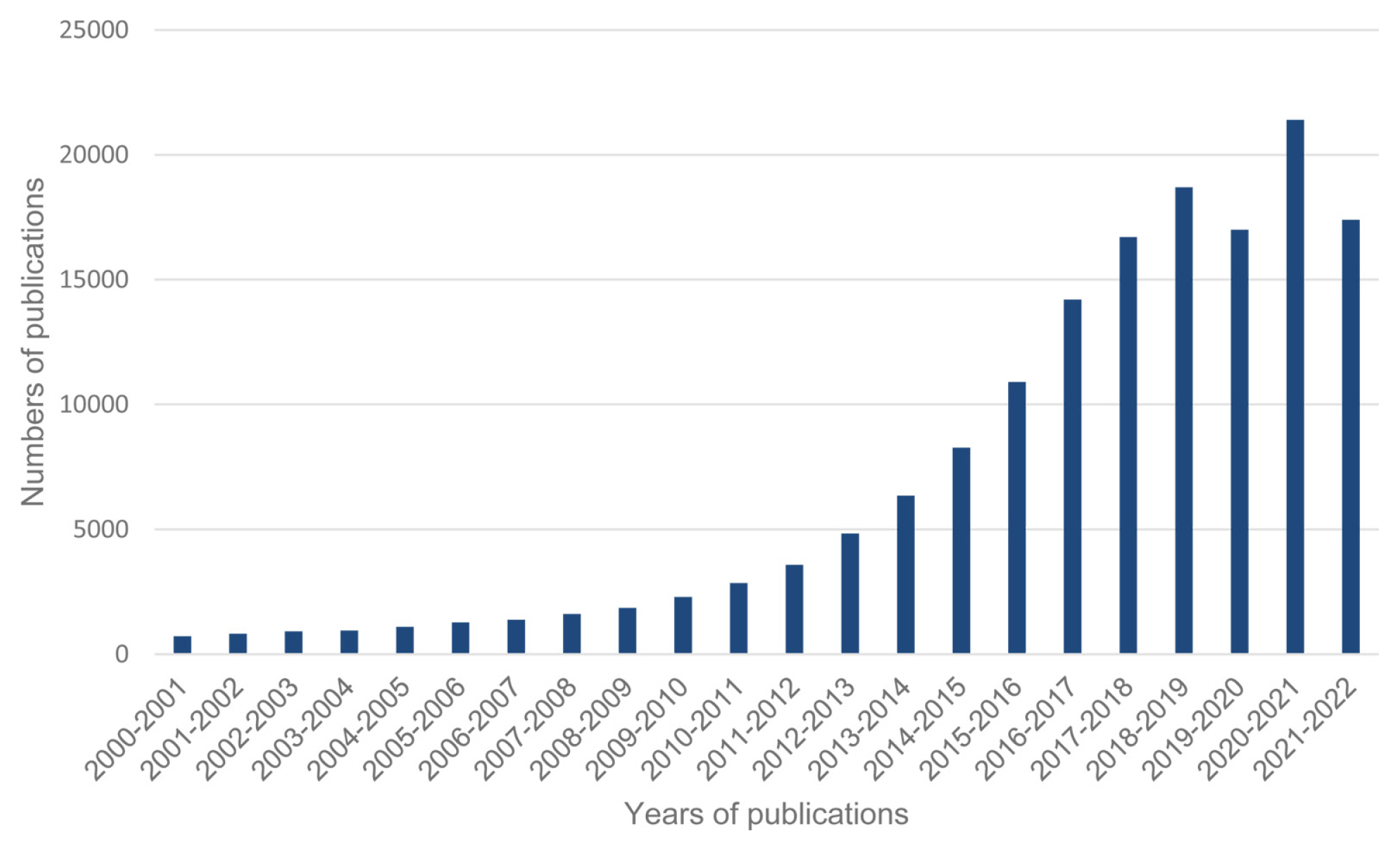
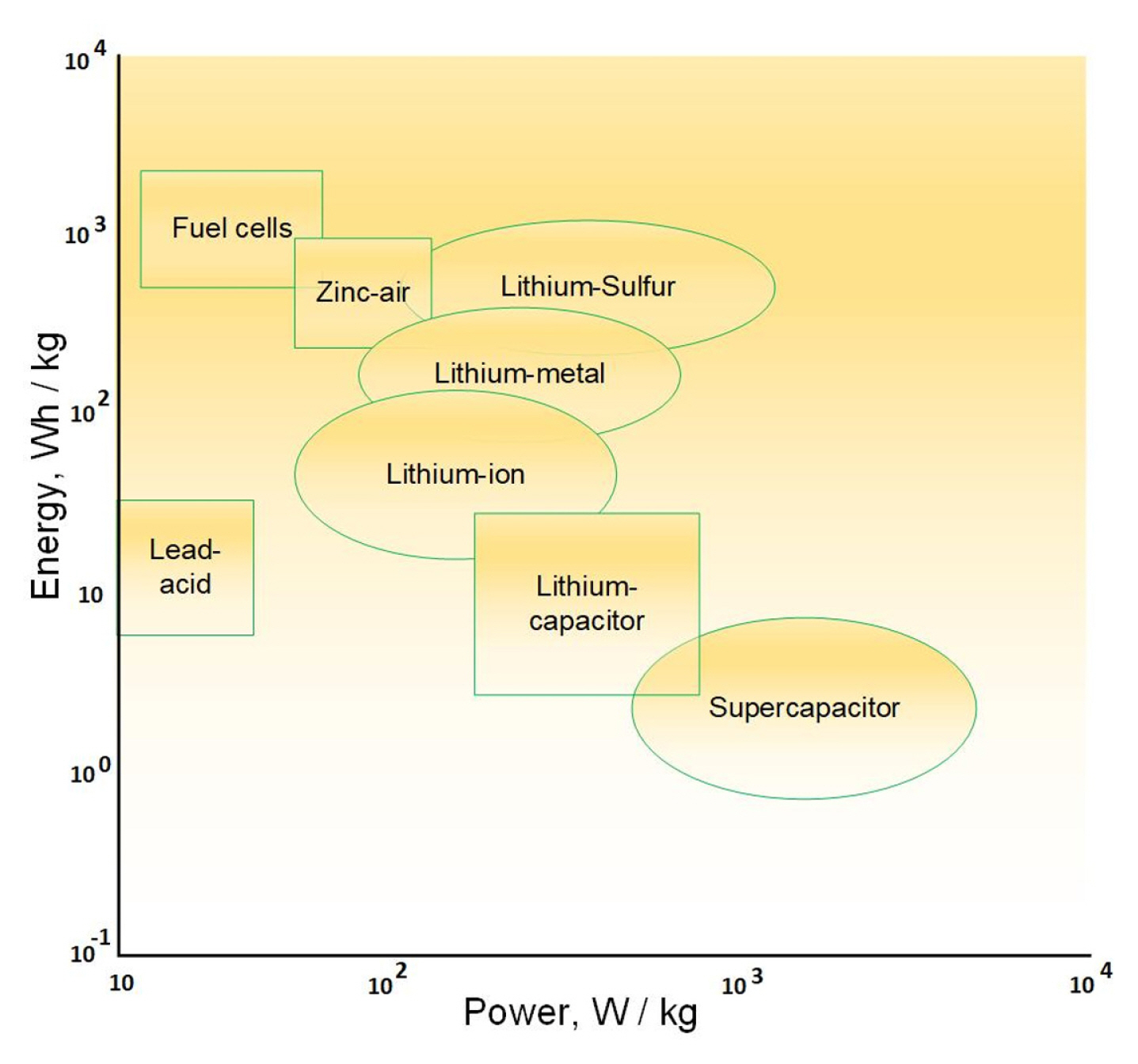
