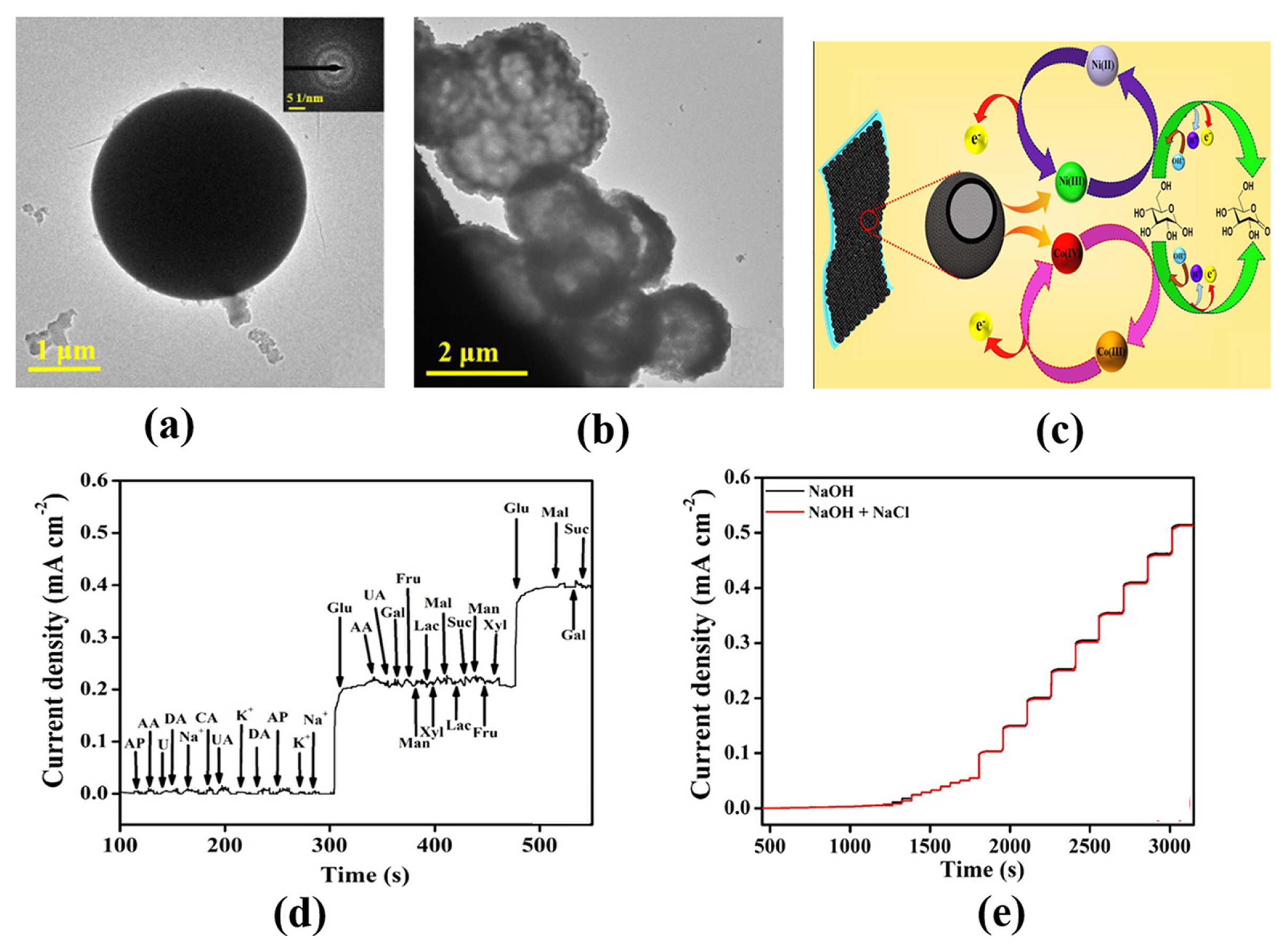
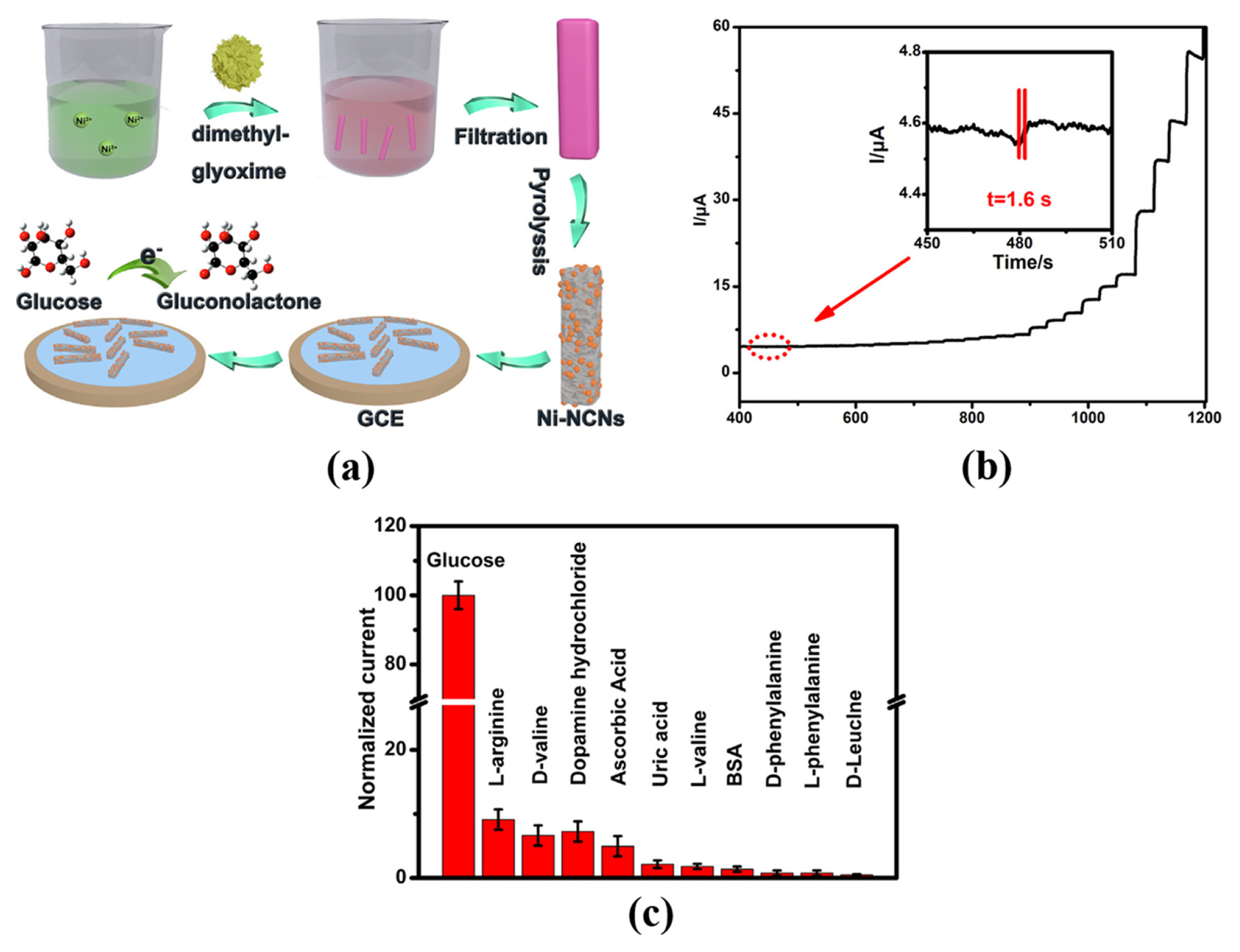
