|
 |
- Search
| J. Electrochem. Sci. Technol > Volume 13(2); 2022 > Article |
|
Abstract
In this study, electrochemical treatment of urban wastewater with electrical conductivity of 1000 μS cm−1 and chemical oxygen demand of 250 mg L−1 was investigated using the variables of initial pH value, current density and flow rate. Electrocoagulation was used, in which aluminum and stainless steel were selected, as the electrochemical treatment process. The electrocoagulation process was operated in continuous mode. The data obtained in experimental studies show that the best COD removal efficiency occurred in experiments where the initial pH value was 6. The increase in current density from 5 A to 15 A decreased the removal efficiency from 79 to 67%. The increase in flow rate under constant current density also reduced the efficiency of removal as expected. In experiments in which current density and flow rate were examined together, the increase in flow rate allowed the application of higher current densities. This situation led to considerable reductions in energy consumption values, even if the COD removal efficiency did not significantly increase. The high COD removal obtained with the use of high flow rate and high current density indicates that the electrocoagulation process can be used for high flow rate municipal wastewater treatment.
Untreated discharge of urban wastewater into the aqueous environment poses potential risks for humans, animals and ecosystems due to the content of chemical and microbiological pollutants. Among the targets and priorities for 2030 in the United Nations Sustainable Development Goals (2015) are the improvement of water quality by reducing the discharge of hazardous materials and the proportion of untreated wastewater, and safe reuse of water. Today, urban wastewaters are discharged into the receiving environment after they have been treated in wastewater treatment plants. However, water scarcity caused by the increase in population and agricultural and industrial activities has led to the development of methods for obtaining safe treated water for use in irrigation, aquifer feeding and drinking water production [1].
Wastewater treatment plants consist of primary, secondary, and sometimes advanced treatment processes with different biological, physical, and chemical technologies. Nowadays, domestic wastewater treatment plants around the world use many treatment processes, including conventional activated sludge treatment, anaerobic-anoxic-oxic (A2/O), anaerobic-oxic (A/O), sequencing batch reactor (SBR) and oxidation ditch. The removal efficiency of a wastewater treatment plant depends on the process used and also depends on the capacity of the wastewater treatment plant [2].
Electrocoagulation (EC) has been successfully employed for treatment of various wastewaters such as vegetable oil refinery wastewater [3], surfactant [4,5], food processing [6], semiconductor [7], printing [8], mining [9], heavy metal [10,11], tannery [12], boron [13], dairy [14], poultry [15], slaughterhouse [6], pulp and paper mill [16], dye effluent [17] and arsenic contaminated drinking water [18]. EC involves in situ generation of coagulants by electrolytic oxidation as a result of applying direct current with an appropriate anode (for example, iron and aluminum) [19]. The metal ions generated during EC form charged metal hydroxide ions and neutral metal hydroxides The low solubility of these hydroxides, mainly at pH values in the range of 6.0–7.0, generates flocs in the wastewater, and pollutants incorporated into these flocs are removed. The EC process removes pollutants principally by coagulation, adsorption, precipitation and flotation [20]. The electrocoagulation process is influenced by many factors such as the contact time, current density, initial pH of wastewater and distance between the electrodes.
Classical activated sludge systems are generally used in the treatment of domestic wastewater. However, in addition to the removal of solid and organic matter, the necessity of removing other inorganic materials during the treatment process led to the necessity to develop different treatment methods. Electrocoagulation is one of these methods.