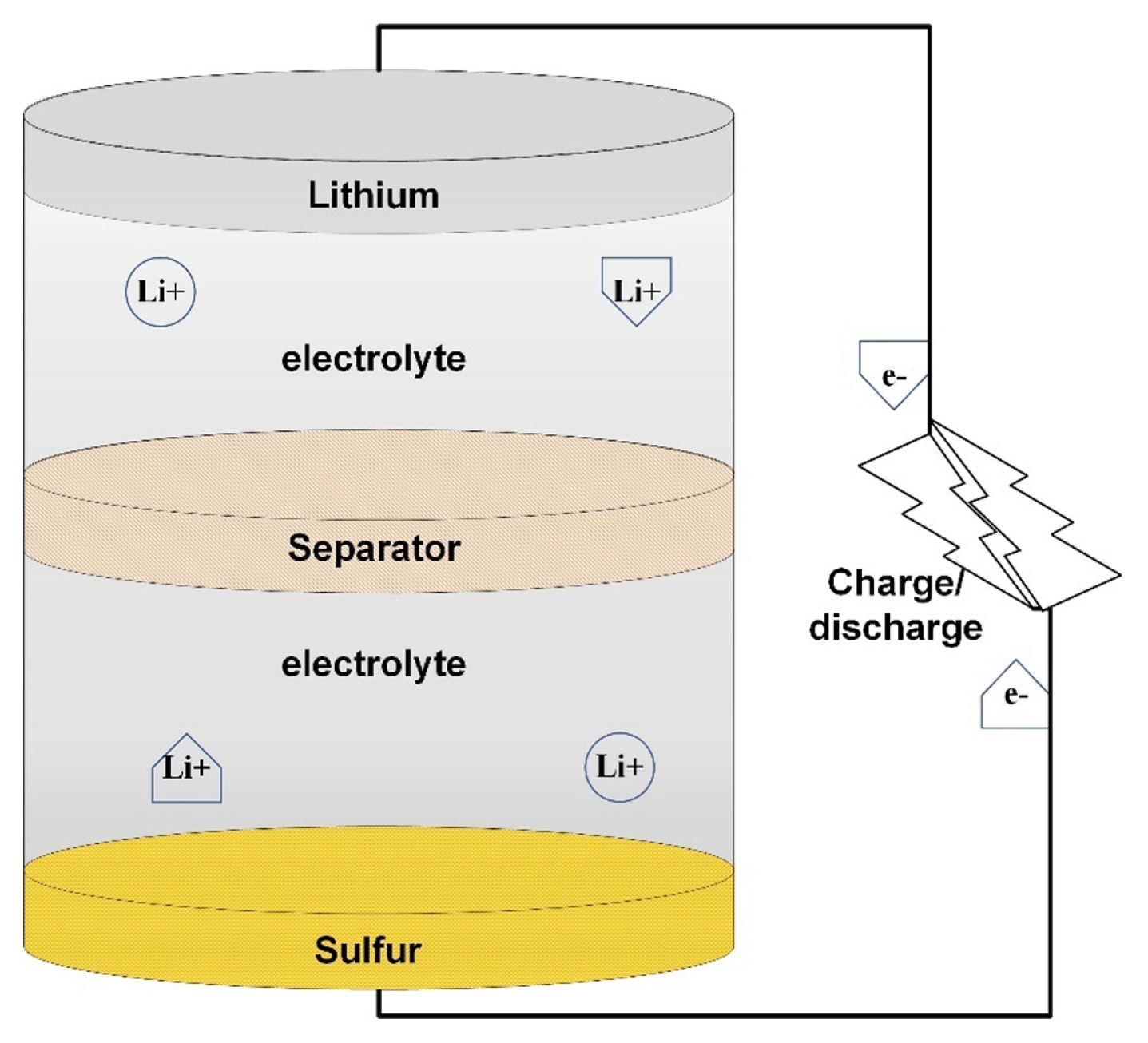
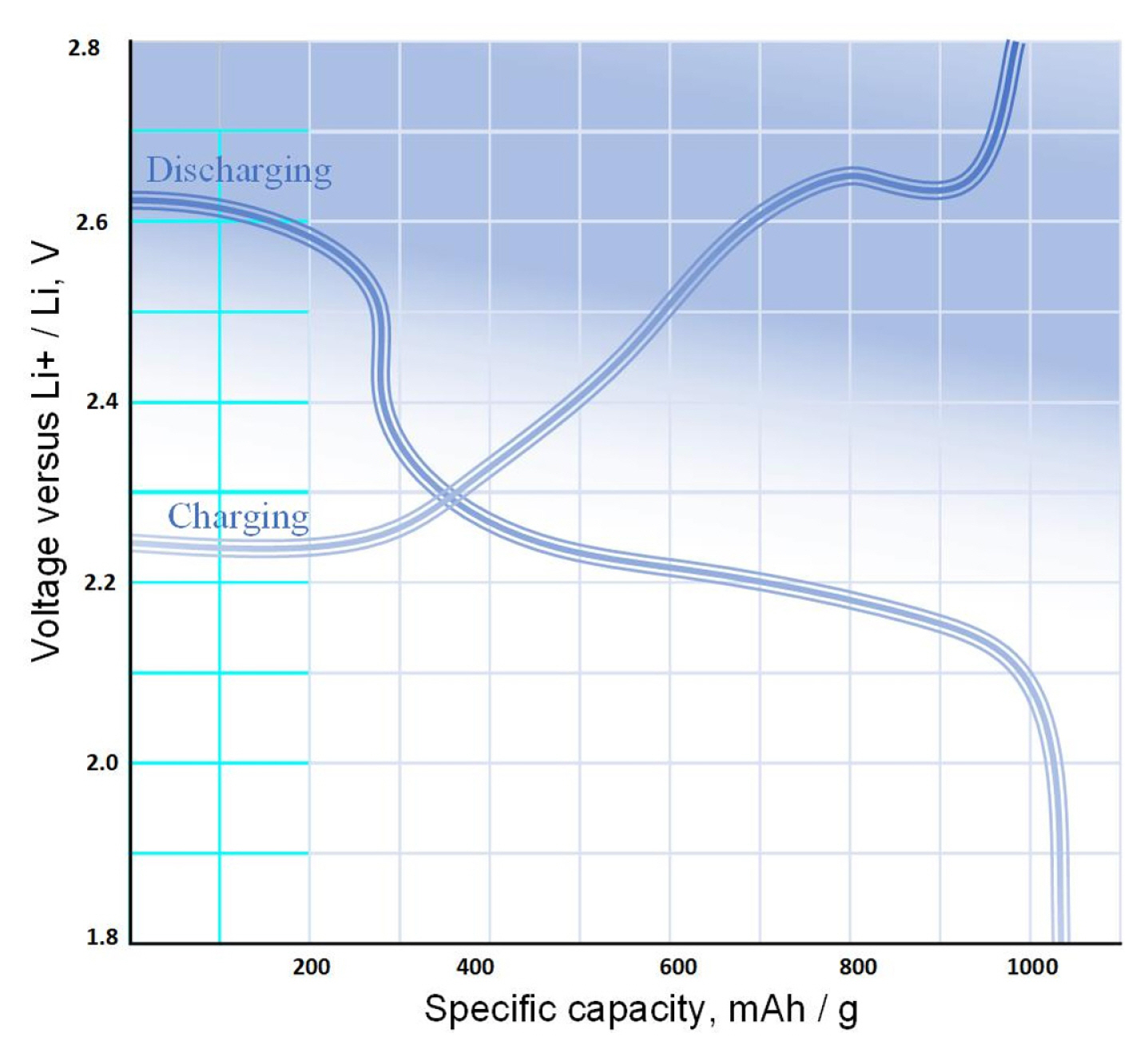
[1] J. H. Kim, Front. Batter. Electrochem., 2022, 1, 1066276.
[2] A. Manthiram, Y. Fu and Y. Su,
Acc. Chem. Res.,
2013,
46(
5), 1125–1134.

