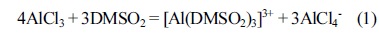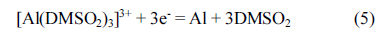1. Introduction
Aluminum is used in many applications including structural, anti-corrosive, electronic, and optical materials. Its widespread use is due to its excellent properties, which include electrical conductivity, heat/light reflectivity, high thermal conductivity, and corrosion resistance. These properties can also be imparted to other substrates when aluminum films form on their surfaces. Smelting of Al has been industrialized by the Hall-H├®roult process: Al is electrowon from alumina-dissolved cryolite molten salt at 1273 K [1]. As its low redox potential (ŌłÆ1.68 V vs. SHE) does not permit its electrowinning in aqueous solutions, recent attempts to fabricate Al films have employed mechanical and chemical methods such as hot dipping [2], vapor phase deposition [3,4], thermal spraying [5,6], and electrodeposition [7-9]. Of these methods, the electrodeposition of Al boasts many advantages, including the ability to coat complex shapes evenly, relatively high deposition rates, and easily controlled coating thickness. Unlike many conventional metallic coatings, those consisting of Al films cannot be electrodeposited from aqueous solutions [10]. However, Al-coating has been achieved using non-aqueous media such as organic solvents, molten salts, and ionic liquids [11-19]. Of these options, the organic solvent dimethylsulfone (DMSO2, (CH3)2SO2) is advantageous, as it is inexpensive compared to ionic liquids and more stable than ethers and aromatic hydrocarbons [20-28].
Raman analyses [26] and 27Al NMR [29] of the AlCl3-DMSO2 mixtures indicate that two main soluble Al complex ions, namely AlCl 4ŌłÆ and [Al(DMSO2)3] 3+, formed according to the following reaction:
Electrodeposition of Al can occur from the solvated cation [Al(DMSO2)3]3+, whereas the reduction of AlCl4- is not observed within the electrochemical window of the electrolytes. It has been demonstrated that dense, uniform Al coatings with a high corrosion resistance can be electrodeposited from AlCl3-DMSO2 baths at 383 K [30-32].
In this study, we investigated the electrochemical behavior of Al using DMSO2 as a solvent. The mechanism of Al electrodeposition was also determined by NMR spectroscopy. The effect of changing [Al(DMSO2)3]3+ concentration on Al electrodeposition was also investigated. Cyclic voltammetry (CV) was used to examine the deposition process. Scanning electron microscopy (SEM) and X-ray diffraction (XRD) analyses were performed to determine the morphology and phase composition of the Al films, respectively.
2. Experimental
2.1 Electrode preparation
Al, Cu, and Pt plates and Pt coil were used as electrodes. The Al and Cu plates were wet-polished with 400 emery paper. The Pt plate and coil were immersed in hydrochloric acid to clean their surfaces and then washed with distilled water. Electrodes washed with acetone for 20 min before being used for CV. Cu and Pt plates were coated with Nitoflon insulating tape to form a working electrode with an area of 1 cm2.
2.2 Bath preparation
DMSO2 (Tokyo Chemical Industry, Japan, 99 mass%) and anhydrous aluminum chloride grains (AlCl3, Nacalai Tesque, crystallized, 98 mass%) were used as the solvent and Al source, respectively. The amount of DMSO2 used in each experiment corresponded to 0.74 mole. DMSO2 was pretreated to remove water by holding the powder at 333 K for at least 48 h in a vacuum. To prepare the experimental baths, DMSO2 powder was heated in a 100-cm3 glass beaker at 403 K. AlCl3 powder was then carefully added to the melted DMSO2. Bath preparation was conducted in an Ar-filled glovebox.
2.3 Electrochemical measurements and characterization of deposits
Electrochemical properties were measured in the Ar-filled glovebox with a potentiostat/galvanostat (BioLogic, SP 150). Cyclic voltammograms were between ŌłÆ2 V and 2 V vs. Al/Al3+ at a scan rate of 100 mVs-1. Galvanostatic electrodeposition was performed at a current density of 40 mAcm-2. The temperature was maintained at 403 K using a hotplate. Cu (Nilaco, >99% purity) and Al (Nilaco, >99.99% purity) plates were used as the working and counter electrodes, respectively. An Al rod was immersed in the bath as the reference electrode (Al/Al3+). A portion of the copper plate was masked with insulating tape, leaving an exposed surface area of 1.0 cm2. The electrolyte was agitated at 150 rpm during the experiment using a magnetic stirring device at the bottom of the beaker.