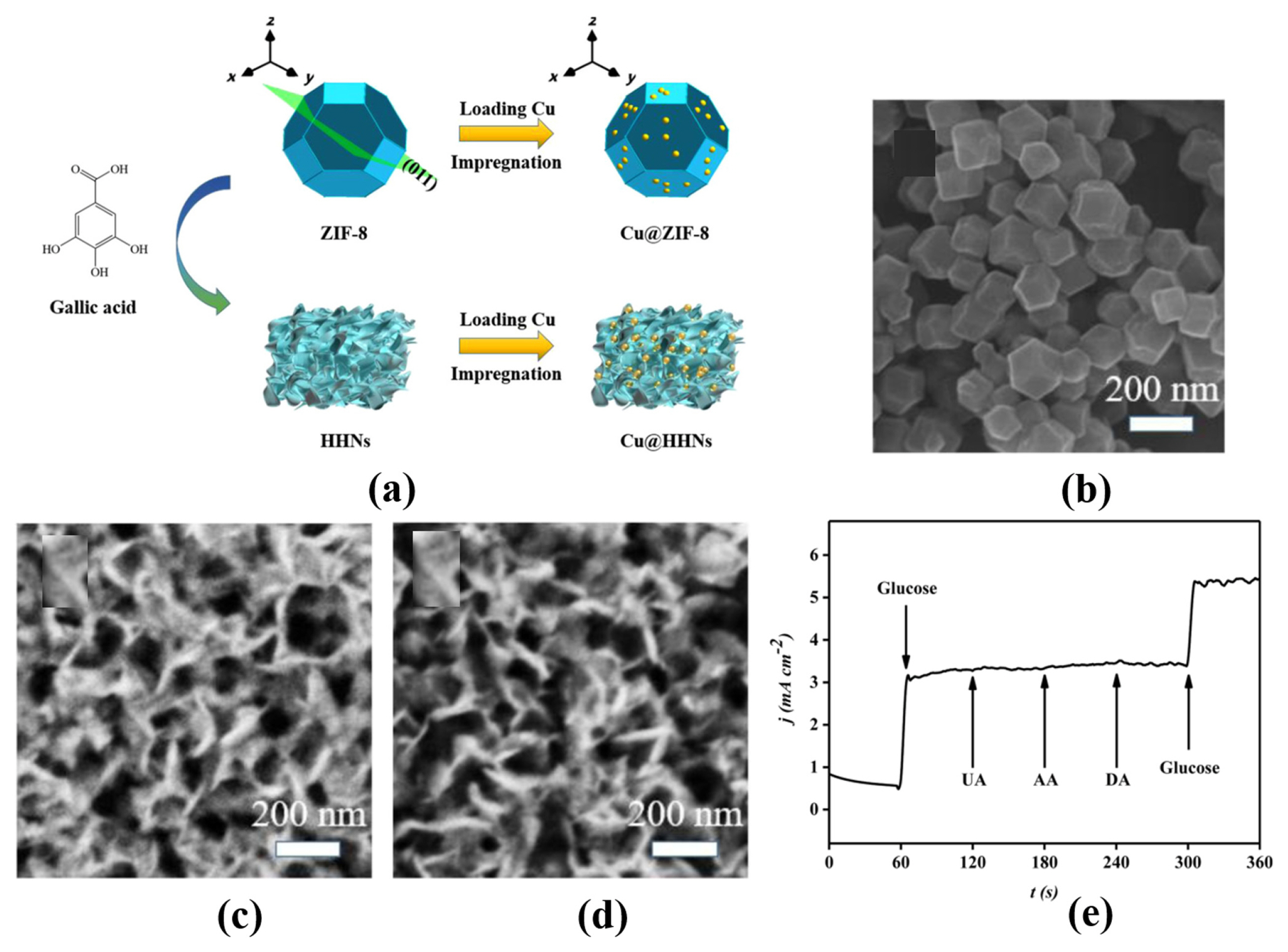
