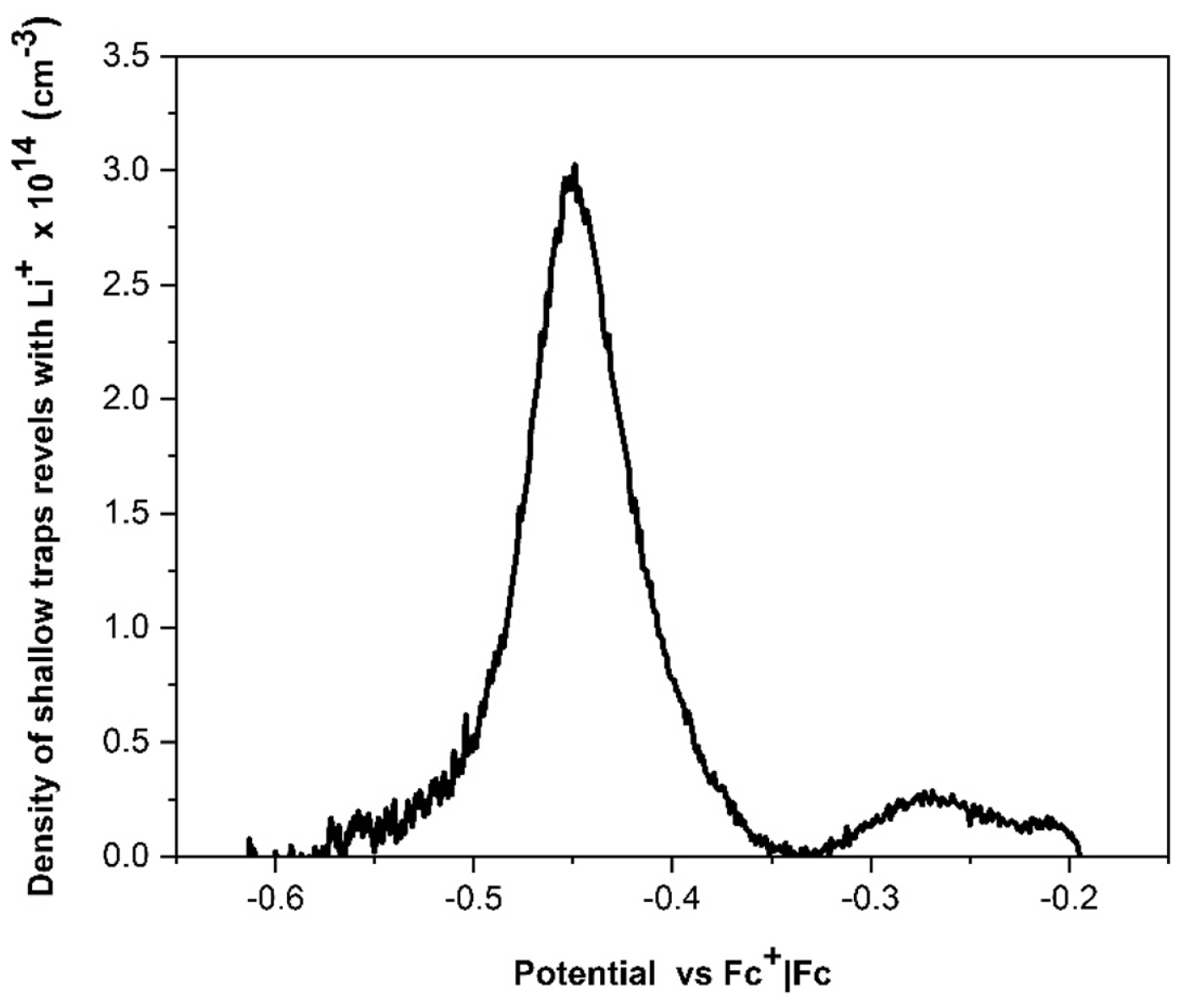
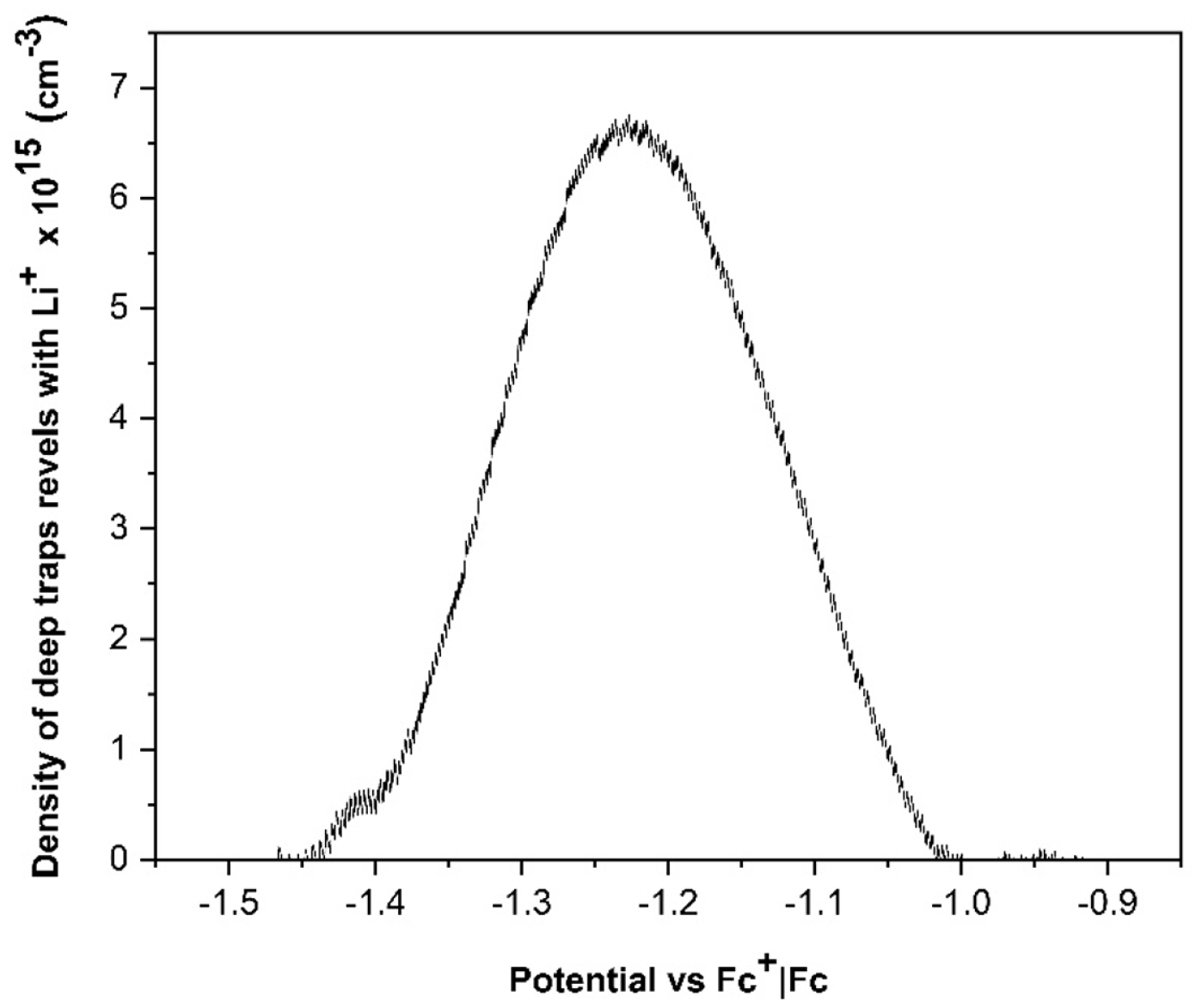
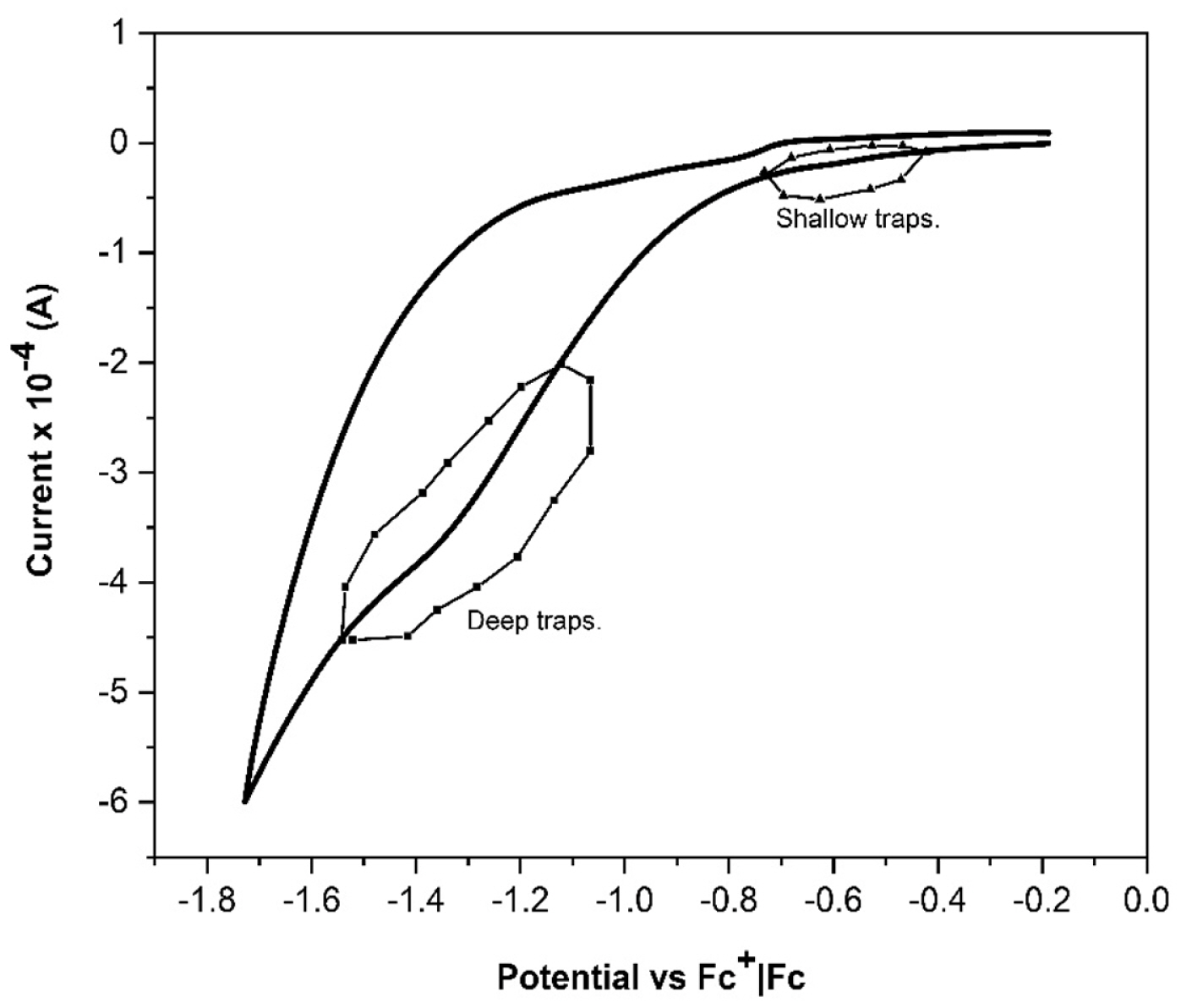
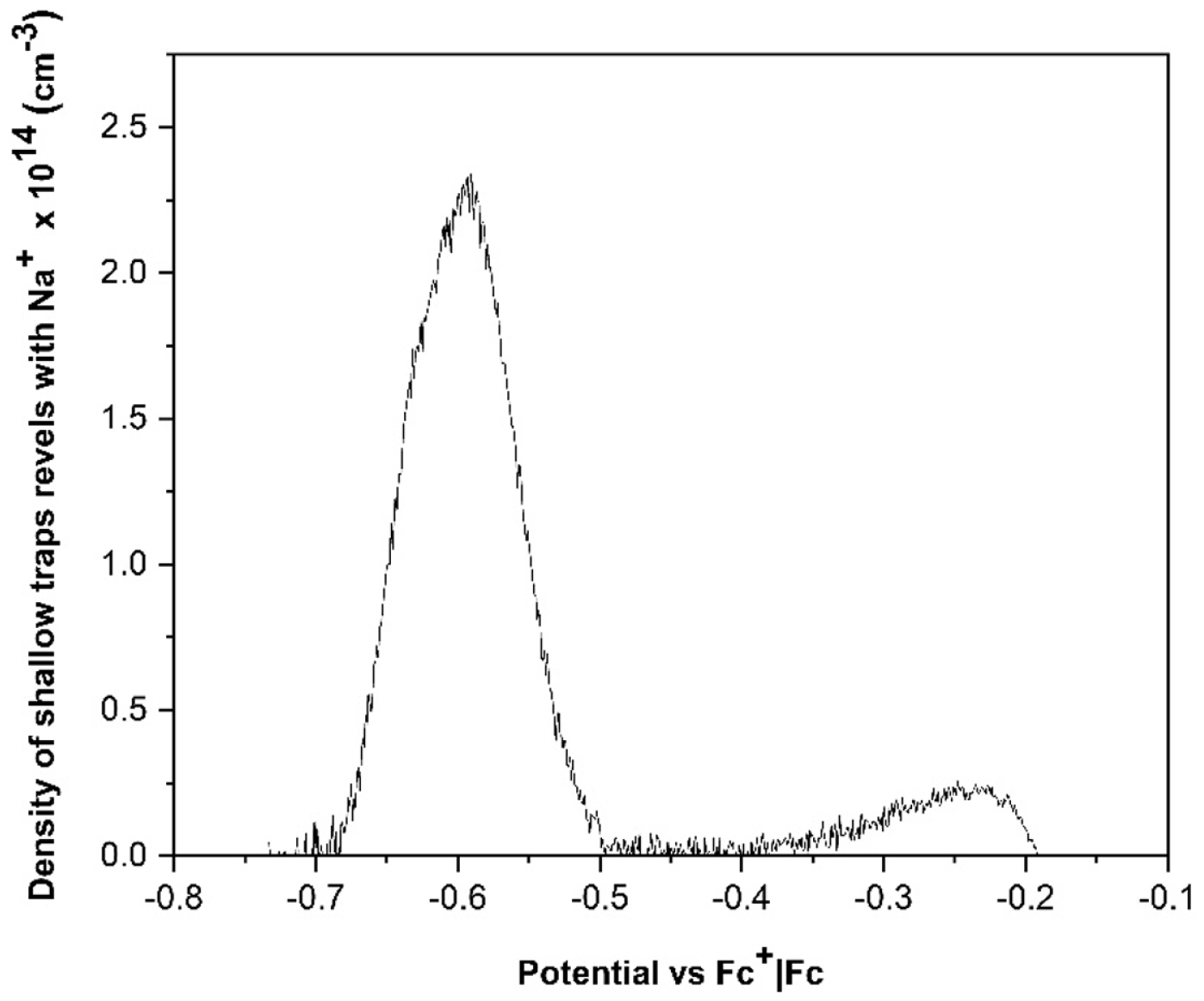
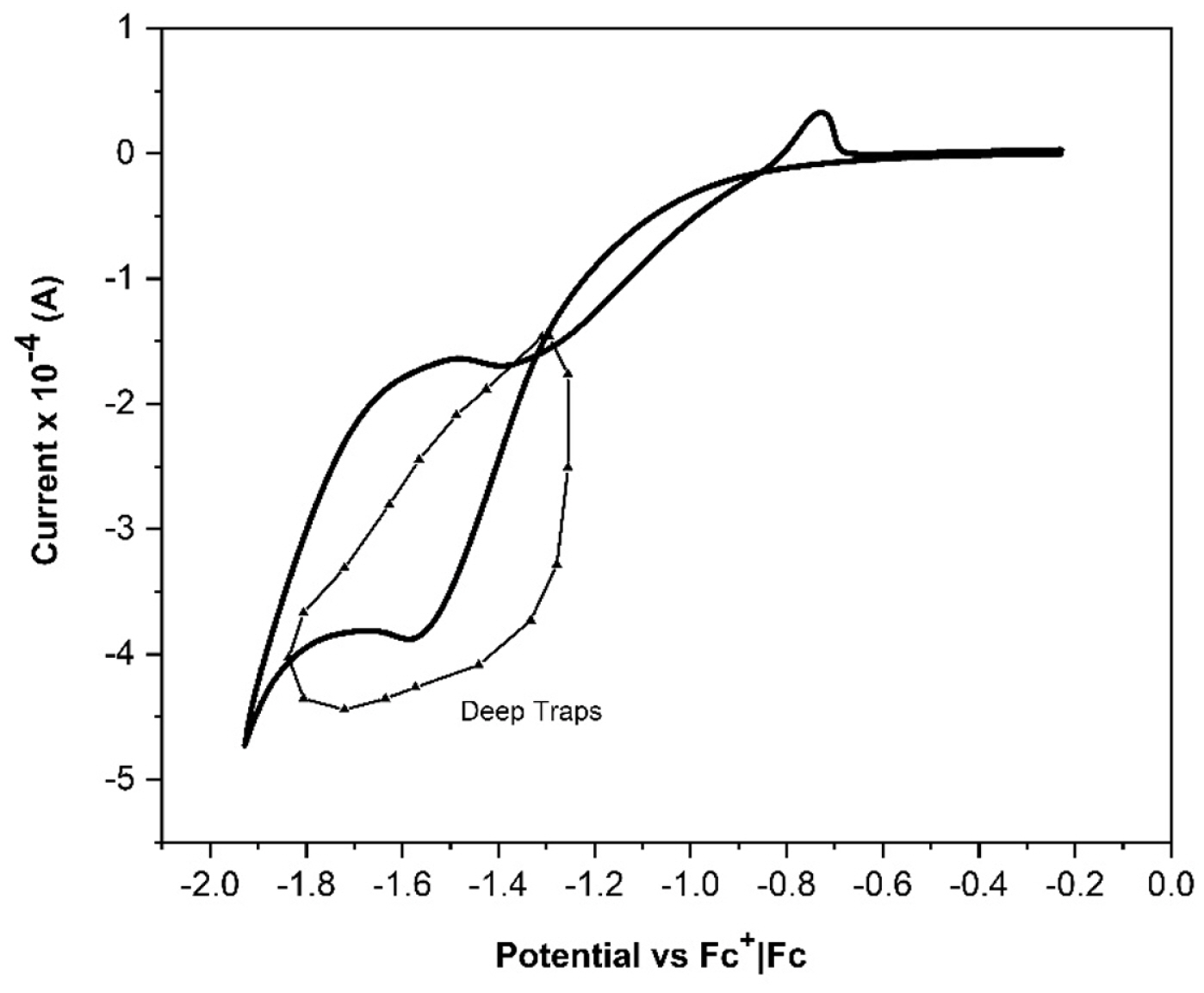
