3.1 Morphology and structural characterization
The surface morphology of the carbon felt was observed before and after modification.
Fig. 1 shows SEM images of the unmodified sample (
Fig. 1a); the TFPO sample (
Fig. 1b), which was treated under a mixed gas (21% O
2 + 79% N
2) in a tube furnace; the BFWA sample (
Fig. 1c), which was treated in a normal box furnace without air flow; and the TFWO sample (
Fig. 1d), which was treated under airless conditions in a tube furnace. As shown in
Fig. 1b, the microstructure of the TFPO sample was characterized by the roughest surface among the samples, whereas the BFWA sample exhibited moderate surface roughness. The surface of the TFWO sample was smooth, and the microgrooves were almost undetectable.
Fig. 1.
SEM images of bare (a), TFPO (b), BFWA (c), and TFWO (d) samples.

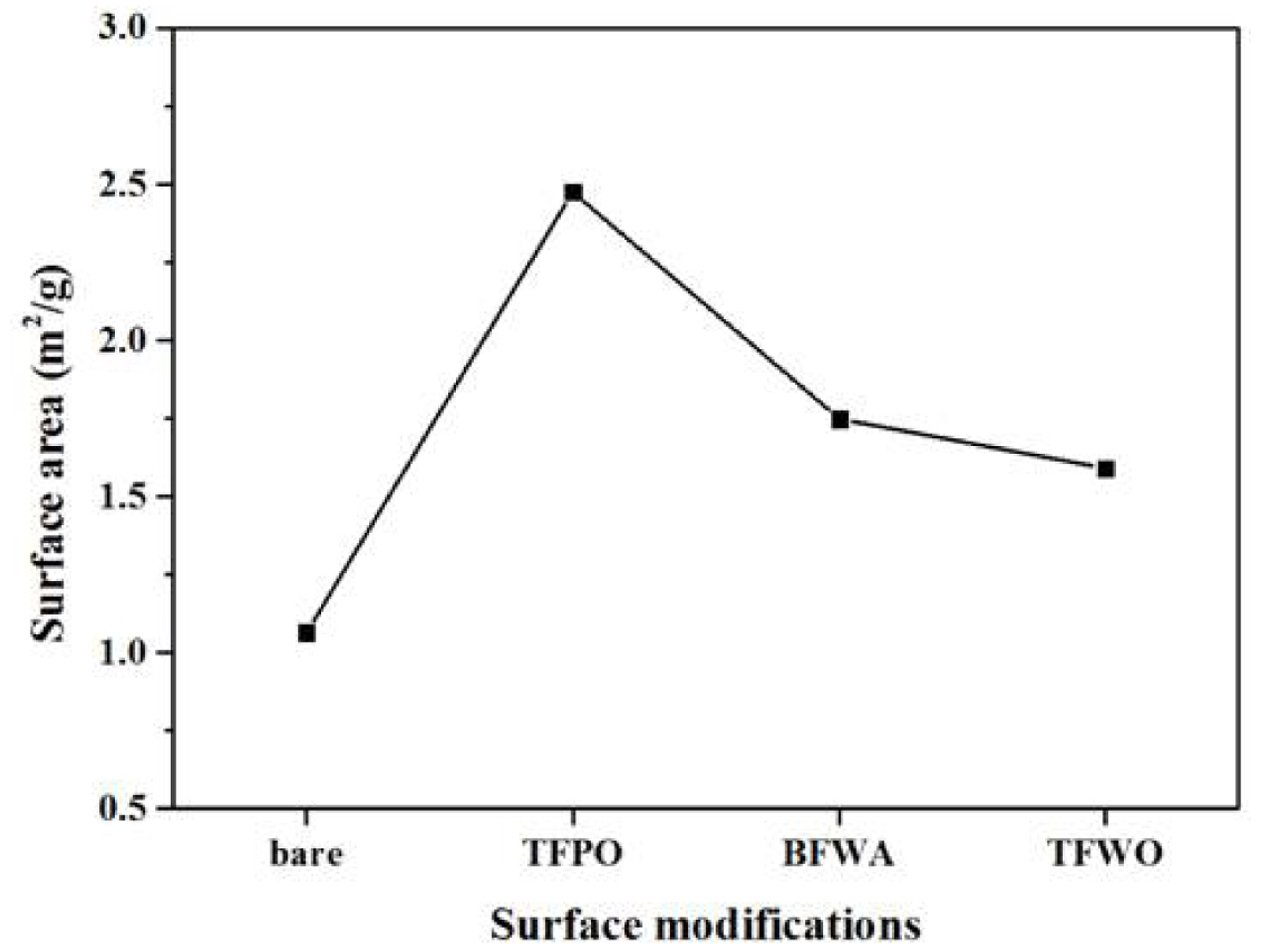
To further investigate the morphological changes, the specific surface area of the carbon felts was evaluated by BET analysis, as shown in
Fig. 2. The surface area of the carbon felt increased notably after treatment under mixed gas (232% vs. bare sample) and without airflow (164%). The TFWO carbon felt sample (treated under airless conditions) had the lowest surface area (149%) among the samples. Therefore, the surface roughness and area of the carbon felt were found to depend on the oxygen content of the mixed gas. The weight loss of the TFPO, BFWA, and TFWO carbon felt samples after surface treatment was measured as approximately 5.0%, 0.5%, and 2.0%, respectively. Notably, the surface of the carbon felt could be easily oxidized under a high oxygen content, leading to greater carbon loss from the surface via CO and/or CO
2 evolution.
Fig. 2.