2.4 Bath characterization
Spectra for Al3+ complexes in AlCl3ŌĆōDMSO2 solution were recorded using UV-visible spectroscopy (UV-vis spectrophotometer, JASCO model V-650, Japan). Absorbance measurements were performed with a quartz cell with a path length of 1 cm. The amount of DMSO2 was fixed at 0.74 mole, while the concentration of AlCl3 varied from 6.2 to 28.3 mol% AlCl3 at 403 K. The absorbance of each solution was measured at 255 and 600 nm.
In this study, the mol% of AlCl3 was calculated using Eq. (2):
27Al NMR spectroscopy was performed at 130.3 MHz using a 500 MHz NMR spectrometer (Agilent Technology). All chemical shifts were referenced to D2O containing 1.5 M Al(NO3)3, which was used as an external reference. Samples were placed in 10 mm NMR tubes with a 5-mm coaxial tube filled with DMSO-d6 as a lock solvent. Spectra were gathered at 403 K, as in the electrochemical measurements.
Ru K-edge XAFS was measured in the transmission mode at 20 K at the NW10A station with a Si(311) double-crystal monochromator. The energy and current of electrons in the storage ring were 6.5 GeV and 60 mA, respectively. Ionization chambers filled with pure Ar and pure Kr were used to monitor the incident and transmitted X-rays, respectively.
2.5 Characterization of electrodeposits
After electrolysis, deposits were washed with a AlCl3-DMSO2 solution and vacuum-dried prior to characterization. Crystal orientation and surface morphology of the deposits were characterized by X-ray diffraction (XRD; Rigaku Ultima IV; 40 kV, ŌłÆ30 mA, 0.4┬░ min-1) and field emission scanning electron microscopy (FE-SEM; NeoScope JCM-5000, JEOL). Chemical characterization and elemental analysis for Al, S, and Cl was performed using energy dispersive X-ray spectroscopy (EDX; EDAX Genesis 2000). In addition, X-ray photoelectron spectroscopy (XPS; Shimadzu, ESCA-3300) was carried out to examine the chemical composition. XPS was performed using a Mg-K╬▒ X-ray source with an Ar+ etching rate of approximately 0.5 nm/min, which was determined using SiO2 as a standard film. The XPS spectra were calibrated relative to the C 1s peak at 285.0 eV.
3. Results and Discussion
3.1 Cyclic voltammetry with different concentration ratios of AlCl3/DMSO2
Cyclic voltammetry was carried out from ŌłÆ2 to 2 V at a rate of 100 mVs-1 in a DMSO2 bath containing 6.2ŌłÆ28.3 mol% AlCl3 at 403 K. The electrode potential was scanned from the open-circuit potential near 0 V in the negative direction, followed by scanning in the positive direction and then cycling. As shown in Eq. 3-5, AlCl3 dissociates in DMSO2 to produce Al3+ and Al3+ and DMSO2 and form the ionic complex [Al(DMSO2)3]3+. Sweeping in the cathodic direction causes metallic Al to electrodeposit from [Al(DMSO2)3]3+. When sweeping in the anodic direction, the precipitated Al is redissolved, resulting in the formation of Al3+ via a reversible reaction [24]. Fig. 1 shows voltammograms recorded after 20 cycles in a DMSO2 bath. The shapes of the curves did not change after the third cycle. The rise in the cathodic current close to ŌłÆ0.5 V corresponds to precipitation of Al, while the increase in the anodic current indicates dissolution of Al.
Fig.┬Ā1.
Cyclic voltammograms of Pt electrode in AlCl3-DMSO2 baths at 403 K with AlCl3 concentrations : (a) 0 (pure DMSO2), (b) 6.2, (c) 11.6, (d) 23.3, and (e) 28.3 mol%.