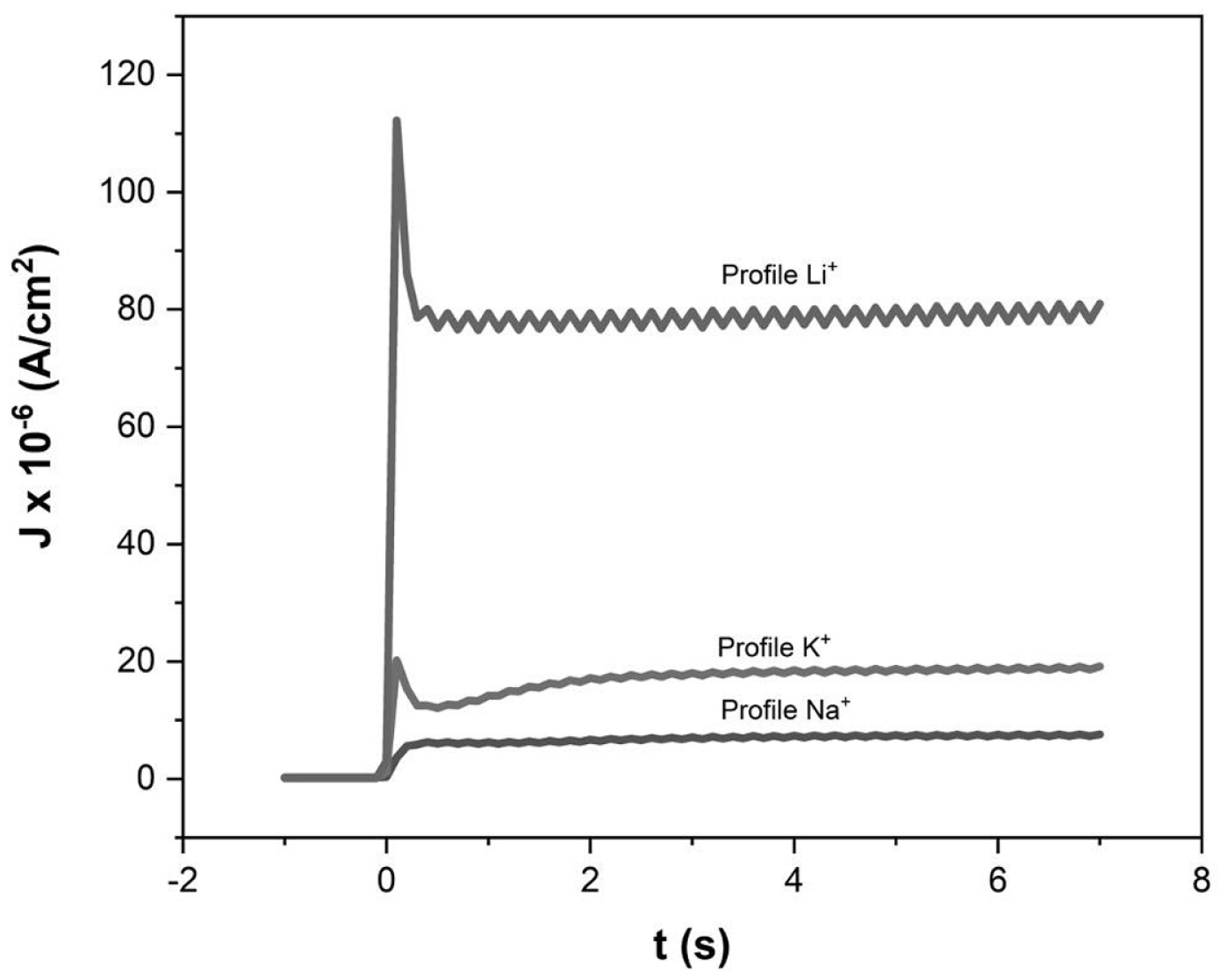
When a DSSC undergoes an abrupt dark-to-light transition, the performance of the cell is influenced by the electron diffusion coefficient, Dn(n). It depends on the electron density and the characteristics and concentration of the added ions; these produce important variations on the magnitude and shape of the photocurrent profiles. The diffusion coefficients of both, the transient and steady states in dark and illumination conditions, were obtained by Cyclic Voltammetry (CV). The chemical capacitance for each ion in illumination condition,
Cö¥ionã(Cö¥Li+,ãCö¥Na+,ãCö¥K+), was first determined; these can be associated with the trap states of the TiO2 film; this information allows to determine the electron density in that state; it is possible to mention that the characteristics and concentration of added ions control in some way affect the electron distribution on the mesoporous surface.