Surface area of bare, TFPO, BFWA, and TFWO samples.
The carbon felt samples before and after thermal treatment were characterized by XRD analysis (
Fig. 3). All of the peaks could be indexed to the typical hexagonal (ICSD 98-005-3781) structure, indicating a pure single phase of graphite.
Fig. 3.
XRD analysis of bare, TFPO, BFWA, and TFWO samples.

The most intense diffraction peak for all the samples was that of the (002) plane, located at 26.4°. The two weak diffraction peaks at about 43.5° and 54.3°correspond to the (100) and (004) planes, respectively
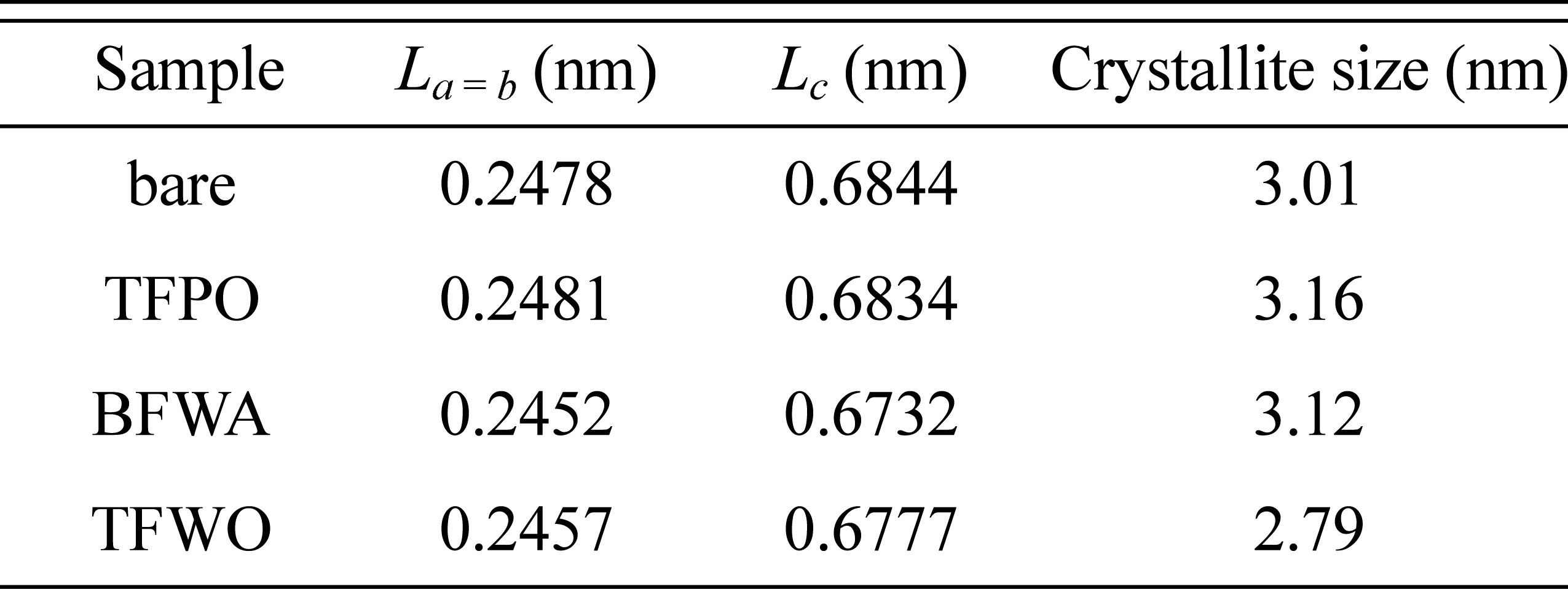
[14]. Compared with the XRD pattern of bare carbon felt, the XRD pattern of the TFWO sample showed a slight decrease in the diffraction intensity, and the peaks were shifted to the right. The lattice parameters and crystallite size of all the carbon felts were obtained by Rietveld refinement. As shown in
Table 1, the lattice parameters and crystallite size varied depending on the sample, where the dimensions of the
a =
b and
c axes of BFWA and TFWO were smaller than those of the TFPO sample. Furthermore, the crystallite size of the TFWO sample decreased more steeply than that of the other samples. These results demonstrate that more structural defects were introduced by modification under airless conditions.
Table 1.
Lattice parameters and crystallite size of carbon felt samples.
3.2 Surface chemistry
Figure 4 shows the C and O XPS spectra, which indicate the surface chemical state of the carbon felts before and after the modification processes. The O 1s peak could be deconvoluted into 3-5 peaks in the range of 528-539 eV, where the first peak, around 533-534 eV, is attributed to the phenolic (C-O and/or C-OH or C-O-C) groups, and the second peak, around 531-532 eV, can be assigned to the carboxylic and carbonyl (C=O) groups. The additional peak around 535-536 eV may be ascribed to absorbed water and some chemically absorbed oxygen