The reduction potential for Al electroplating varied with the concentration of [Al(DMSO2)3]3+. Current density depends on the Al ion contents. Thus, this potential was used as a reference.
UV-visible spectrophotometry was performed to further investigate the reversible reaction. The absorption spectra of various concentrations of AlCl3 in DMSO2 are shown in Fig. 2. DMSO2 exhibits a strong absorption band at 392 nm. Spectra following the addition of Al3+ to the solution show a decrease in the absorption peak at 392 nm, while a band centered at 456 nm increases with up to 23.3 mol% AlCl3. For concentrations of AlCl3 greater than 6.2 mol%, a second isosbestic point is observed at 470 nm, accompanied by the increase in a new band at 475 nm, which is characteristic of the formation of a complex ion. As all spectra cross at least one of the two isosbestic points, the simultaneous presence of the free ion and the two main complex ions is not observed. This indicates that Al3+ is cleanly converted into the [Al(DMSO2)3]3+ complex. Our results are in agreement with the data presented by Cornard et al. for quercetin [33].
Fig.┬Ā2.
UV-visible spectra of DMSO2-Al(III) complexes with various AlCl3 concentrations at 403 K: (a) 0 (pure DMSO2), (b) 6.2, (c) 11.6, (d) 23.3, and (e) 28.3 mol%.
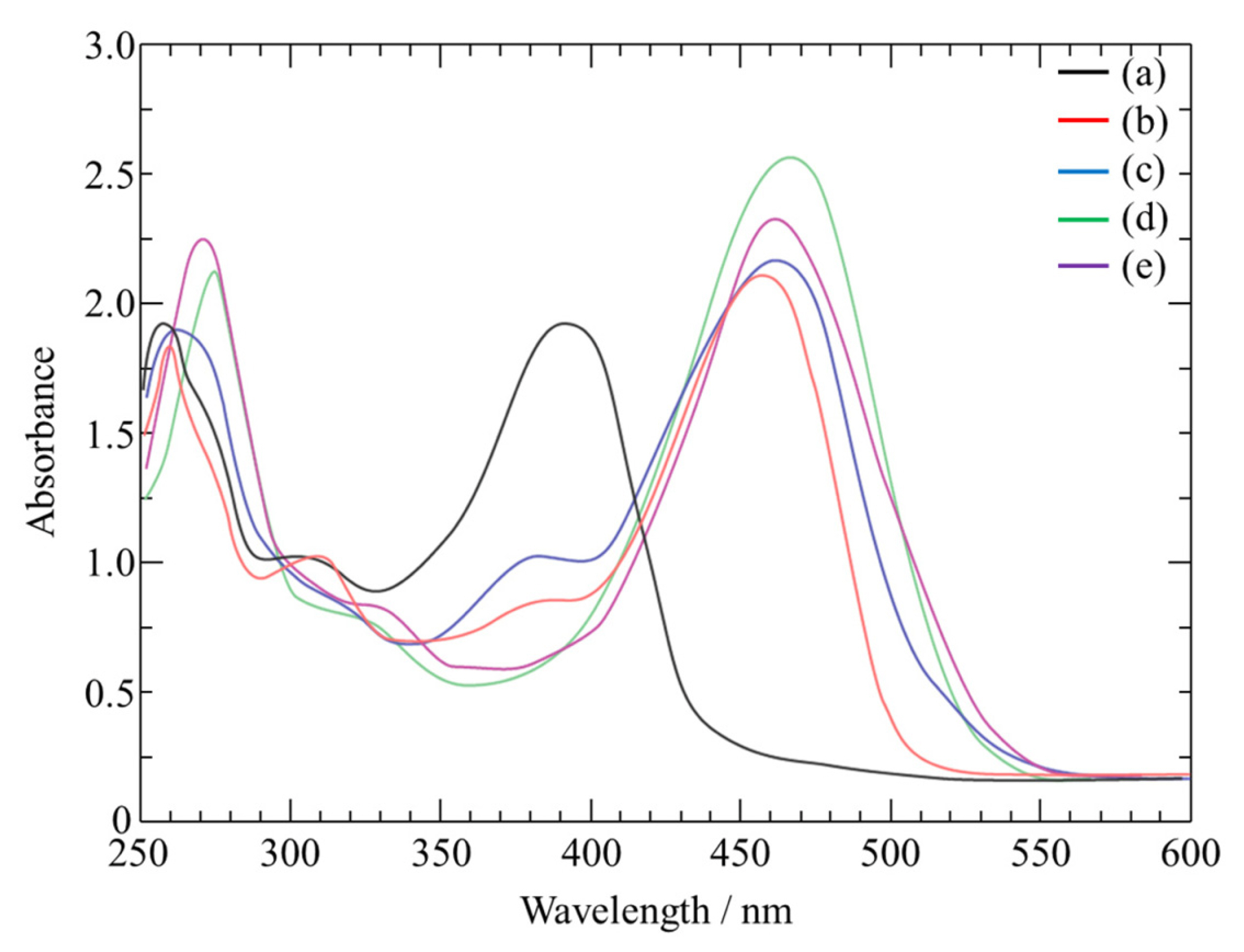