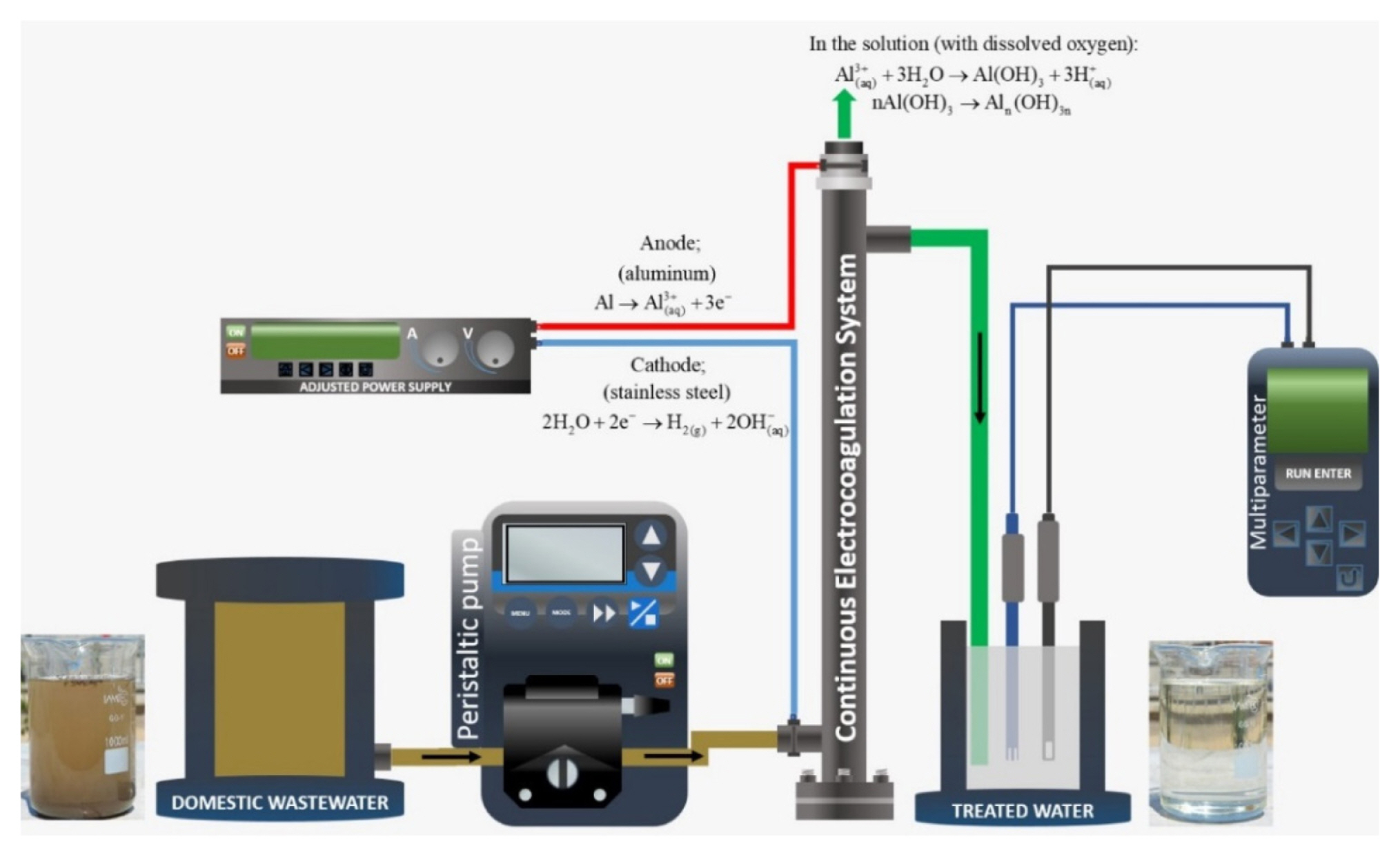
In this process, metal ions are produced due to the application of direct current through the metal electrode and these metal ions form metal hydroxides in the electrochemical cell. In electrocoagulation processes, the coagulating species are produced in situ and the process generally consists of three successive stages: (i) formation of coagulants by electrolytic oxidation of the sacrificial electrode, (ii) destabilization of the contaminants, particulate suspension and breaking of emulsion, and (iii) aggregation of the destabilized phases to form flocs [21].
If aluminum is used as the anode electrode, the reactions given in equations 1–4 occur.