[15-
18]. The intensity of the O 1s-A peak of the TFPO and BFWA samples (which is related to the C-O group) increased, whereas that of the O 1s-B peak (C=O group) declined in comparison with that of the bare sample (
Fig. 4a-
c). It seems that the surface of the carbon felt changed because of CO and/or CO
2 evolution
[19]. The TFWO sample exhibited different behavior, as shown in
Fig. 4d. The intensity of the peaks related to the C-O group (O 1s-A) and the C=O group (O 1s-B) decreased, whereas a peak corresponding to the C-OH group (O 1s-C) appeared and became more intense than the peaks of the other samples. Moreover, the profile of the sample showed two additional peaks corresponding to chemisorbed H
2O and C-OH groups. The activity toward vanadium species reactions reportedly increases with increasing surface concentration of C-OH functional groups produced on the electrode surface
[14,
15,
17].
Fig. 4.
XPS analysis of bare (a), TFPO (b), BFWA (c), and TFWO (d) samples.

From the XPS spectra, it is possible to estimate the surface atomic ratio of C 1s versus O 1s, which provides more information on the functional groups formed by the surface modifications.
Fig. 5 shows the O 1s/C 1s atomic ratio for the bare, TFPO, BFWA, and TFWO samples, which had O 1s/C 1s ratios of 1.72, 9.18, 10.45, and 12.83, respectively. It is found that the carbon felt could be more easily functionalized with oxygen by airless treatment than by the other methods.
Fig. 5.
O 1s/C 1s atomic ratio of bare, TFPO, BFWA, and TFWO samples.

