1. Introduction
2. Experimental
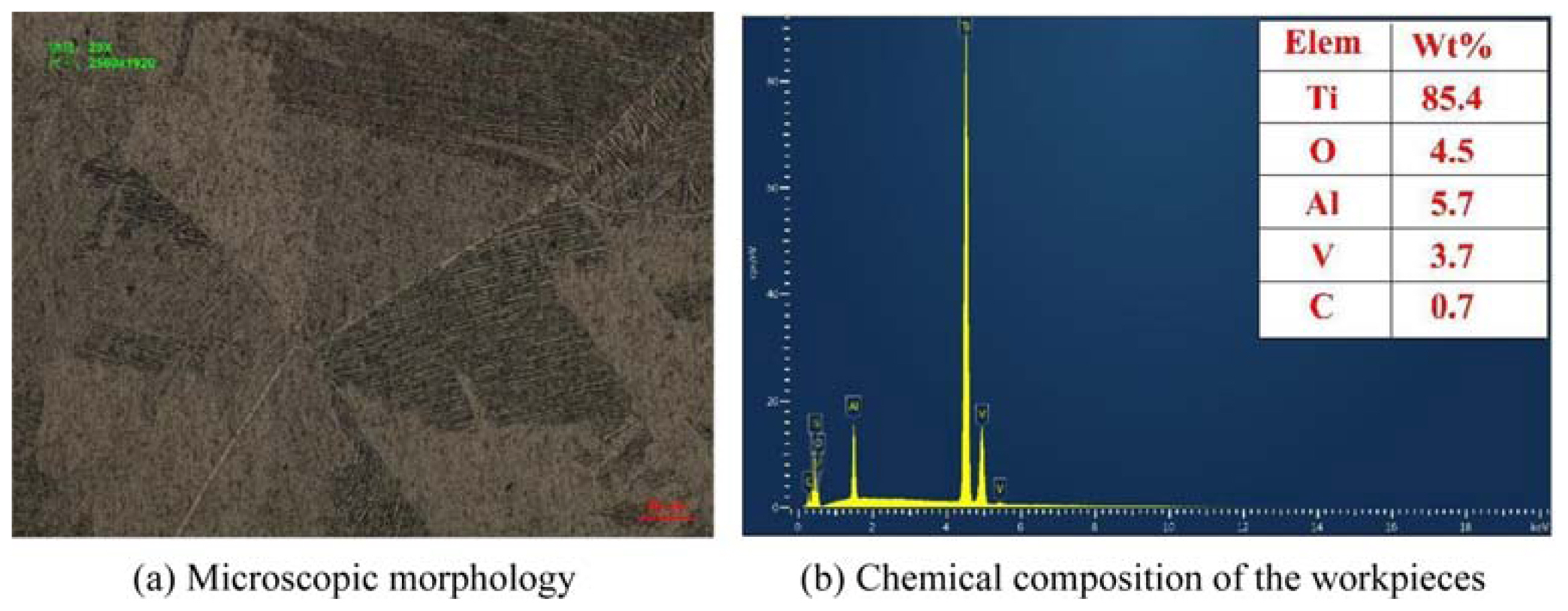
2.1 Experimental details
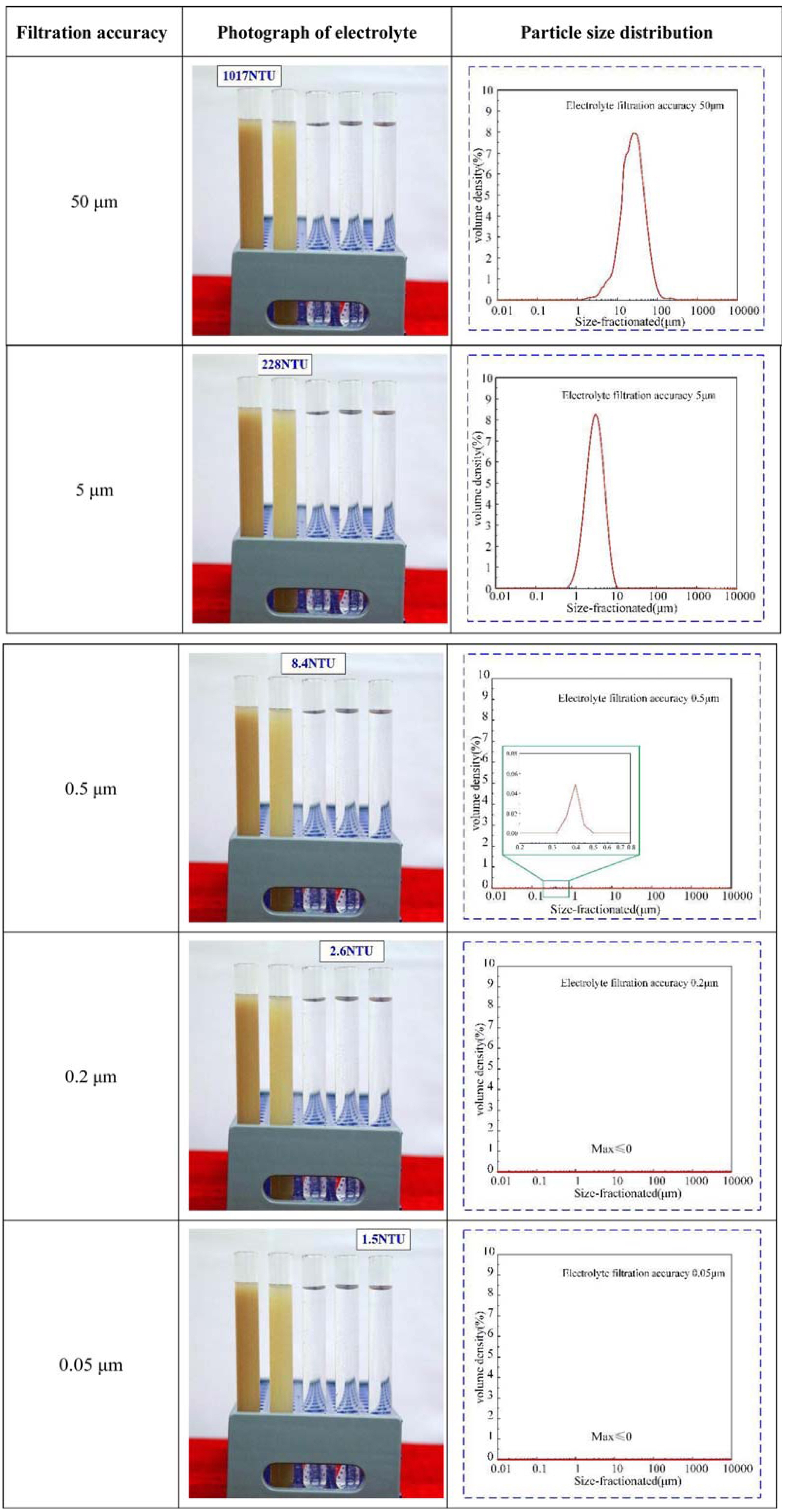
2.2 Electrolyte preparation and characterization
3. Simulations
3.1 Simulation conditions
3.2 Analysis of simulation results
4. Machining Experiments
5. Results and Discussion
5.1 Material dissolution
5.2 Surface quality
5.3 Cross-sectional profile and repeatability
6. Conclusions
The simulation results indicated that the electrolyte filtration accuracy improves from 50 to 0.05 μm, and the Joule heating decreases in the processing area from 315.7K to 311K. This indicates that the Joule heating in the processing area correlates certain with the electrolyte filtration accuracy. Simultaneously, the bubble rate in the processing area decreases from 20.2% to 11.6%, i.e., by a decrease of nearly 43.5%. The results mean that the electrolyte filtration accuracy has a more significant impact on the bubble rate compared to the temperature in the processing area.