[3] V. K. Bharti, S. K. Cherian, M. M. Gaikwad, A. D. Pathak and C. S. Sharma, Chemistry and operation of lithium–sulfur batteries.
Lithium-Sulfur Batteries, Elsvier,
2022, 37–55.

[4] S. Gu, C. Sun, D. Xu, Y. Lu and J. Jin,
Electrochem. Energ. Rev.,
2018,
1, 599–624.


[5] W. Ren, W. Ma, S. Zhang and B. Tang,
Energy Storage Mater.,
2019,
23, 707–732.

[6] G. D. Donato, T. Ates, H. Adenusi, A. Varzi, M. A. Navarra and S. Passerini, Batter. Supercaps, 2022, 5(7), e202200097.
[7] C. Fu and J. Guo,
Curr. Opin. Chem. Eng.,
2016,
13, 53–62.

[8] Y. Mo, L. Liao, D. Li, R. Pan, Y. Deng, Y. Tna and H. Zhou,
Chin. Chem. Lett.,
2023,
34(
1), 107130.

[9] S. J. Park, S. Y. Yang, S. A. Han, Y. J. Choi, T. Kim, M.-S. Park, J. H. Kim and K. J. Kim,
Chem. Eng. J.,
2023,
460, 141620.

[10] G. S. Martynková, G. Kratošová, S. Brožová and S. K. Sathish, Recyclability ,circular economy, and environmental aspects of lithiumesulfur batteries. Lithium-Sulfur Batteries. Elsevier, 2022. p.653–672.
[11] D. H. Yoon and Y. J. Park,
J. Electrochem. Sci. Technol.,
2021,
12(
1), 126–136.