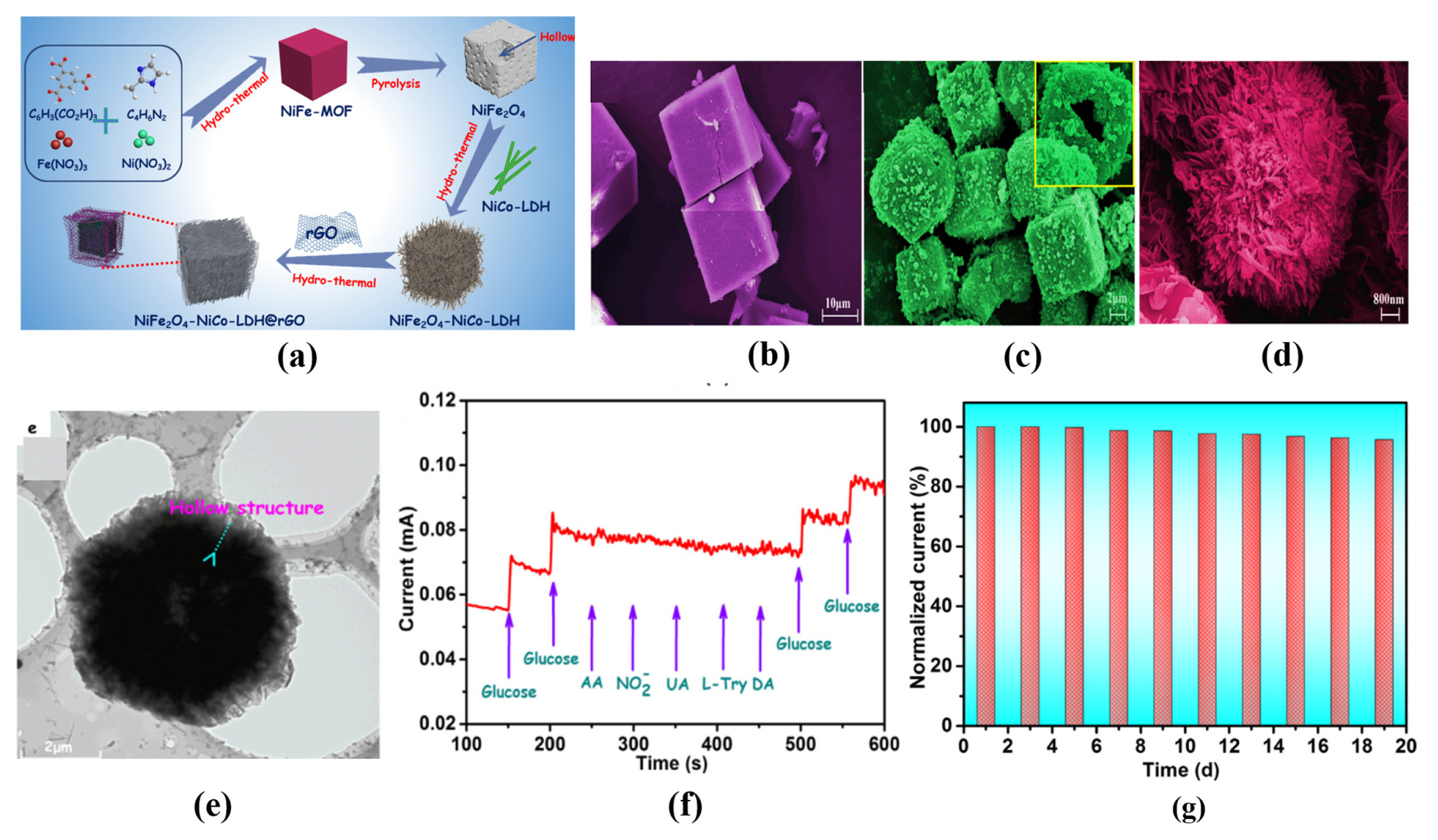
[1] D. W. Hwang, S. Lee, M. Seo and T. D. Chung,
Anal. Chim. Acta.,
2018,
1033, 1–34.