The electrocoagulation technique has some advantages such as shorter reaction time, rapid sedimentation of electro-generated flocs, less sludge production, absence or less additional chemicals, simple equipment and easy operation. The objective of this study is to optimize urban wastewater removal by electrocoagulation. Aluminum was used as anode material and stainless steel was used as cathode in the monopolar configuration. The effects of initial pH of wastewater, current density, reaction time and flow rate on the COD removal efficiency and energy consumption were investigated.
In the study, domestic wastewaters with weak characteristics were used (Metcalf and Eddy, 2002). COD was 250 mg L−1, while BOD5 was 175 mg L−1, suspended solids (SS) were 80–100 mg L−1 and 1000–1050 μS cm−1 of electrical conductivity. The experiments were carried out at 15±3°C. In the continuously operated electro-coagulation system, a tubular reactor with total volume of 500 mL was used. The reactor consisted of a telescopic stainless-steel cathode with inner diameter of 60 mm and aluminum anode with outer diameter of 50 mm. A Chroma brand digitally controlled direct-current power supply (62024P–40–120 model 0–40V, 1–120A) was used to supply the required power. A WTW brand multi-meter was used to adjust pH, conductivity and temperature of the wastewater at the beginning of the reaction and to read these values instantly during the reaction. Total surface area of the plates was 1400 cm2 and the distance between the electrodes was 5 mm. In the experiments, the effect of wastewater flow rate, initial pH of wastewater and current density were examined as wastewater treatment parameters. The experimental system is given in Fig. 1.
Experimental conditions and work intervals are shown in the table below (Table 1).
Chemical oxygen demand (COD) analyses were used to examine the parameters affecting the electrocoagulation process for domestic wastewater treatment. COD analyses were conducted according to the closed system (reflux) method as specified in the standard methods [22]. The following equations were used for calculation of the experimental data [23, 24];
Calculation of treatment efficiencies,
C0 initial pollutant concentration (mg L−1) and Ct the concentration of the pollutant remaining in the wastewater at time t (mg L−1).
Calculation of energy consumptions
W, the value of energy consumption (kW-h m−3), I applied current density (A), V potential difference in the system (volt), t reaction time (minute), and θ total wastewater volume (m3).
Effects of the wastewater initial pH value, flow rate and current density on COD removal efficiency were examined in the system.
pH is a significant operating parameter that influences performance in electrochemical processes. The formation and stability of aluminum hydroxyls during electrolysis, as well as solubility of their products, are primarily dependent on the initial pH of wastewater. Therefore, initial wastewater pH influences COD removal in both acid and basic solutions [25]. Accordingly, the initial pH values of the wastewater were adjusted to 5, 6, 7 and the natural pH value (7.8) to examine effect of pH on the removal efficiency, and the experiments were carried out. During the experiment, the pH value of the wastewater was not interfered with from the outside. The pH of the wastewater changed as a result of electrochemical reactions.
To investigate the effects of these parameters flow rates of 10, 25, 50, 75 and 100 mL min−1 were used with current density of 10 A. At all pH values, the effluent water pH values tended to fall with the increase in flow rate, as seen in Table 2.
In addition, none of the initial pH values exceeded the final pH 9.5. This shows that the content of wastewater has buffering properties around pH=9.5. Under high pH conditions, Ca+2 and Mg+2 in wastewater may precipitate together with Al(OH)3 in the form of hydroxide, causing a pH drop. It is thought that this situation is caused by the total hardness of approximately 150 mg L−1 CaCO3 in the wastewater. Ca+2 ions, which have a large share of the total hardness, tend to precipitate in the form of CaCO3 due to the following reaction around pH 9–9.5.
The withdrawal of OH-ions from the solution for CaCO3 precipitation caused the pH value to remain at a certain level. This situation has a similar trend to studies conducted by various researchers in the literature [26,27] because when pH >11, Al flocs turn into soluble Al compounds and cause a decrease in floc formation. This negatively affects the separation of pollutants from the wastewater. Thanks to this feature of domestic wastewater treatment, the electrocoagulation process offers flexible operation in terms of pH. Fig. 2 shows the distribution of Al species depending on the pH of the environment [28].