[12] S. Gifford and J. Robinson, Lithium-sulfur batteries: lightweight technology for multiple sectors. Faraday Insights The Faraday Institution. UK,
2020, 8, Available:
www.faraday.ac.uk
.
[13] G. Palissat, Lithium-sulphur batteries: opportunities and challenges for space applications. In: 8th european conference for aeronautics and space sciences (EUCASS); 2019; pp 1–12.

[14] A. Fotouhi, D. J. Auger, L. O’Neill, T. Cleaver and S. Walus,
Energies,
2017,
10(
12), 1937.

[15] L. Fan, M. Li, X. Li, W. Xiao, Z. Chen and J. Lu,
Joule,
2019,
3(
2), 361–386.

[16] M. Zhao, B. Li, X. Zhang, J. Huang and Q. Zhang,
ACS Cent. Sci.,
2020,
6(
7), 1095–1104.


[17] M. J. M. Al Essa, J. Energy Storage, 2018, 18, 380–388.
[18] M. J. M. Al Essa, J. Energy Storage, 2020, 31, 101762.
[19] M. J. M. Al Essa, J. Energy Storage, 2023, 59, 106514.
[20] M. J. M. Al Essa,
J. Control. Autom. Electr. Syst.,
2020,
31, 1520–1532.


[21] G. Li, Z. Chen and J. Lu,
Chem,
2018,
4(
1), 3–7.

[22] A. M. Vassallo, Applications of batteries for grid-scale energy storage. Advances in Batteries for Medium and Large-Scale Energy Storage. Woodhead Publishing,
2015. p.587–607.

[23] O. Babayomi, Z. Zhang, T. Dragicevic, J. Hu and J. Rodriguez,
Int. J. Electr. Power Energy Syst.,
2023,
147, 108812.

[24] M. J. M. Al Essa and L. M. Cipcigan, Effects of randomly charging electric vehicles on voltage unbalance in micro grids. In: 2015 50th International Universities Power Engineering Conference (UPEC); Stoke on Trent, UK. 2015; pp 1–6.

[25] M. J. M. Al Essa and L. M. Cipcigan, Appl. Sci., 2016, 6(2), 53.
[26] M. J. M. Al Essa and L. M. Cipcigan, Integration of renewable resources into Low Voltage grids stochastically. In: 2016 IEEE International Energy Conference (ENERGYCON); Leuven, Belgium. 2016; pp 1–5.

[27] M. J. M. Al Essa,
Electr. Eng.,
2023,
105, 1761–1773.


[28] D. Kaur, M. Singh and S. Singh, Lithiume-Sulfur batteries for marine applications. Lithium-Sulfur Batteries. Elsevier, 2022. p.549–577.
[29] N. Deng, Y. Li, Q. Li, Q. Zeng, S. Luo, H. Wang, W. Kang and B. Cheng,
Energy Storage Mater.,
2022,
53, 684–743.

[30] S. A. Han, H. Qutaish, J.-W. Lee, M. Park and J. H. Kim, EcoMat., 2023, 5(2), e12283.
[31] A. B. Haruna, T. P. Mofokeng, J. J. Ogada, O. Zoubir, A. Lallaoui, F. C. El Moursli, Z. Edfouf and K. I. Ozoemena,
Electrochem. Commun.,
2022,
136, 107248.

[32] K. Subasinghage, K. Gunawardane, N. Padmawansa, N. Kularatna and M. Moradian,
Energies,
2022,
15(
20), 7752.

[33] E. A. Worsley, S. Margadonna and P. Bertoncello,
Nanomaterials,
2022,
12(
20), 3600.

[34] A. Swiderska-Mocek, E. Rudnicka and A. Lewandowski,
J. Solid State Electrochem.,
2020,
24, 1157–1164.


[35] T. Glossmann, A. Raj, T. Pajan and E. Buch, Introduction to the lithium-sulfur system?: Technology and electric vehicle applications. Lithium-Sulfur Batteries. Elsevier,
2022. p.3–15.

[36] S. Yang, Z. Zhang, J. Lin, L. Zhang, L. Wang and S. Chen, Front. Energy Res., 2022, 10, 945003.
[37] R. Mori,
J. Solid State Electrochem.,
2023,
27, 813–839.