3.3 Electrochemical evaluations
Figure 6 shows cyclic voltammograms of the carbon felts in 0.3 M VOSO
4 + 0.5 M H
2SO
4 solution acquired at a scan rate of 5 mV·s
−1 over five cycles. The oxidation peak of the TFWO sample occurred at 1.0 V, whereas those of the other samples appeared at approximately 1.2 V, indicating the excellent electrochemical activity of the former. The oxidation and reduction peaks are associated with the VO
2+/VO
2+ couple, where the anodic peaks at 1.0-1.2 V are due to oxidation of VO
2+/VO
2+, whereas the cathodic peaks at 0.3-0.5 V correspond to reduction of VO
2+ to VO
2+ [15]. The redox peaks of the bare sample occurred at the lowest energy, even at the fifth cycle. Moreover, the TFWO sample presented a quasireversible and faster kinetic reaction than the TFPO and BFWA samples. The current peak for oxidation and reduction of the TFWO sample displayed a high degree of symmetry: The peak potential separation (D
Ep) decreased from 0.9 to 0.5 V.
Fig. 6.
Cyclic voltammetric curves of bare, TFPO, BFWA, and TFWO samples at fifth cycle.

Figure 7 shows the Nyquist plots of the carbon felts in 0.3 M VOSO
4 + 0.5 M H
2SO
4 solution at the open-circuit potential in the frequency range of 0.01 Hz to 10 kHz with an AC amplitude of 5 mV. All the Nyquist plots included a semicircular region at high frequency and a linear region at low frequency, suggesting that the electrochemical process was a combination of charge transfer and diffusion steps.
Fig. 7.
Nyquist plots of bare, TFPO, BFWA, and TFWO samples.

The high-frequency semi-arc arose from the charge transfer reaction at the electrolyte– electrode interface. The radius of the semi-arc reflects the charge transfer resistance. The low-frequency linear region can be attributed to the diffusion processes associated with the diffusion of VO
2+/VO
2+ in the pore channel of the electrode
[12,
20]. The bare sample had the largest semi-arc radius, indicating that it had the largest charge transfer resistance for VO
2+/VO
2+. The magnitude of the high-frequency semi-arc decreased with increasing oxygen groups on the surface of the carbon felts in moving from the TFPO to the TFWO sample. This result is consistent with the CV results. Thus, both the EIS and CV data indicate that the reaction of the VO
2+/VO
2+ redox couple was greatly enhanced on the TFWO sample.
