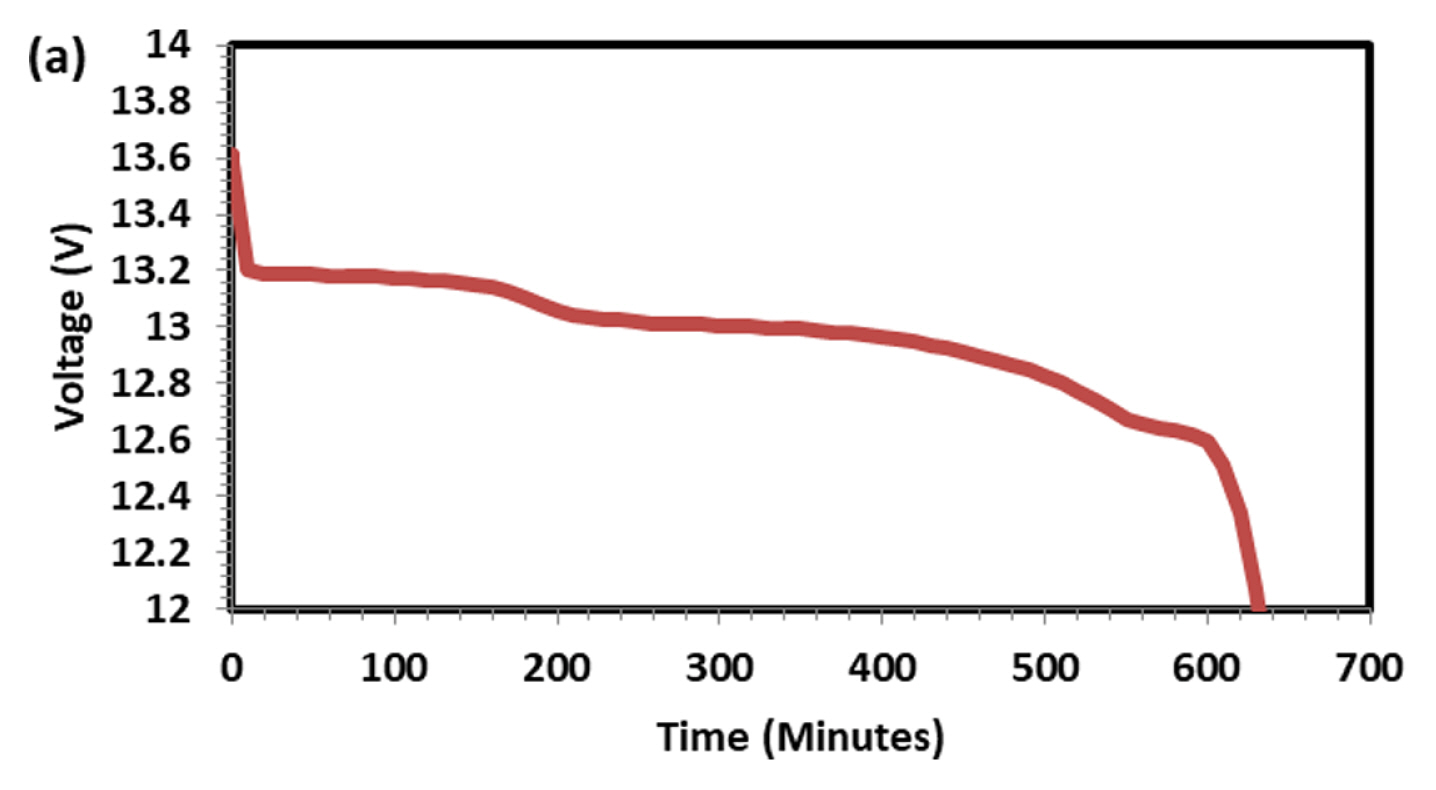
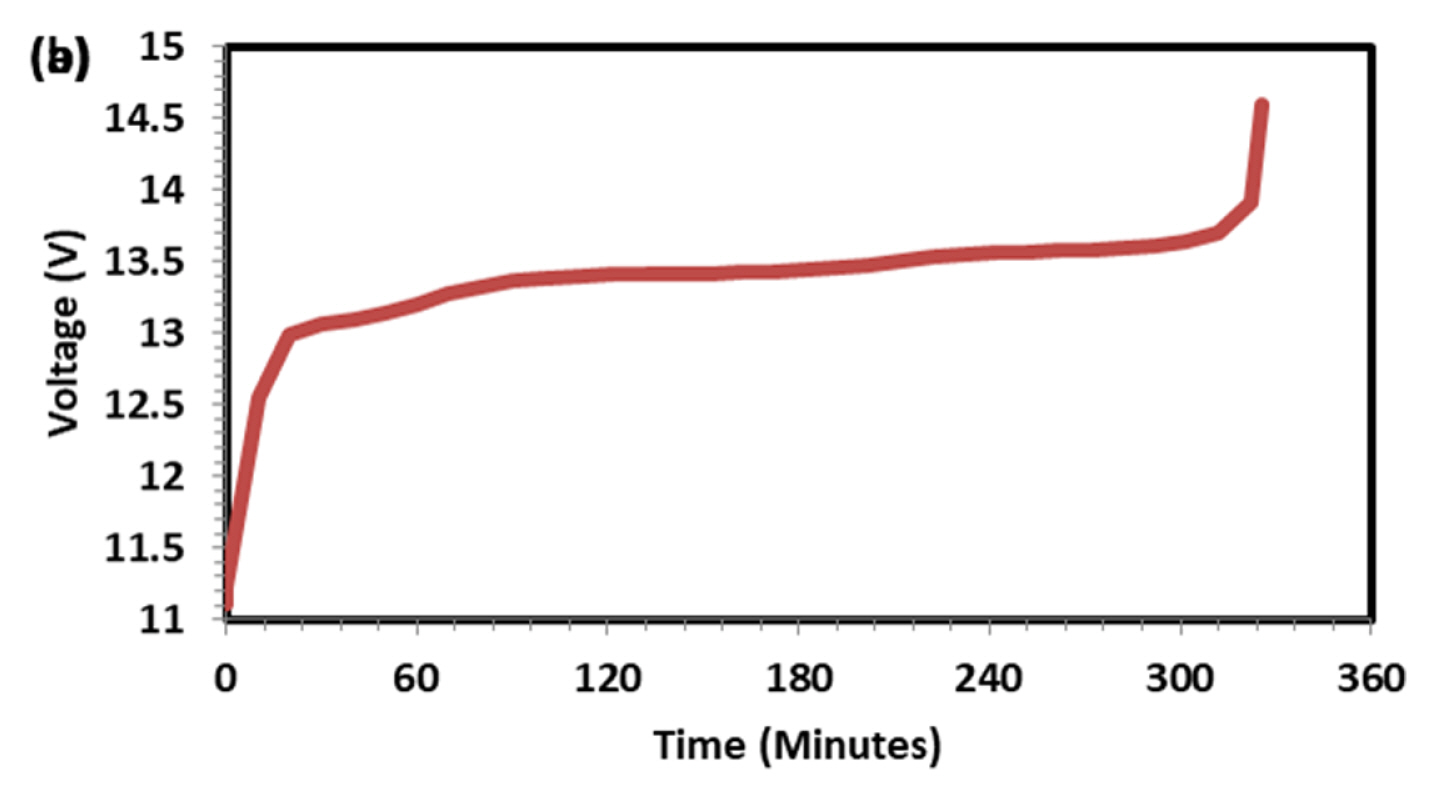
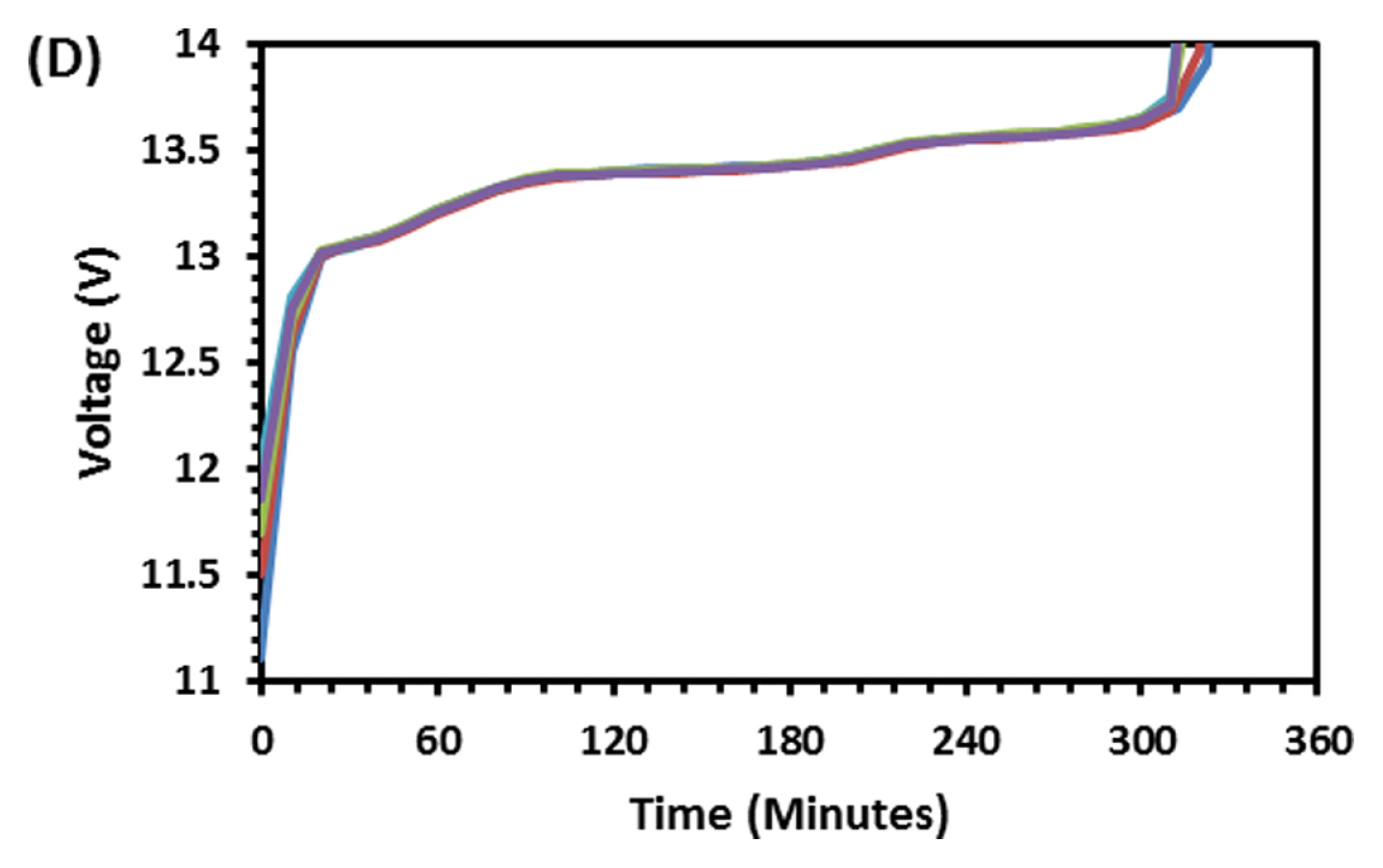
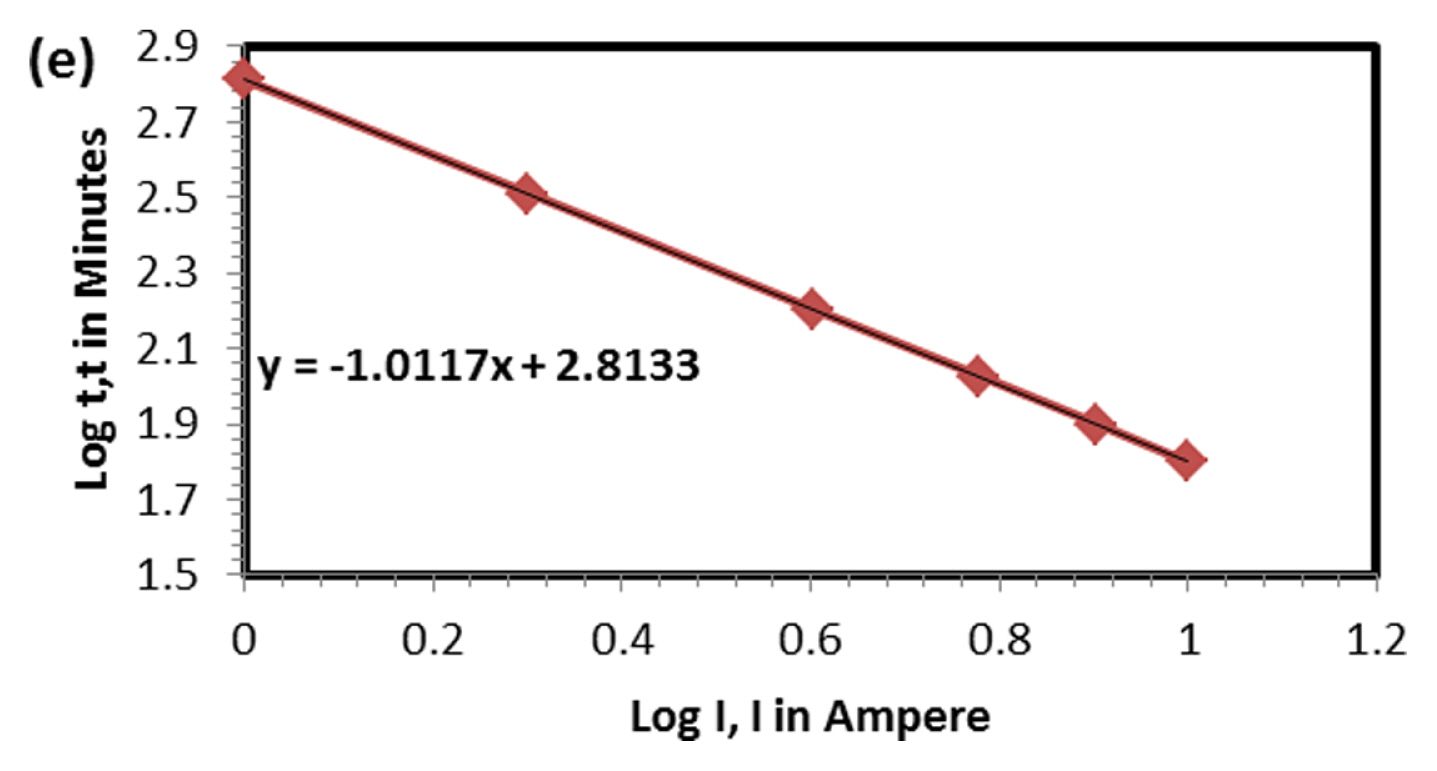
Figs. 1 and 2 Shows discharge (1 A current) and subsequent charging capacity (2 A current) of LFP battery. As observed and discussed in our previous paper [12] five distinct regions are seen in voltage-time graph both in charge and discharge operation. The capacity of the battery was 10.82 Ah at 1 A discharge current which is higher than the rated capacity 10 Ah. For PeukertŌĆÖs study the battery was discharged at various currents, namely 1, 2, 4, 6, 8 and 10 A to find out the impact of rate of discharge on capacity output. As expected, the rate of discharge hardly made an impact on discharge capacity. Though rate of discharge increased by 10 times the capacity drop is just 2.77% indicating the discharge kinetics is very fast in LFP system. Voltage-time plot at various discharges current is shown in Fig. 3. As the discharge current increases, the number of distinct regions is getting reduced from 5 to 2. Fig. 4 explains charging behavior of battery after various discharge currents. Since charging current is the same, the previous discharge current does not have any specific effect on charging behavior and capacity. Fig. 5 shows typical PeukertŌĆÖs plot. The slope of 1.0117 indicates the reaction is very fast and independent on rate of discharge. The preliminary capacity and PeukertŌĆÖs study indicate that the battery quality is good and can be subjected for life cycle test.