The photoelectrochemical impedance technique was used to determine the diffusion coefficients for each added ion:
Dnss,.lightLi+=5.59û10-4ãcm2ãs-1,Dnss,.lightNa+=3.46û10-4ãcm2ãs-1 and
Dnss,.lightK+=1.33û10-4ãcm2ãs-1. To determine the dark and transient state diffusion coefficients, an dark-light (8 seconds, total time) profile with an intensity 100mW cmã2 (7 seconds-light) was used for each DSSC allowing to obtain the dark diffusion coefficients for each ion:
Dnss,.darkLi+=38.1ãcm2ãs-1ûDnss,.darkNa+=9.86ãcm2ãs-1 and
Dnss,.darkK+=12.41ãcm2ãs-1. For the transient state:
Dntrans.Li+=1.86û10-5ãcm2ãs-1,Dntrans.Na+=2.494û10-5ãcm2ãs-1 and
Dntrans.K+=6.598û10-3ãcm2ãs-1.
2.2 Modeling the electron diffusion coefficient
As it is known, in a DSSC, electrons are generated through the dye molecules and diffuse throughout the film via the surface trap states and conduction band, moreover, the generated electrons can recombine with the oxidized species at the TiO
2/E interface [
16,
25]. Such processes can be modeled by the electrochemical impedance proposed by Bisquert
et al. [
15]. For the study of the transition from the dark state to the light state, a hybrid model of the carrier source is given by:
Such a model consists of a Heaviside step function H(t) in time for which a constant function in time has been taken for t ãË 0 in the illumination state, but dependent on the position given by the Lambert-Beer absorption law. Here öÝ represents the effective adsorption coefficient of the pigment molecules distributed on the film of thickness L, and I0 is the irradiance or the coefficient of the effective flux of incident photons. Thus, the model describing the system is given by:
Where
C2 represents the concentration of carriers in the solid phase (
2), (electrons),
C20 is the initial concentration of the same species in the same phase,
k is the recombination frequency, and
Dn the effective electron diffusion coefficient in the film.
However, during the dark-to-light transition, which is when the electronic charge is injected in an order of femtosecond [
26], the recombination can be neglected since it is of the order of 10
1ã10
2 Hz in this case. Therefore,
Eq. (4) can be rewritten in the transient state as:
In reported studies [
27], it has been shown that the electron diffusion coefficient is highly dependent on the same electronic concentration
C2:
Where
l ã2ã3, is a parameter related to the distribution of the trap states [
28]. Then, the model is a nonlinear partial differential equation with no analytical solutions. At Ref. [
23], solutions were determined by numerical methods for illum,ination pulses of
Eq. (5), but those results do not fit the experimental results. Therefore, in the study of dark-illumination transients proposed in this work, an approximation is used in which
Dn is time-dependent rather than electron concentration-dependent:
Where
Dnãý is the effective dark steady-state diffusion coefficient (DSDC) in t ãÊ 0, or when C2 = C20 and Dn is the effective diffusion coefficient in the illuminated state (EDCI) in t ãË 0 of the transient part. This allows for avoiding the nonlinearity of the model as it is shown in the following equation for the approximate model in the transient:
Where the carrier source given by
Eq. (3) has also been used. On the other hand, the boundary conditions when measuring the photocurrent generated by the DSSC in short circuit will be given by:
The first condition implies that in TCO the concentration is only time-dependent and is proportional to the concentration gradient evaluated in x = 0, where the proportionality constant is given by the ratio of the electron extraction rate kext. at the TCO contact.