Fig. 1 also shows that the reduction potential of Al3+ in ionic liquids decreases in the order 23.3 mol% AlCl3 > 28.3 mol% AlCl3 > 11.6 mol% AlCl3 > 6.2 mol% AlCl3. Similar negative shifts have been observed when Al/Al3+ was directly immersed in baths [34].
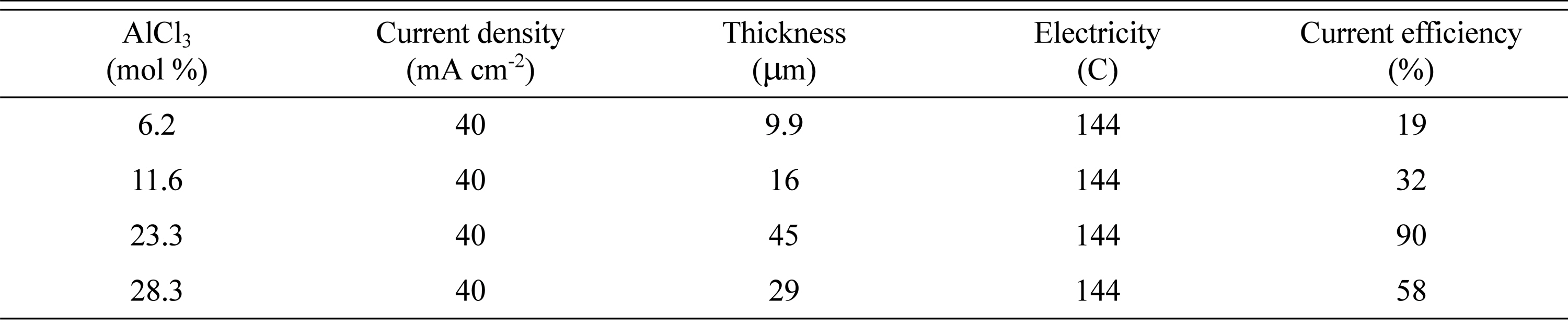
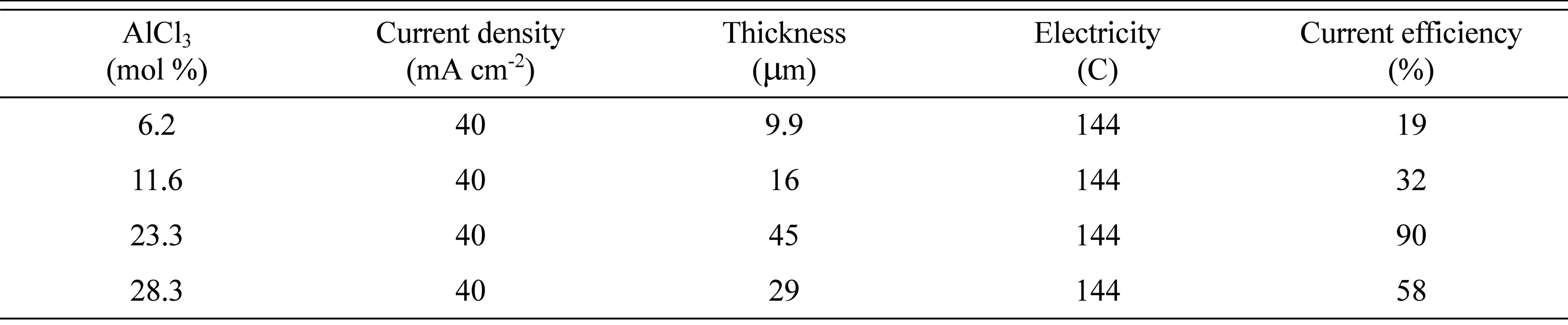
Our observations for the effect of the mole percent of AlCl3/DMSO2 are similar to data presented by Jiang et al. [24]. They reported that electrolytes with AlCl3/DMSO2 ratios of 0.2:1 and 0.3:1 have wider ranges of operating current densities. To confirm the operating range of the current density, experiments were performed from 20 mAcm-2 to 80 mAcm-2 at 403 K. The results are shown in Table 1. In terms of current efficiency, a current density of 20 mAcm-2 was the best, but resulted in a black surface on electrodeposited Al surface. Thus, we selected a current density of 40 mAcm-2, which provided a clean film with a high current efficiency. However, further investigation was needed to determine the cause of the wider operating current density. To that end, the influence of the AlCl3 concentration on the ion species in the bath and the electrodeposition of Al was examined using NMR.
3.2 NMR measurements using various AlCl3/DMSO2 mixture