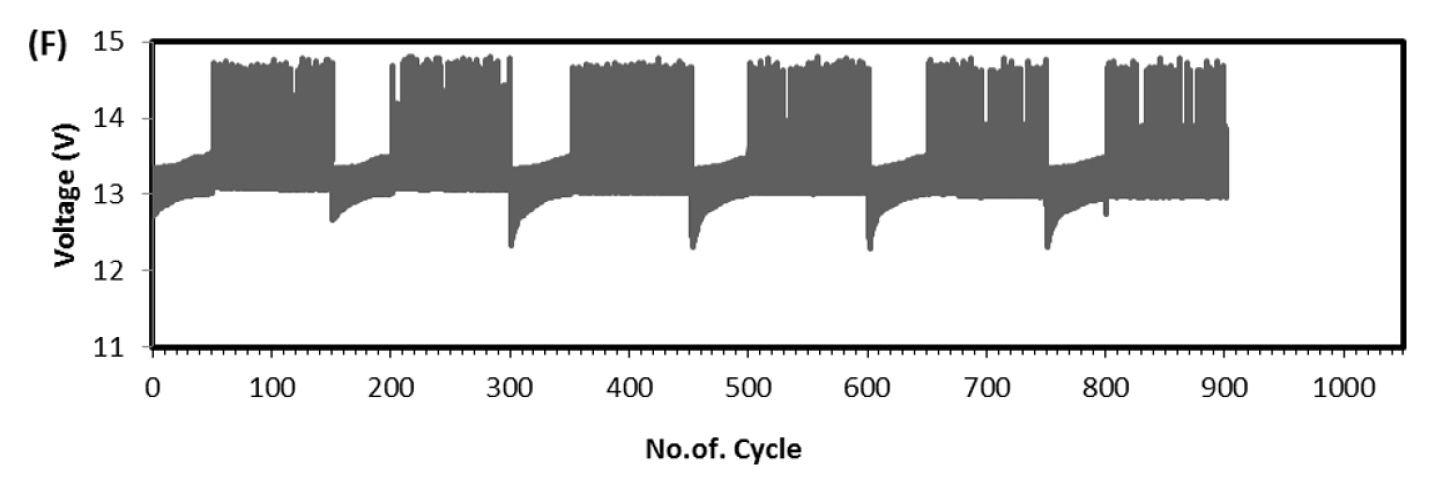
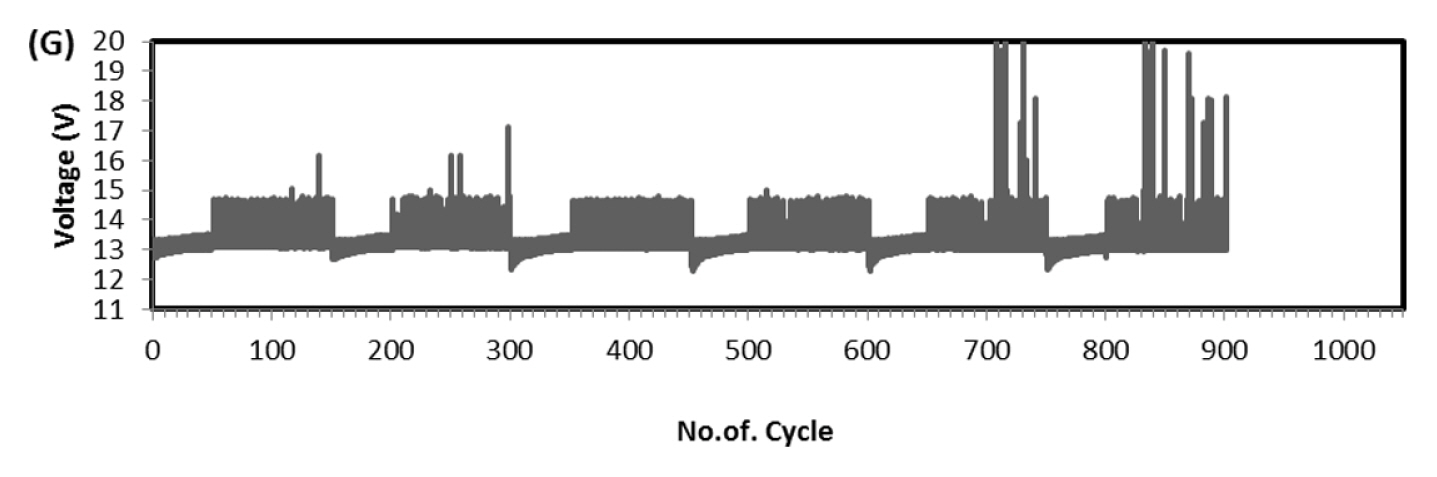
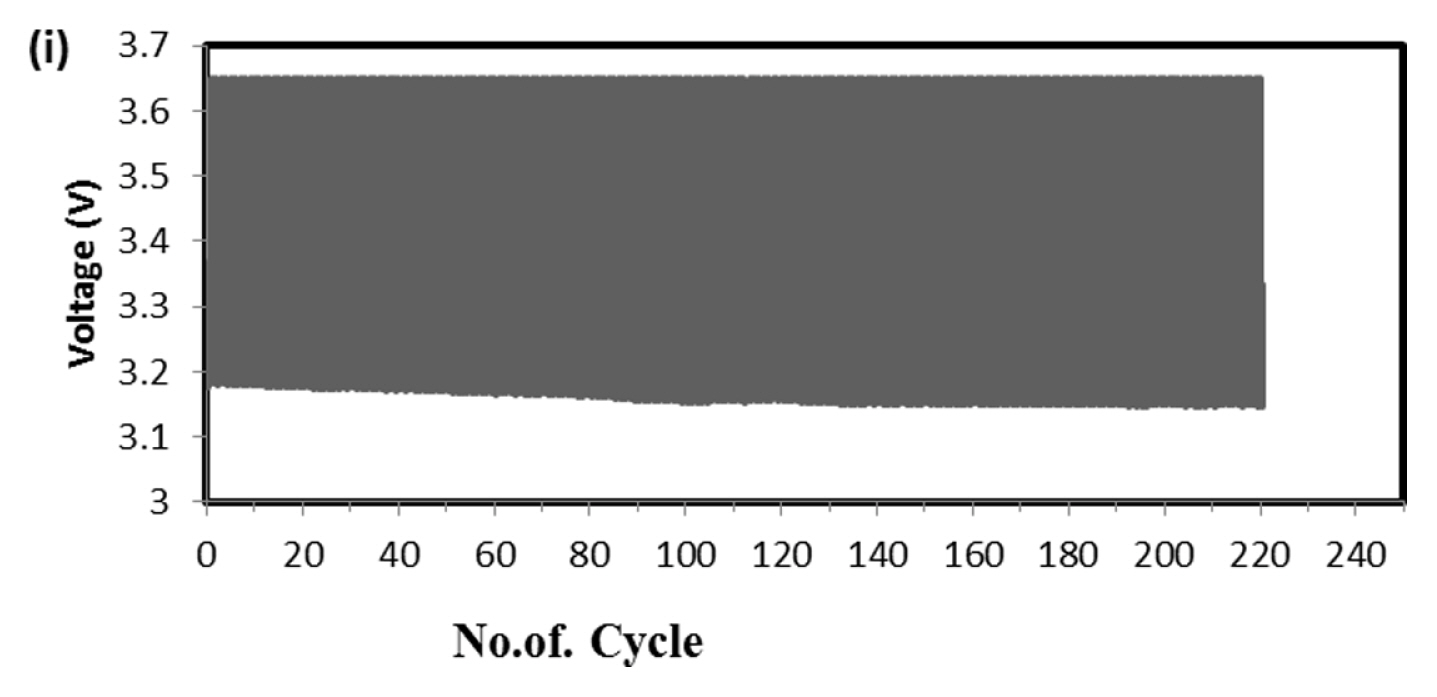
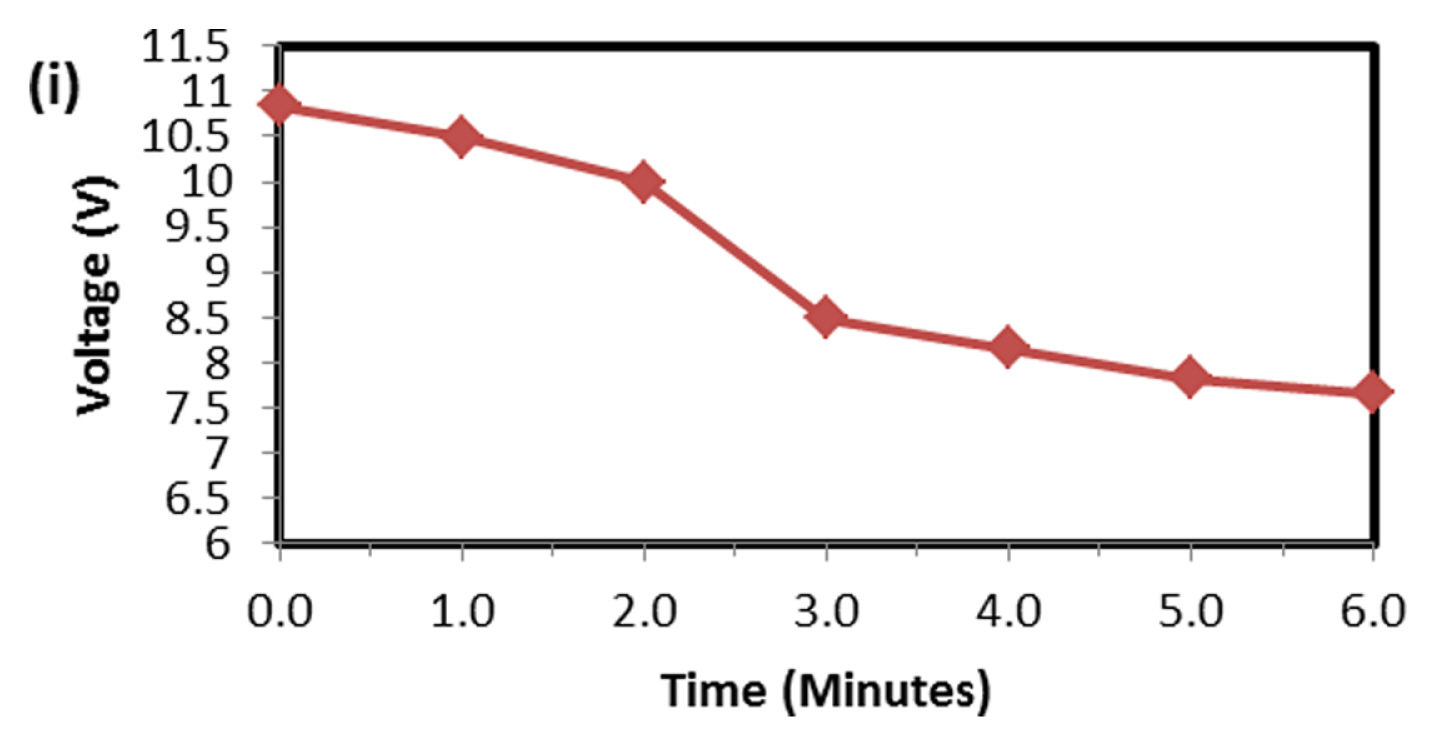
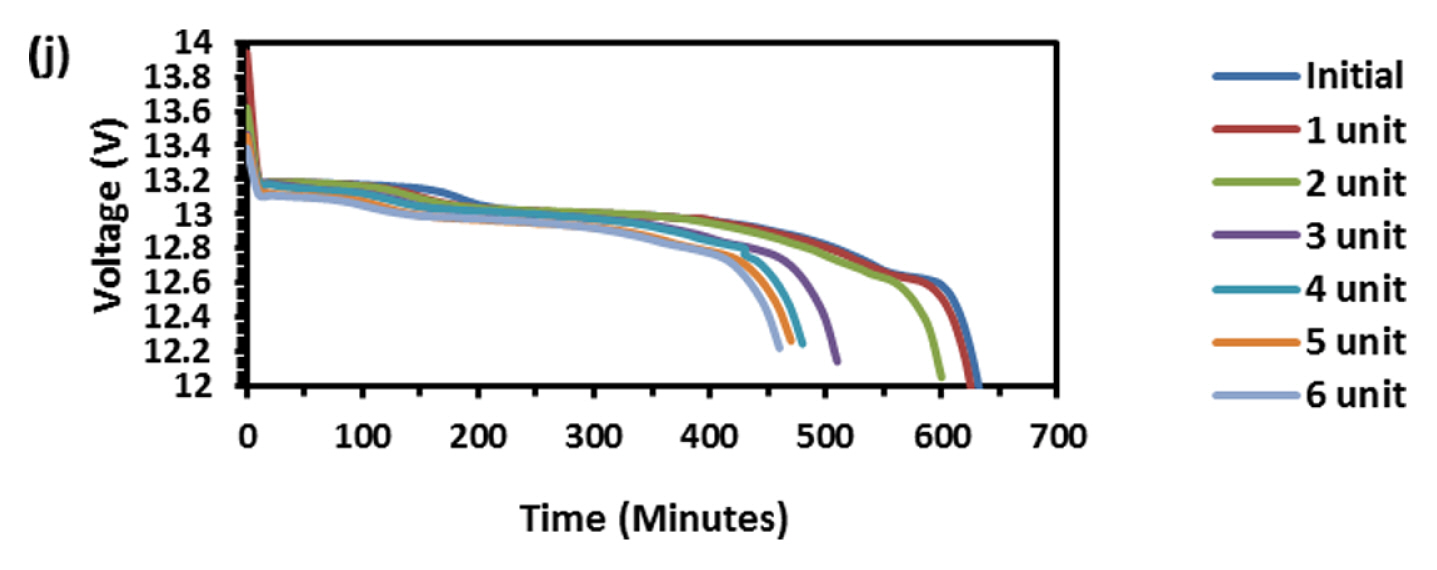
This test is very robust and accepted by international community for solar-off grid application. As per the standard one endurance unit corresponds to 1 year life in the field and comprises of Phase-A, 49 cycles at low SOC (10 to 40%), Phase-B, 99 cycles at high SOC (75 to 100%) and one capacity health check. The test protocol was designed to take care of seasonal variations such as, for more than 2 weeks the battery might not get charged due to absence of solar radiations. Cumulative Ah output in Phase-A is 14.7 times and Phase-B is 24.75 times of rated capacity. Hence total Ah output in one endurance unit is 39.45 times of rated capacity (C-10 in case of lead-acid and C-1 in case of Lithium battery). Since the test is conducted at 40┬░C, the ambient temperature Ah turnover is double the value of 40 C and in this case, it is 78.9. At maximum, solar battery is discharged for 20% of rated capacity in a day. Hence one endurance unit corresponds to 395 days, i.e., little more than one year in the field. In the present study the test was conducted at room temperature (25 to 28┬░C), hence two endurance units correspond to one year life in the field. Fig. 6 is the real time voltage vs. cycle evolution plot. In Phase-A, as the life cycle progresses the end discharge voltage is becoming more positive because the charge input is 1.03% of previous discharge output which increases SOC by 0.03%. In Phase-B, during change over stage between 14.6 V charge and 1.25 A CC discharge, voltage spike is taking place due to cell balancing act. The changeover is taking place in less than 50 ms interval. Since two cells are in parallel in each module (4S/2P configuration) the balancing current is equal to difference in voltage between the cells and its internal resistance. This huge inflow of current results in occasional voltage spike and as the cycle progresses more spikes should occur due to cell divergence. Fig. 7 explains this phenomenon. In four endurance units 7 spikes has occurred out of which 3 are up to 15 V, 3 are up to 16 V and 1 is up to 17 V. In 5th and 6th endurance units, more than 30 spikes occurred in the voltage range 17 to 20 V. This is an indication of cell divergence due to inherent variations in raw material, design and process [13]. Since voltage excursion is up to 20 V in the battery and 5 V at cell level, the impact will be huge in oxide-based cathode cells. Solar off-grid application needs battery to perform its duty close to 100 and 0% SOC, the inherent variation and voltage spike due to cell balancing is a challenge in real life applications. To confirm that the cell voltage excursion is only due to cell balancing, after the life cycle the battery was dismantled and two individual cells were subjected to discharge/charge study. The voltage-time behavior is depicted in Fig. 8 and fluctuation is not happening at all during changeover from charge to discharge. This clearly shows that the battery after reaching 14.6 V, during change over time (less than 50 ms) from charge to discharge, cell balancing is happening and during this gap the current which is flowing between the cells results in polarizing the cells in an uncontrolled way. Fig. 9 explains health check capacity data after each endurance unit. The plot shows nearly 4% capacity decay per unit for first two units and sudden drop in capacity by 14% and further a drop of capacity by 3% per unit. After sixth endurance unit battery lost 30% of its original capacity indicating that LFP battery can reliably work in solar off-grid application for just 2 to 3 years. This is startling and reasons to be known for this poor performance.