[38] Z.-L. Xu, J.-K. Kim and K. Kang,
Nano Today,
2018,
19, 84–107.

[39] J. E. Knoop and S. Ahn,
J. Energy Chem.,
2020,
47, 86–106.

[40] X. Sun, S. Liu, W. Sun and C. Zheng,
Chin. Chem. Lett.,
2023,
34(
1), 107501.

[41] N. Ding, S. Wei, T. S. A. Hor, Z. Liu and Y. Zong,
J. Power Sources,
2014,
269, 111–116.

[42] K. R. Bugga, Prospects and Challenges of Lithium-Sulfur Batteries What makes Li-S attractive. In: Li-S Symposium, Military Power Sources Committee (MPSC) Workshop; 2021.
[43] A. I. Kamisan, T. I. T. Kudin, A. S. Kamisan, A. F. C. Omar, M. F. M. Taib, O. H. Hassan, A. M. M. Ali and M. Z. A. Yahya,
Int. J. Hydrogen Energy,
2022,
47(
13), 8630–8657.

[44] D. Lv, J. Zheng, Q. Li, X. Xie, S. Ferrara, Z. Nie, L. B. Mehdi, N. D. Browning, J.-G. Zhang, G. L. Graff, J. Liu and J. Xiao,
Adv. Energy Mater.,
2015,
5(
16), 1402290.

[45] C. Xu, T. Cleary, D. Wang, G. Li, C. Rahn, D. Wang, R. Rajamani and H. K. Fathy, Online state estimation for a physics-based Lithium-Sulfur battery model.
J. Power Sources,
2021,
489, 229495.

[46] R. Demir-Cakan, Introduction to Rechargeable Lithium–Sulfur Batteries. Li–S Batteries. World Scientific,
2017. p.1–30.

[47] H. Wang, J. Dong, K. Schelkun, S. Penski, C. Silkowski, M. Wixom and L. Alexander, Development of Lithium Sulfur Batteries for High Energy Applications. In: 2020 NASA Aerospace Battery Workshop; 2020.
[48] R. Thangavel, A. G. Kannan, R. Ponraj, K. Kaliyappan, W.-S. Yoon, D.-W. Kim and W.-S. Lee,
Nanomaterials,
2020,
10(
6), 1220.

[49] W. Han, Z. Li and R. Wang,
IOP Conf. Ser.: Earth Environ. Sci.,
2022,
1011, 012004.


[50] S. Dörfler, H. Althues, P. Härtel, T. Abendroth, B. Schumm and S. Kaskel,
Joule,
2020,
4(
3), 539–554.

[51] Q. Zhu, C. Ye and D. Mao,
Nanomaterials,
2022,
12(
20), 3612.

[52] X. Zhao, C. Wang, Z. Li, X. Hu, A. A. Razzaq and Z. Deng,
J. Mater. Chem. A,
2021,
9, 19282–19297.

[53] W. Xue, L. Miao, L. Qie, C. Wang, S. Li, J. Wang and J. Li,
Curr. Opin. Electrochem.,
2017,
6(
1), 92–99.

[54] U. Stoeck, J. Balach, M. Klos, D. Wadewitz, E. Ahrens, J. Eckert and L. Giebeler,
J. Power Sources,
2016,
309, 76–81.

[55] M. Yan, W.-P. Wang, Y.-X. Yin, L.-J. Wan and Y.-G. Guo,
EnergyChem,
2019,
1, 100002.

[56] S. Ni, S. Tan, Q. An and L. Mai,
J. Energy Chem.,
2020,
44, 73–89.

[57] P. Modiba, M. Matoetoe and A. M. Crouch,
Anal. Lett.,
2011,
44(
11), 1967–1975.

[58] X. Wang, Y. Tan, G. Shen and S. Zhang,
J. Energy Chem.,
2020,
41, 149–170.

[59] D. A. Dornbusch, R. P. Viggiano, J. W. Connell, Y. Lin and V. F. Lvovich,
Electrochim. Acta,
2022,
403, 139406.

[60] S. Bandyopadhyay and B. Nandan,
Mater. Today Energy,
2023,
31, 101201.

[61] F. Shi, L. Zhai, Q. Liu, J. Yu, S. P. Lau, B. Y. Xia and Z.-L. Xu,
J. Energy Chem.,
2023,
76, 127–145.