Cyclic voltammetry measurements indicated that the composition of ions in the AlCl3-DMSO2 mixture changed with AlCl3 concentration. Thus, the relationship between the concentration of AlCl3 and the ionic species observed in the bath and on the electrodeposition of Al was investigated by NMR spectroscopy. 27Al NMR spectroscopy was used, as this technique allows the environment of Al atoms to be unambiguously determined in most cases. As previously reported, octahedral Al is found at chemical shifts of around 0 ppm in the 27Al NMR spectra, while tetrahedral Al is found around 100 ppm [35-37].
27Al NMR spectra obtained for the four mixtures are shown in Fig. 3. The results showed that AlCl4- and [Al(DMSO2)3]3+ species were formed in DMSO2 baths containing 6.2-28.3 mol% AlCl3. The sulfone-solvated Al3+ species is generated along with the formation of AlCl4- [29]. Since the source of Cl- is AlCl3, asymmetric cleavage of AlCl3 forms AlCl4ŌĆō and [Al(DMSO2)3]3+. We confirmed the structure of the complex ion by conducting XAFS analysis of the AlCl3-DMSO2 bath in more detail. Fig. 4 shows the K-edge XAFS analysis for S in DMSO2 and in the AlCl3-DMSO2 bath. For only DMSO2, the peak at 2479 eV is larger in the AlCl3-DMSO2 mixture compared to what is observed for DMSO2 alone. This is because the O of DMSO2 is coordinated to Al3+, the electrons of O are attracted to Al3+, and the electrons of S are attracted to O. Therefore, O is thought to have caused some new bonding with AlCl3.
Fig.┬Ā3.
27Al NMR spectra of AlCl3-DMSO2 baths containing AlCl3 concentrations: (a) 6.2, (b) 11.6, (c) 23.3, and (d) 28.3 mol%.