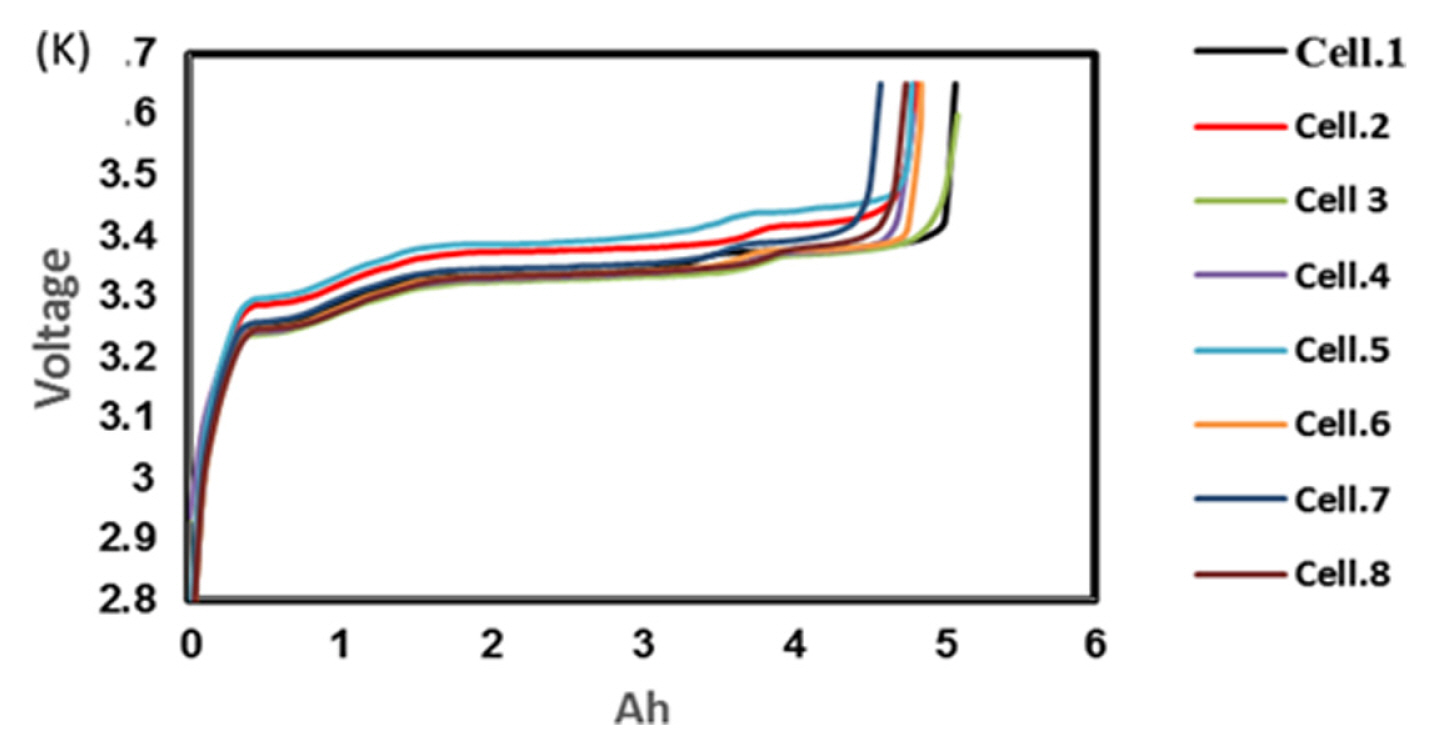
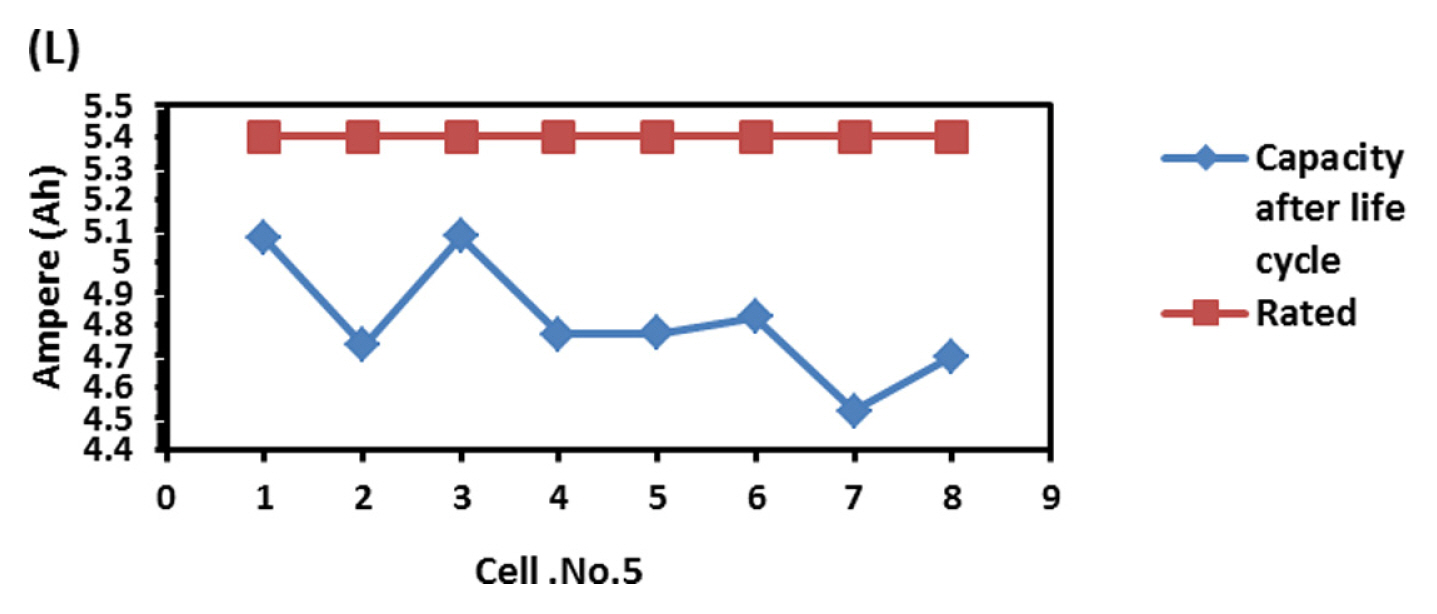
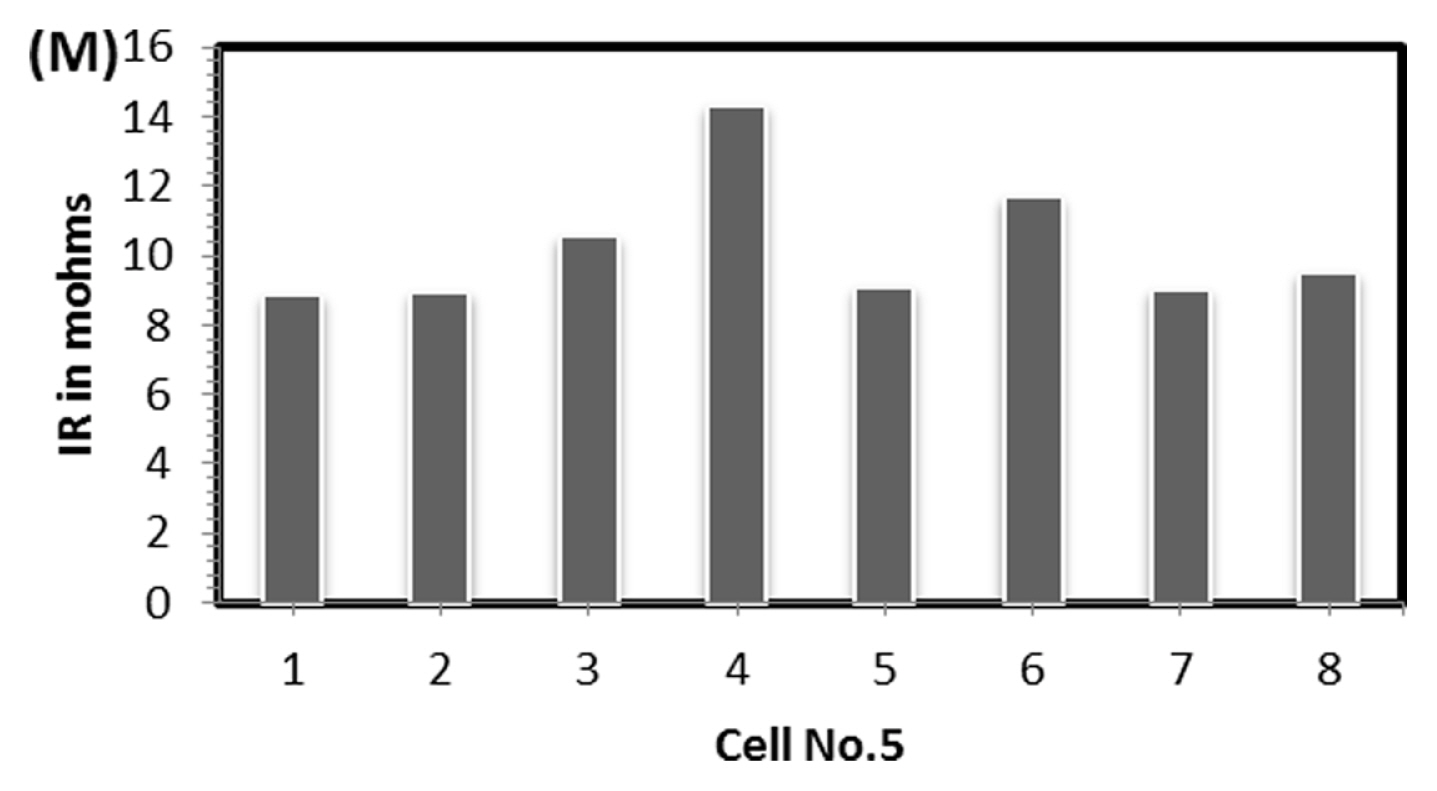
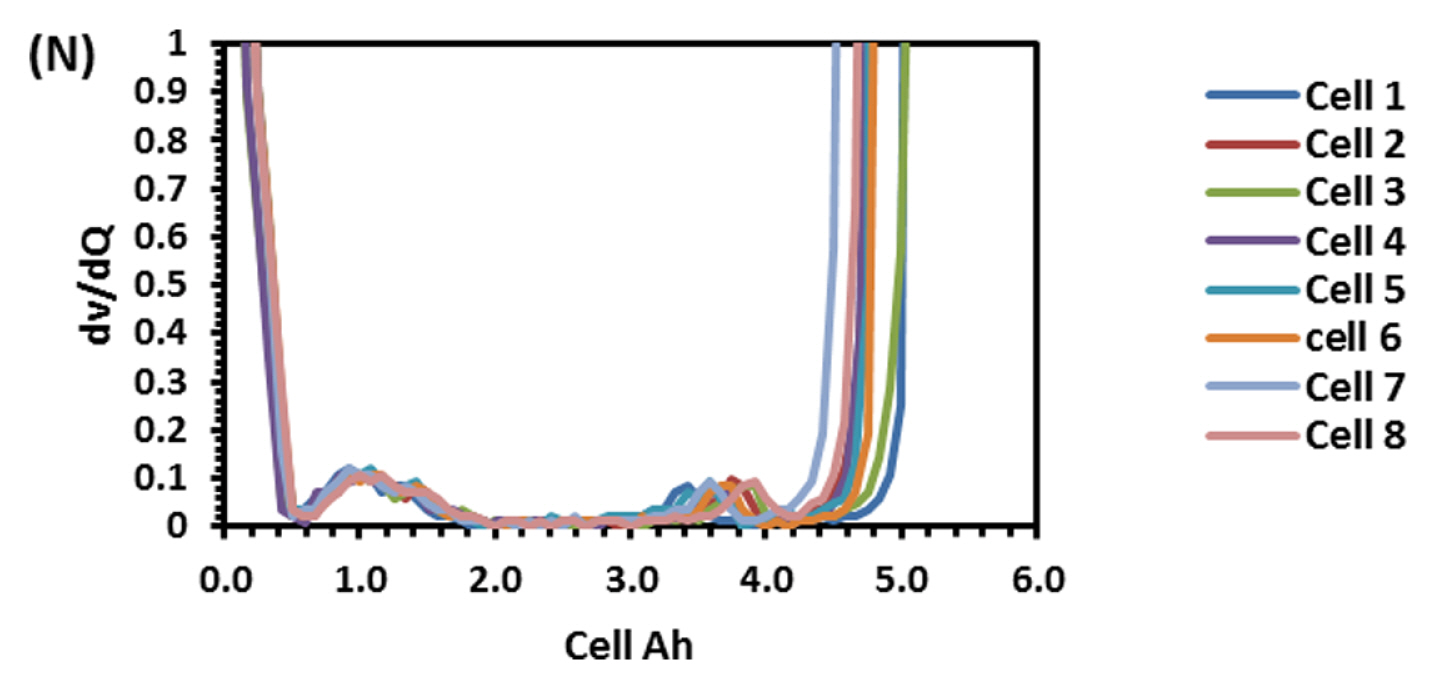
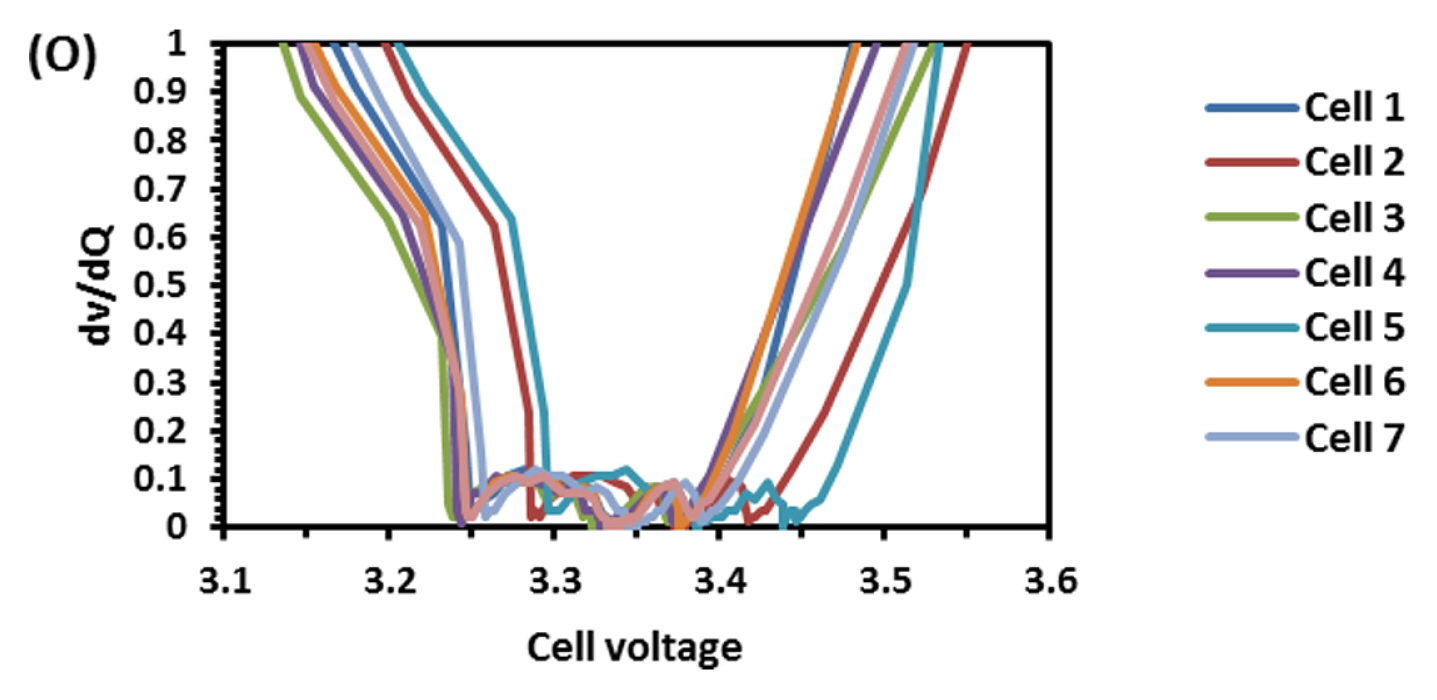
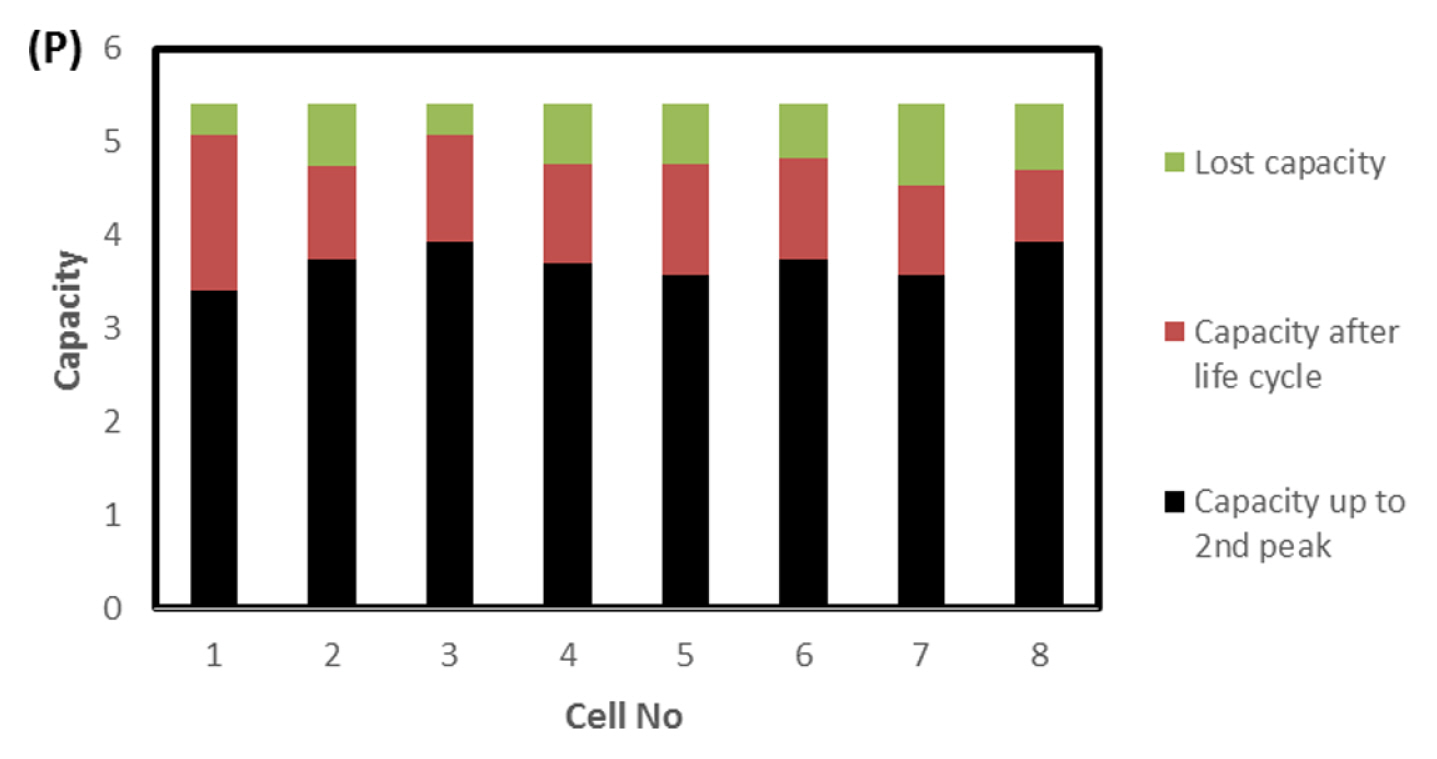
To find out the reason, the discharge capacity curve as in Fig. 10 was plotted for every health check study after each endurance unit. It is clear that up to 2 cycles, the end voltage drop started at 12.6 V and from 3 to 6 cycles, the voltage drop started at 12.8 V indicating the phenomena happening between 12.6 to 12.8 V is missing in the later endurance units. To probe further, the battery was discharged and cells were dismantled and individually charged at 0.5 A current till the voltage becomes 3.65 V and Fig. 11 shows charging capacity of individual cells. All the cells are diverging after 3.4 Ah input, and cell No. 2 and 5 are polarized at 60 mV higher than the other cells. The individual capacity of the cells is shown in Fig. 12. Assuming that the original capacity of the cells is 5.4 Ah, out of 8 cells, 5 cells lost 25 to 30%, one cell 20% and 2 cells around 10% of rated capacity. IR (Internal resistance) of the cells measured using Hioki instrument is shown in Fig. 13. Variation in the IR is in the range of 0 to 46%. Capacity and IR variation between individual cells clearly indicating that the cells are diverging during life cycle test. To unravel more, the data in Fig. 11 was rearranged in the form of dV/dQ vs. Ah and dV/dQ vs. voltage plots as in Figs. 14 and 15 [14,15]. They demonstrated that incremental capacity study (dQ/dV vs. voltage) can be effective for identifying the root causes for capacity fading such as loss of active material, change