Figure 8 shows the galvanostatic charge-discharge profiles at the first cycle of VRFB single cells with the carbon felts before and after modification. A comparison of the charge–discharge curves of the VRFB cells employing the bare, TFPO, BFWA, and TFWO electrodes showed that the VRFB with the bare electrode had a higher charge voltage plateau and a lower discharge voltage plateau than the cells with the other electrodes at the same current density. Moreover, the TFWO electrode exhibited a lower charge voltage plateau and a higher discharge voltage plateau than the other electrodes; thus, the voltage efficiency of the VRFB with the TFWO electrode was higher than that of the VRFB with the other electrodes, suggesting that the oxygen groups on the TFWO electrode reduced the polarization resistance during the charge–discharge process.
Fig. 8.
Galvanostatic charge-discharge profiles of VRFB cells employing bare, TFPO, BFWA, and TFWO samples.
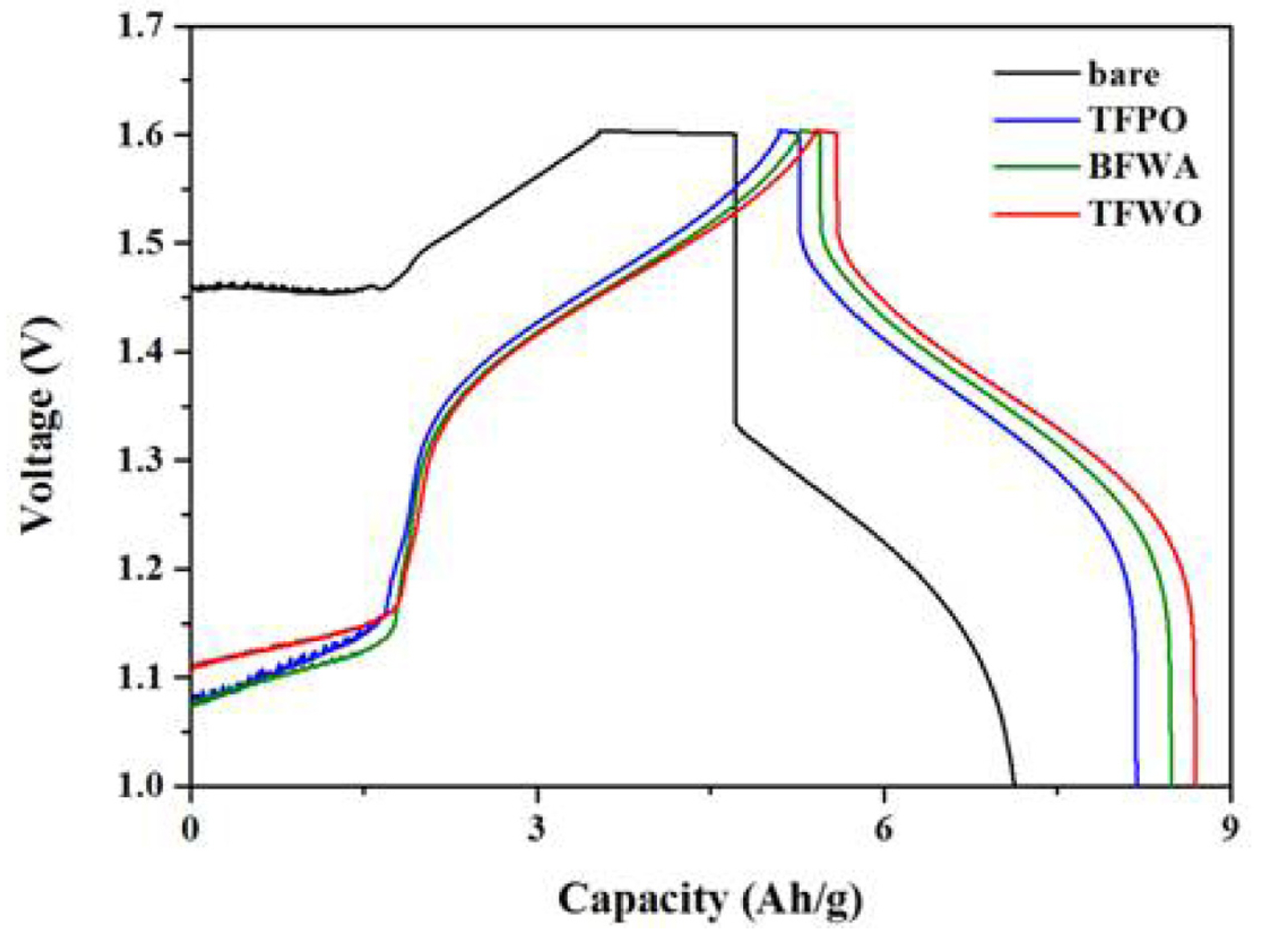
Figure 9 shows the charge-discharge and efficiency profiles of the TFWO electrode during cycling. As shown in
Fig. 9a, a large, irreversible charge curve was obtained at the first cycle, which is common to all the samples owing to the oxidation of V
3+ to VO
2+/VO
2+ during the initial charging process
[15]. The discharge capacities at the first and second cycles did not differ greatly, unlike the case for the charge process. The Coulombic efficiency (CE), voltage efficiency (VE), and energy efficiency (EE) of the TFWO electrode with cycling are summarized in
Fig. 9b. The CE, VE, and EE of the TFWO electrode at the fifth cycle were 89%, 92%, and 82%, respectively, and the EE values of all the samples except for the bare sample were almost the same, indicating their high performance compared with that previously reported for carbon felts
[8,
19].
Fig. 9.
Galvanostatic charge-discharge profile (a) and efficiency (b) of VRFB cells employing TFWO sample.

These results reveal that although its surface area was relatively small, the TFWO electrode exhibited good electrochemical performance; the essential factor affecting the electrochemical activity of the carbon felt electrodes is the oxygen functional groups, which can, along with the large surface area, provide more redox reaction sites for vanadium ions at a constant flow rate. Therefore, the phenolic group (C-O and/or C-OH or C-O-C) is more effective for increasing the energy efficiency of VRFB cells than the carboxylic or carbonyl groups (C=O) from the thermodynamic point of view.


