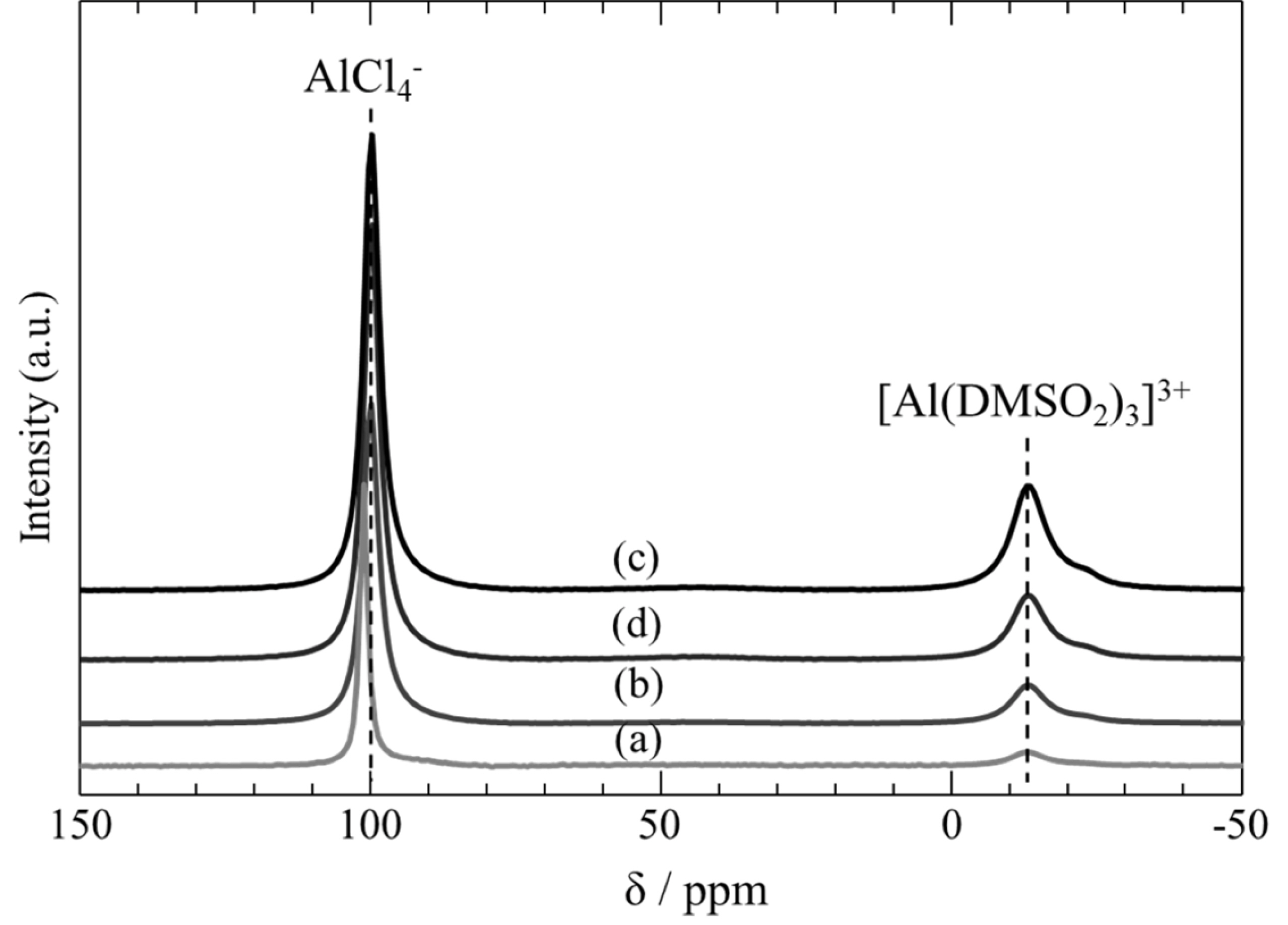
Fig. 5 shows the results of K-edge XAFS analysis of Cl in an AlCl3-DMSO2 bath and in AlCl3 alone. Peaks at 2824 eV and 2830 eV were observed for AlCl3 alone, whereas in the AlCl3-DMSO2 bath, one large peak at 2826 eV was observed. We suggest that the large peak at 2826 eV obscures those at 2824 and 2830 eV, giving rise to a single large peak for the mixture. The peak energy value depends on bond length rather than atom to which Cl is bonded. Therefore, Cl of AlCl3 is unlikely to form a direct bond with DMSO2, and it is thought that Al, which forms a new bond with DMSO2, forms a complex ion [Al(DMSO2)3]3+.