in cell chemistry, undercharging and under discharging phenomena. dV/dQ vs. Ah plot was used in [16] for studying the degradation mechanism. In the present study dV/dQ is used in y-axis because the change in voltage during charging and discharging for LFP system is zero or very minimum due to involvement of two-phase systems. In addition, the 10 minutesŌĆÖ data collection frequency made dV very small and as a result dV at the denominator is distorting the data and hence dV/dQ is used. Fig. 14 shows that all the cells behave more or less same till the Ah input during charging is 3.4 Ah. Divergence is occurring after 3.4 Ah and the second peak position also appeared at different Ah for the cells and the capacity range varies from 4.57 to 5.07 Ah. Appearance of second peak at different Ah indicates that the amount of lithium intercalated in graphite structure is different for the cells under study. The original rated capacity for the cell is 5.4 Ah. Hence the individual cells reduced capacity by 6 to 16% of its rated value. Fig. 15 reveals that above 3.4 V the slope of dV/dQ vs. voltage plot is varying for all the cells. This charging voltage regime corresponds to lithium plating reaction on carbon electrode. Variation in the slope indicates indeed lithium is plating on carbon edge at different rate [17,18]. For cell No. 2 and 5, the shift in the dV/dQ vs. voltage plot by nearly 60 mV indicates higher internal resistance which reflects in IR vs. cell plot in Fig. 13. The variations in (a) cell to cell capacity, (b) IR, (c) appearance of second peak in dV/dQ vs. Ah plot and (d) slope in dV/dQ vs. V plot after 3.4 V suggests loss of active lithium due to plating on negative electrode and formation of additional SEI layer due to plated lithium.