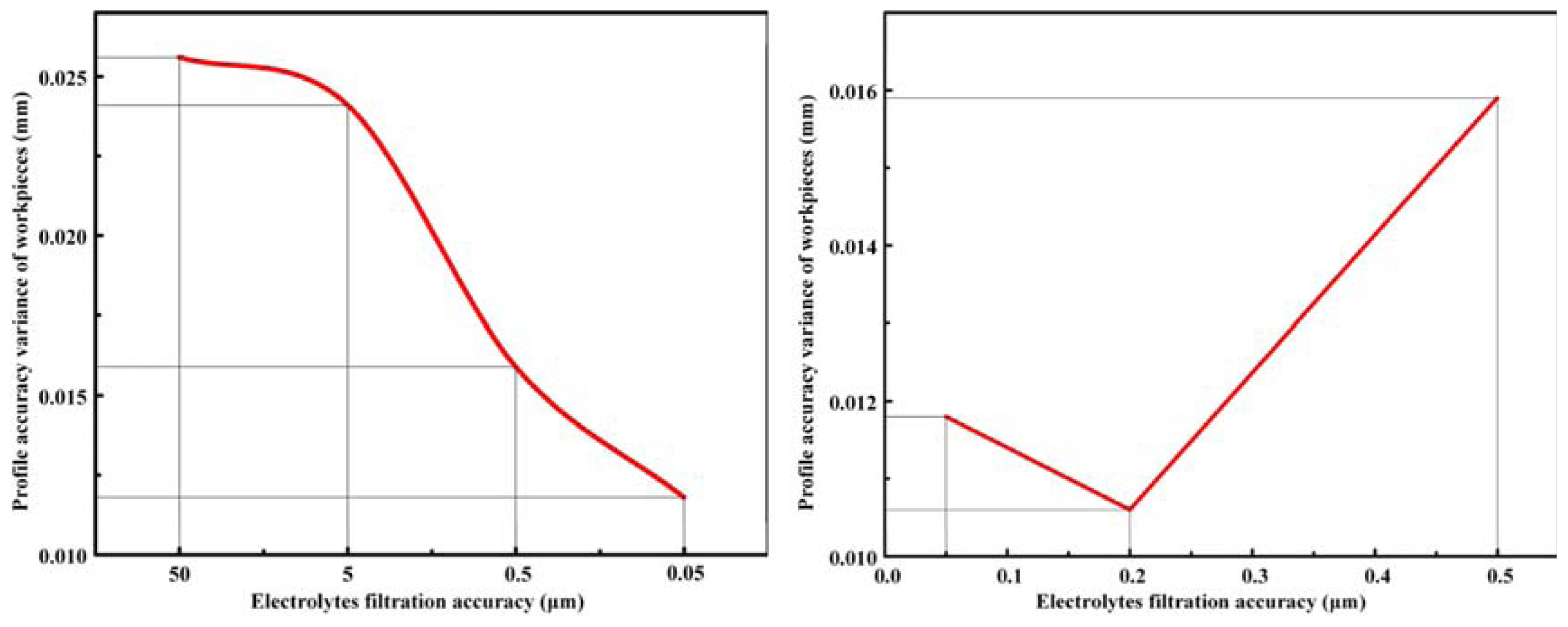
The results of the ECM experiments showed that increasing the electrolyte filtration accuracy improves the workpiece machining quality. Measurement of the workpieces found that the surface roughness decreased from Ra=1.425 μm to Ra=0.753 μm as the filtration accuracy improved from 5 μm to 0.05 μm, the workpieces surface quality has been significantly improved, reaching 89.24%. As the electrolyte filtration accuracy improved from 50 μm to 0.05 μm, the machining accuracy improved from 0.0863 mm to 0.0425 mm, and the repetition accuracy improved from 0.0689 to 0.0403 mm. When the electrolyte filtration accuracy was higher than the level range of 0.5 μm, further improvements in filtration accuracy produce only a very limited gain in machining quality.
The trade-off between the improved machining quality and increasing economic cost means, that an electrolyte filtration accuracy maintained within the level range of 0.5 μm is a reasonable choice for further large-scale development of precision ECM technology. Especially for manufacturing blade profiles or inlet and exhaust edges, the electrolyte filtration accuracy in the level range of 0.5 μm is contributed to obtaining better machining quality.