27Al NMR of AlCl3-DMSO2 confirmed the presence of AlCl4- (110 ppm) and [Al(DMSO2)3]3+ (ŌłÆ10 ppm) at 403 K. These chemical shifts were assigned based on the work of Legrand et al. [29]. AlCl4- is electrochemically inactive, while Al2Cl7- is active in the case of room-temperature ionic liquids. Since the anionic species in the DMSO2 baths containing 6.2ŌłÆ28.3 mol% AlCl3 are AlCl4-, we conclude that [Al(DMSO2)3]3+ is electrochemically active. The peak for [Al(DMSO2)3]3+ intensified as the concentration of AlCl3 increased.
Fig. 6 shows the calculated concentrations of each ion species in 6.2ŌłÆ28.3 mol% AlCl3-DMSO2 baths, as determined from NMR spectra. As the concentration of AlCl3 increased, the observed concentration of [Al(DMSO2)3]3+ also increased. However, in the 28.3 mol% AlCl3 bath, the calculated concentration decreased slightly (a smaller peak was observed). To calculate the absolute amounts of [Al(DMSO2)3]3+ present in each bath, the molar amount of AlCl3 was multiplied by the calculated [Al(DMSO2)3]3+ concentration ratio (Fig. 7). The absolute amount of [Al(DMSO2)3]3+ was calculated using Eq. (7).
Fig.┬Ā6.
Concentrations of ion species in DMSO2 baths with various AlCl3 concentrations: (a) 6.2, (b) 11.6, (c) 23.3, and (d) 28.3 mol% as measured by 27Al NMR.

Fig.┬Ā7.
Concentration of [Al(DMSO2)3]3+ as a function of the AlCl3 concentration in AlCl3/DMSO2 baths.

The concentration of [Al(DMSO2)3]3+ increased with additional AlCl3; however, it remained almost unchanged when the amount of AlCl3 was increased from 23.3 to 28.3 mol%, indicating that almost no additional [Al(DMSO2)3]3+ forms in baths with AlCl3 concentrations of greater than 23.3 mol%. When combined with the observation that AlCl4ŌĆō concentration increases with larger amounts of AlCl3, this finding indicates that the decrease in the current observed in the bath containing 28.3 mol% AlCl3 results from a lower [Al(DMSO2)3]3+ concentration that leads to reduced Al electrodeposition. Furthermore, increased viscosity of the bath may compromise the deposition. The results show that an AlCl3 concentration of approximately 23.3 mol% is optimal for the electrodeposition of Al from AlCl3-DMSO2 baths.
3.3 Influence of AlCl3 concentration on the plated film
Galvanostatic electrodepositions were carried out to confirm the properties of the deposited Al and the effect of the concentration of AlCl3 in the DMSO2 baths. Fig. 8 shows photographs of the Al deposits obtained from AlCl3-DMSO2 baths held at a constant current density of 40 mAcm-2 for 60 min at 403 K. Cu and Al plates were used as the cathode and anode, respectively. As shown, grayish-white Al plating was observed with high concentrations of AlCl3 (23.3 and 28.3 mol%).
Fig.┬Ā8.
Surface photographs of Al films electrodeposited on Cu substrates at 40 mAcm-2 for 1 h in AlCl3/DMSO2 baths at 403 K with AlCl3 concentrations: (a) 6.2, (b) 11.6, (c) 23.3, and (d) 28.3 mol%.

XRD measurements shown in Fig. 9 confirmed that the deposits consisted of crystalline Al metal without trace impurities such as Al2O3. The height of the Cu substrate peak was considered to be constant for normalization purposes (Cu (200) peak at 51┬░). In each pattern, diffraction peaks relating to Al (111), (200), (220), and (311) were observed. The Al (200) reflection was notably higher than the Al (111) reflection, indicating Al (200) as the preferential orientation under the experimental conditions. This result agrees with PangarovŌĆÖs theory [38], which predicts that fcc metals electrodeposited at low overpotentials are preferentially orientated in the (111) direction and that this preference changes to (200) with increasing overpotential.