[2] K. Dhara and D. R. Mahapatra, Microchim. Acta., 2017, 185, 49.
[3] M. Wei, Y. Qiao, H. Zhao, J. Liang, T. Li, Y. Luo, S. Lu, X. Shi, W. Lu and X. Sun,
Chem. Commun.,
2020,
56, 14553–14569.

[4] S. A. Shafiee, S. C. Perry, H. H. Hamzah, M. M. Mahat, F. A. Al-lolage and M. Z. Ramli,
Electrochem. Commun.,
2020,
120, 106828.

[5] R. Ahmad, M. Khan, P. Mishra, N. Jahan, M. A. Ahsan, I. Ahmad, M. R. Khan, Y. Watanabe, M. A. Syed, H. Furukawa and A. Khosla,
J. Electrochem. Soc.,
2021,
168, 017501.


[6] H. Jia, N. Shang, Y. Feng, H. Ye, J. Zhao, H. Wang, C. Wang and Y. Zhang,
J. Colloid Interface Sci.,
2021,
583, 310–320.

[7] G. A. Naikoo, H. Salim, I. U. Hassan, T. Awan, F. Arshad, M. Z. Pedram, W. Ahmed and A. Qurashi, Front. Chem., 2021, 9, 748957.
[8] J. R. Sempionatto, J.-M. Moon and J. Wang,
ACS Sens,
2021,
6(
5), 1875–1883.

[9] M. Bariya, H. Y. Y. Nyein and A. Javey,
Nat. Electron,
2018,
1, 160–171.


[10] H. Lee, Y. J. Hong, S. Baik, T. Hyeon and D.-H. Kim, Adv. Healthc. Mater., 2018, 7(8), 1701150.
[11] R. Sakamoto, N. Fukui, H. Maeda, R. Toyoda, S. Takaishi, T. Tanabe, J. Komeda, P. Amo-Ochoa, F. Zamora and H. Nishihara,
Coord. Chem. Rev.,
2022,
472, 214787.

[12] O. K. Farha, I. Eryazici, N. C. Jeong, B. G. Hauser, C. E. Wilmer, A. A. Sarjeant, R. Q. Snurr, S. T. Nguyen, A. Ö. Yazaydin and J. T. Hupp,
J. Am. Chem. Soc.,
2012,
134(
36), 15016–15021.

[13] A. Mahmood, W. Guo, H. Tabassum and R. Zou, Adv. Energy Mater., 2016, 6(17), 1600423.
[14] B. E. Lewis, N. Choytun, V. L. Schramm and A. J. Bennet,
J. Am. Chem. Soc.,
2006,
128(
15), 5049–5058.

[15] F. Largeaud, K. B. Kokoh, B. Beden and C. Lamy,
J. Electroanal. Chem.,
1995,
397(
1–2), 261–269.

[16] B. Lertanantawong, A. P. O’Mullane, W. Surareungchai, M. Somasundrum, L. D. Burke and A. M. Bond,
Langmuir,
2008,
24(
6), 2856–2868.

[17] L. D. Burke,
Electrochim. Acta.,
1994,
39(
11–12), 1841–1848.

[18] Y. Wang, R. Jin, N. Sojic, D. Jiang and H.-Y. Chen,
Angew. Chem. Int. Ed. Engl,
2020,
59(
26), 10416–10420.


[19] R. D. Munje, S. Muthukumar and S. Prasad,
Sens. Actuators B: Chem.,
2017,
238, 482–490.

[20] W. Gao, S. Emaminejad, H. Y. Y. Nyein, S. Challa, K. Chen, A. Peck, H. M. Fahad, H. Ota, H. Shiraki, D. Kiriya, D.-H. Lien, G. A. Brooks, R. W. Davis and A. Javey,
Nature,
2016,
529, 509–514.