[62] Y. Huang, X. Gao, X. Han, Z. Guang and X. Li,
Solid State Ion.,
2020,
347, 115248.

[63] S. Choudhury,
Curr. Opin. Electrochem.,
2020,
21, 303–310.

[64] M. J. Seong, S. Manivannan, K. Kim and T. Yim,
J. Electrochem. Sci. Technol. Electrochem.,
2021,
12(
4), 453–457.


[65] A. A. Abdelhamid, A. Mendoza-Garcia and J. Y. Ying,
Nano Energy,
2022,
93, 106860.

[66] M. Byakodi, N. S. Shrikrishna, R. Sharma, S. Bhansali, Y. Mishra, A. Kaushik and S. Gandhi,
Biosens. Bioelectron. X,
2022,
12, 100284.

[67] W. Liu, X. Qiao, S. Liu and P. Chen,
Nanomaterials,
2022,
12(
21), 3780.

[68] X. Yang, X. Li, K. Adair, H. Zhang and X. Sun,
Electrochem. Energ. Rev.,
2018,
1, 239–293.


[69] H. Qutaish, S. A. Han, Y. Rehman, K. Konstantinov, M.-S. Park and J. H. Kim,
Sci. Technol. Adv. Mater.,
2022,
23(
1), 169–188.

[70] Y. Hyeon, J. Lee, H. Qutaish, S. A. Han, S. H. Choi, S. W. Moon, M.-S. Park, D. Whang and J. H. Kim,
Energy Storage Mater.,
2020,
33, 95–107.

[71] J. Lee, S. H. Choi, H. Qutaish, Y. Hyeon, S. A. Han, Y.-U. Heo, D. Whang, J.-W. Lee, J. Moon, M.-S. Park, J. H. Kim and S. X. Dou,
Energy Storage Mater.,
2021,
37, 315–324.

[72] O. A. Oviedo and E. P. M. Leiva,
Curr. Opin. Electrochem.,
2017,
1(
1), 1–6.

[73] Álvaro. Doñoro, D. Cíntora-Juárez and V. Etacheri, Carbon nanomaterials for rechargeable lithium-Sulfur batteries. Micro and Nano Technologies, Carbon Based Nanomaterials for Advanced Thermal and Electrochemical Energy Storage and Conversion. Elsevier, 2019. p.279–309.
[74] J. Park, S.-H. Yu and Y.-E. Sung,
Nano Today,
2018,
18, 35–64.

[75] H. S. Kim, T.-G. Jeong and Y.-T. Kim,
J. Electrochem. Sci. Technol. Electrochem.,
2016,
7(
3), 228–233.


[76] W. Zhao, W. Choi and W. Yoon,
J. Electrochem. Sci. Technol. Electrochem.,
2020,
11(
3), 195–219.


[77] L. Ma, K. E. Hendrickson, S. Wei and L. A. Archer,
Nano Today,
2015,
10(
3), 315–338.

[78] K. Zhu, C. Wang, Z. Chi, F. Ke, Y. Yang, A. Wang, W. Wang and L. Miao, Front. Energy Res., 2019, 7, 123.
[79] J. B. Robinson, K. Xi, R. V. Kumar, A. C. Ferrai, H. Au, M.-M. Titirici, A. Parra-Puerto, A. Kucernak, S. D. S. Fitch, N.. Garcia-Araez and et al,
J. Phys. Energy,
2021,
3, 031501.


[81] W. Zhang, S. Li, A. Zhou, H. Song, Z. Cui and L. Du,
Molecules,
2021,
26(
21), 6341.

[82] J. Sun, T. Wang, Y. Gao, Z. Pan, R. Hu and J. Wang, InfoMat, 2022, 4(9), e12359.
[83] D. Lu, Y. Shao, T. Lozano, W. D. Bennett, G. L. Graff, B. Polzin, J. Zhang, M. H. Engelhard, N. T. Saenz, W. A. Henderson, P. Bhattacharya, J. Liu and J. Xiao,
Adv. Energy Mater.,
2015,
5(
3), 1400993.

[85] S. Nanda, A. Bhargav and A. Manthiram,
Joule,
2020,
4(
5), 1121–1135.

[86] G. Zhou, H. Chen and Y. Cui,
Nat. Energy,
2022,
7, 312–319.


[87] C. Xu, Q. Dai, L. Gaines, M. Hu, A. Tukker and B. Steubing, Commun. Mater., 2020, 1, 99.