The preferential crystal orientation of Al differed among samples. Al formed in baths containing 23.3 mol% or 28.3 mol% AlCl3 was oriented primarily in the (200) plane (Figs. 9 (c) and (d)), while the Al(200) peaks observed in Figs. 9 (a) and (b) are smaller. Therefore, it can be deduced that the Al layer formed using either 23.3 mol% AlCl3 or 28.3 mol% AlCl3 was much thicker than that formed using 6.2 mol% AlCl3 or 11.6 mol % AlCl3 baths.
Fig.┬Ā9.
XRD pattern of deposit obtained on a Cu substrate galvanostatic electrodeposition in AlCl3/DMSO2 baths at 403 K with AlCl3 concentrations: (a) 6.2, (b) 11.6, (c) 23.3, and (d) 28.3 mol%.

Fig. 10 shows the effect of the AlCl3 concentration on the microstructure of the electroplated Al surface formed over 60 min in a DMSO2 bath containing 6.2ŌłÆ28.3 mol% AlCl3 at a constant current density of 40 mAcm-2 at 403 K. Significant cracking was observed on the surfaces of the Al films made in the baths containing 6.2 mol% and 11.6 mol% AlCl3 (Fig. 10 (a) and (b)). A small number of cracks can also be seen in the sample obtained using 23.3 mol% AlCl3, though there are regions that appear entirely crack-free when compared with those of the other specimens. Small white stains are observed, particularly when 23.3 mol% AlCl3 was used, but overall this sample had the cleanest surface. In addition, the deposits were almost flat, and the thickness was 45 ┬Ąm, in good agreement with the calculated values assuming 90% current efficiency (45 ┬Ąm), indicating that the coatings were compact. The current efficiency according to the concentration of AlCl3 is shown in Table 2.
EDX analysis for the area shown in Fig. 10 indicated that 23.3 mol% AlCl3 shows the highest purity of Al in the experimental samples. The results are shown in Table 3. S and Cl were detected as the main impurities. Inclusion of S and Cl is characteristic of coatings electrodeposited from AlCl3-DMSO2 electrolytes [39]. Fe is detected because it is present in trace amounts in AlCl3.
Fig.┬Ā10.
Surface SEM image of Al film electrodeposited on Cu substrate under 40 mAcm-2 for 1 h in AlCl3/DMSO2 baths at 403 K with AlCl3 concentrations: (a) 6.2, (b) 11.6, (c) 23.3, and (d) 28.3 mol%.

XPS analysis was performed to further investigate the impurities. A representative set of XPS spectra of the Cl 2p, S 2p, and S 2s regions is shown in Fig. 11. A signal was detected at ~199 eV, which compares to Cl 2p3/2 binding energies for many chlorides. This result confirms that Cl was not a surface contaminant but rather was incorporated in the Al coating during electrodeposition. As for S, an S 2p signal was detected at ~169.3 eV from the surface of Al electrodeposition. The peak position is close to the reported value for DMSO2 (169.2 eV) [40], thus confirming the presence of DMSO2 on the surface of the Al coating. If sulfides or elemental sulfur were present, signals would appear in the range of 160-165 eV [41]. This region is obscured by the Al satellite peak; thus, the S 2s region was analyzed to clarify whether S is present in other forms. In the S 2s region, a signal was observed at 233.2 eV from the surface of the Al coating. Another signal was found at a lower binding energy (about 226 eV), which is ascribed to a sulfide in the coating, consistent with what is reported in the literature [42,43]. XPS analysis showed no peak for Fe.
4. Conclusions
Al was electrodeposited from AlCl3-DMSO2 baths containing 6.2ŌłÆ28.3 mol% AlCl3. CV indicated that electroplating is a reversible process, while 27Al NMR spectroscopy confirmed that while both [Al(DMSO2)3]3+ and AlCl4- ions are present in the baths, only the cation produces electroplated Al. The concentration of [Al(DMSO2)3]3+ increased as more AlCl3 was added, plateauing at an AlCl3 concentration of 23.3 mol%. This result indicates that little additional [Al(DMSO2)3]3+ formed with AlCl3 concentrations greater than 23.3 mol%; instead, AlCl4ŌĆō formed. Thus, electroplating of Al should be performed in AlCl3-DMSO2 baths with AlCl3 concentrations of approximately 23.3 mol%.