Solar off-grid application demands battery to be operated mainly between 0 to 30% SOC (peak winter and rainy seasons) and 75 to 100% SOC (peak summer). When batteries are operated in low SOC (0 to 30%), preferential lithium plating is occurring due to higher reactivity of carbon edge atom than graphene layer. Repeated cycling at this range preferentially plates out lithium on negative electrode. Figs. 14,ŌĆō16 show the above-described phenomena. Variations in lost capacity and capacity up to 2nd peak as in Fig. 16 indeed explains the challenges the battery faces when it operates at low SOC regime. At high SOC (75 to 100%) the charge balancing due to inevitable inherent variation leads to voltage excursion even up to 5 V per cell (Fig. 7). This may lead to destruction of positive active material. In case of LFP it is not seen but in vase of NMC it may lead to fire hazard. When similar study was conducted using NMC, the battery was gutted into fire in authorŌĆÖs laboratory. The voltage excursion at high SOC and lithium plating at low SOC results in poor life cycle performance of LFP battery in solar off-grid applications. Hence to make lithium battery successful in solar off-grid applications, it has to be operated between 30 to 90% SOC window and hence the battery has to be over designed at least by 40%.
Secondary batteries play a pivotal role in solar off-grid application. IEC-61427-2013-part-1, define criteria for a battery to qualify in solar off-grid application. The test demands battery to be operated at low SOC (10 to 40%) and at high SOC (75 to 100% SOC). Repeated cycling of lithium battery at low SOC results in preferential lithium plating on graphite surface and reduces intercalation efficiency and hence capacity. The reduction in capacity is uneven between the cells. Operating lithium battery at high SOC results in voltage excursion of cell even up to 5 V due to cell balancing act during change over stage. High