[21] J. Zhu, S. Liu, Z. Hu, X. Zhang, N. Yi, K. Tang, M. G. Dexheimer, X. Lian, Q. Wang, J. Yang, J. Gray and H. Cheng,
Biosens. Bioelectron,
2021,
193, 113606.

[22] M. Wei, Y. Qiao, H. Zhao, J. Liang, T. Li, Y. Luo, S. Lu, X. Shi, W. Lu and X. Sun,
Chem. Commun.,
2020,
56, 14553–14569.

[23] X. Zha, W. Yang, L. Shi, Y. Li, Q. Zeng, J. Xu and Y. Yang,
ACS Appl. Mater. Interfaces,
2022,
14, 37843–37852.


[24] S. Hu, Y. Lin, J. Teng, W.-L. Wong and B. Qiu, Mikrochim. Acta., 2020, 187, 670.
[25] T. Meng, N. Shang, J. Zhao, M. Su, C. Wang and Y. Zhang,
J. Colloid Interface Sci.,
2021,
589, 135–146.

[26] M. A. Kachouei, S. Shahrokhian and M. Ezzati, Sens. Actuators B: Chem., 2021, 344, 130254.
[27] X. Li, H. Dong, Q. Fan, K. Chen, D. Sun, T. Hu and Z. Ni,
Microchem. J.,
2022,
179, 107468.

[28] K. Kim, J. Kim and Y.-S. Bae,
ACS Sustain. Chem. Eng.,
2022,
10(
35), 11702–11709.


[29] P. Li, Y. Bai, G. Zhang, X. Guo, X. Meng and H. Pang,
Inorg. Chem. Front,
2022,
9, 5853–5861.

[30] A. Vignesh, P. Vajeeston, M. Pannipara, A. G. Al-Sehemi, Y. Xia and G. G. kumar,
Chem. Eng. J.,
2022,
430, 133157.

[31] H. Jia, N. Shang, Y. Feng, H. Ye, J. Zhao, H. Wang, C. Wang and Y. Zhang,
J. Colloid Interface Sci.,
2021,
583, 310–320.

[32] G. Li, G. Xie, D. Chen, C. Gong, X. Chen, Q. Zhang, B. Pang, Y. Zhang, C. Li, J. Hu, Y. Chen, L. Yu and L. Dong,
Appl. Surf. Sci.,
2022,
585, 152683.

[33] J. Ding, L. Zhong, X. Wang, L. Chai, Y. Wang, M. Jiang, T.-T. Li, Y. Hu, J. Qian and S. Huang,
Sens. Actuators B: Chem.,
2020,
306, 127551.

[34] Y. Zhou, Q. Hu, F. Yu, G.-Y. Ran, H.-Y. Wang, N. D. Shepherd, D. M. D'Alessandro, M. Kurmoo and J.-L. Zuo,
J. Am. Chem. Soc.,
2020,
142(
48), 20313–20317.

[35] Q. Zhu, S. Hu, L. Zhang, Y. Li, C. Carraro, R. Maboudian, W. Wei, A. Liu, Y. Zhang and S. Liu,
Sens. Actuators B: Chem.,
2020,
313, 128031.

[36] C. Wei, X. Li, W. Xiang, Z. Yu and Q. Liu,
Sens. Actuators B: Chem.,
2020,
324, 128773.

[37] D. Chu, F. Li, X. Song, H. Ma, L. Tan, H. Pang, X. Wang, D. Guo and B. Xiao,
J. Colloid Interface Sci.,
2020,
568, 130–138.

[38] C. Chen, D. Xiong, M. Gu, C. Lu, F.-Y. Yi and X. Ma,
ACS Appl. Mater. Interfaces,
2020,
12(
31), 35365–35374.

[39] Y. Zhang, Y.-Q. Liu, Y. Bai, W. Chu and J. Sh,
Sens. Actuators B: Chem.,
2020,
309, 127779.

[40] H. Zhao, L. Tang, M. Zhou, K. Li, J. Hu, Y. Zhao and Z. Cai,
J. Mater. Chem. C,
2022,
10, 2988–2997.