At
x = 0, due to the low charge generated by the NOP used [
29], it is considered that there is no electrical or transport impediment for the electrons to reach the collector, this is reflected due to the low screening of the electrons arriving at
x = 0 and that there is a maximum in the concentration gradient in that region. Therefore, at this boundary, it can be considered that the charge control is first-order kinetic (Arrhenius type) and consequently the diffusion coefficient in that region will be given by
At the other boundary, in x = L, at the mixed-phase electrode boundary (MPEB) and the bulk of the electrolyte was determined with the electron flow completely blocked, thus the concentration gradient is minimal and equal to zero and hence there is no electron diffusion in that zone.
The solution for the model C2 (x, t) is given by:
That through the continuity conditions of the carrier density: ntãÊ0(0,0) = ntãË0(0,0) and the current density in the whole film: JtãÊ0(x,0) = JtãË0(x,0), it is obtained the parameter relationship with the coefficients: c1, c2, c3 and Bn
Parameters of model the DSSC for Li+; c4 and c5 are parameters determined by fitting experimental results.
As it can be observed
Eq. (10b) for
t ãË 0 is a summation of different exponentials, which could represent the contribution of each of the charge species contributing to the current in phase (
2). Each species is identified by its diffusion frequency given by:
Where ün is the diffusion frequency of the umpteenth component of the current.
Up to this point, it has been considered in the model that the charge transfer at the bottom, i.e., at the phase (
1) or TCO/electrolyte (TCO/E) interface of the pore is zero, however, sometimes the photocurrent measured at the TCO (
x = 0) is affected by the charges accumulated at the bottom of the pores of the mixed electrode and in the steady state can be taken into account by means of the impedance
ZB of
Fig. 2. When this area is exposed directly with the electrolyte both recombination and charge accumulation can arise [
13]. Since both processes demand charge in that region, they can modify the photocurrent profile measured in the transient.
In these cases, the model is modified if it is initially assumed that a quantity of carriers: öÇ(
x)[
C2|
t ãÊ 0(
x = 0,
t = 0)], is injected into the TCO from the sensitized TiO
2 layers at the TCO/TiO
2 + dye interface; where
C2|
t ãÊ 0 indicates that the evaluation is done in part of the solution when
t ãÊ 0. Once the charge is injected into the TCO, some carriers tend to return to the mixed-phase electrode by a new reinjection at the TCO/E interface but in a different direction (from the TCO to the electrolyte). As discussed above, the movement of carriers in the opposite direction is due to the accumulation of ions of different signs at the TCO/E interface and may cease until their steady state impedance Z
B in direct current is obtained. Since charge neutrality tends to be preserved in the film, it is equivalent to thinking that in
x =
L, a number of holes has been injected equal to the quantity of electrons injected in
x = 0, and that the concentration in the same region is zero. One way to guarantee this boundary condition is to establish a charge of equal magnitude, but different sign at
x = ã
L, the other condition will be given by the blocking of electrons at
x =
L. Considering that the same model of
Eq. (8) for
t ãË 0 and the initial condition mentioned above we have that the electron concentration will be given by:
Since the measured current density is given by:
J=qDnãC2ãx|x=0, where q is the electron charge. In addition, it is noted that the current density profile will depend on the hole diffusion coefficient Dh and the electron diffusion coefficient Dn of the mixed phase electrode:
For the part of
t ãÊ 0 the solution will be given in the same way by
Eq. (10) respectively.