voltage surge due to cell balancing at higher SOC and lithium plating at lower SOC is inevitable in lithium battery. Hence the life cycle performance is not as expected. To alleviate this battery has to be operated between 30 to 90% SOC in other words battery has to be overdesigned by 40%. Due to overvoltage excursion LFP is more suitable than NMC in solar off-grid application considering safety into account.
References
[1] V. Vega-Garita, A. Hanif, N. Narayan, L. Ramirez-Elizondo and P. Bauer, J. Power Sources, 2019, 438, 227011.

[2] IRENA, Electricity storage and renewables: Costs and markets to 2030. International Renewable Energy Agency, Abu Dhabi, 2017.978-92-9260-038-9.
[3] T. Chen, Y. Jin, H. Lv, A. Yang, M. Liu, B. Chen, Y. Xie and Q. Chen, Trans. Tianjin Univ, 2020, 26, 208ŌĆō217.


[5] G. Berckmans, M. Messagie, J. Smekens, N. Omar, L. Vanhaverbeke and J.V. Mierlo, Energies, 2017, 10(9), 1314.

[7] D.H. Doughty and E.P. Roth, Electrochem. Soc. Interface, 2012, 21(2), 21ŌĆō37.
[9] L. Yunjian, L. Xinhai, G. Huajun, W. Zhixing, H. Qiyang, P. Wenjie and Y. Yong, J. Power Sources, 2009, 189(1), 721ŌĆō725.

[11] International Standard IEC 61427-1, Secondary cells and batteries for renewable energy storageŌĆōGeneral requirements and methods of testŌĆōPart 1: Photovoltaic off-grid application. IEC, Geneva, Switzerland, 2013.978-2-83220-763-5.
[12] S. Lakshmana, S. Annadurai, V. Sekar, R. Subban and N. Sugumaran, J. Electrochem. Soc, 2021, 168, 010539.


[14] M. Dubarry, V. Svoboda, R. Hwu and B.Y. Liaw, Electrochem. Solid-State Lett, 2006, 9(10), A454.

[15] V.A. Sethuraman, L.J. Hardwick, V. Srinivasan and R. Kostecki, J. Power Sources, 2010, 195(11), 3655ŌĆō3660.

[16] A.J. Crawford, Q. Huang, M.C.W. Kintner-Meyer, J. Zhang, D.M. Reed, V.L. Sprenkle, V.V. Viswanathan and D. Choi, J. Power Sources, 2018, 380, 185ŌĆō193.

- TOOLS