The reason for the decrease in the effluent pH values due to the increasing flow rate is thought to be the decrease in residence time of the wastewater in the reactor linked to the increased flow rate. Therefore, the decrease in the contact time between the pollutant and the electrochemically-produced Al(OH)3 flocs is an important factor affecting the pH variation. The change of COD removal efficiencies vs initial pH as function of flow rate is given in Fig. 3.
For all initial pH values, which were investigated at constant current density, the increase in flow rate decreased COD removal efficiencies. The reason for this is that as the flow rate increases, the duration of the electrical field to which the wastewater is exposed becomes shorter and the pollutants cannot be sufficiently oxidized. In addition, the highest removal efficiencies at all flow rates occur when the initial pH value of the wastewater is 6–7. When Fig. 2 is examined, the solubility of aluminum is very low at pH values between 6.5–7.8. In this pH range, where aluminum has the lowest solubility, it is thought to provide the fastest COD removal since there is sufficient flocculant in the aquatic environment [29].
The effect of initial pH of wastewater on energy consumption was investigated at constant current density of 10 A at all flow rates, and is shown in Fig. 4.
The highest energy consumption value was obtained at the lowest flow rate (25 mL min−1) under constant current density. This trend is applicable to all pH values studied, and it is believed that the basic reason is that the electrical resistance increases depending on the increased contact time inside the reactor.
The conductivity of the solution is an important factor that affects the efficiency and power consumption of EC; the higher the conductivity, the lower the power consumption of EC [30]. The highest energy consumption occurred at pH 7.8, which is the natural pH of the wastewater, at all current densities. This is an expected result. Concentrated HCl was added to the wastewaters to obtain pH values below the natural pH value. This increased the conductivity of the wastewater. The increased conductivity decreased the potential difference applied to the system under constant current density. This decrease caused a decrease in energy consumption values according to Equation 6. Flow rate values of 10 mL min−1 were also examined with respect to energy consumption, but they are not shown in Fig. 4 because the results were very high.
The applied current density (j) controls the electrochemical reactions that occur in solution (electrodissolution rate, gas evolution, electroflotation, water reactions, etc.) as well as their continuation and kinetics. Consequently, j defines (with the applied potential) the energy consumption associated with the operation of the electrochemical process [31].
Experiments were conducted using wastewater with initial COD concentration of approximately 250 mg L−1 at the current densities of 5, 10 and 15 A to investigate the effect of current density on COD removal efficiency in a continuous system using the electrocoagulation method. The experiments were carried out at 10 mL min−1 of flow rate and pH=6. COD removal efficiencies and energy consumption values at the different current densities are shown in Fig. 5.
The increase in current density increased the effluent pH values. Effluent pH of wastewater was obtained as 8.2 for 5 A, 9.08 for 10 A and 9.21 for 15 A. Increasing the current density increases the amount of dissolved Al3+ and causes the pH to rise faster. In this case, the pH range of 6–7, where the highest COD removal is achieved, is quickly exceeded. This means that the produced Al3+ ions are used inefficiently. In this case, the COD removal efficiencies decrease [32]. According to Fig. 5, the highest removal efficiency was obtained at 5 A. In Fig. 5, the COD removal efficiency decreased from 79% to 67% depending on the increase in current density. This situation contradicts the known theory. However, examining the event only by considering the increase in current density will cause an error. In order to understand the negative effects of increase in current density on COD efficiency at low flow rate values, current density, flow rate and electrolyte pH value should be considered together. Current density determines the coagulant matter amount electrochemically generated in the reactor. The increase in current density increases the amount of Al3+ ions released per unit time. However, when pH values are examined carefully, the current densities of 10 and 15 A raised pH values of the effluent above the pH interval that is suitable for Al(OH)3 formation.
In Fig. 5, the increase in the current density also increased the energy consumption. According to the V=I*t equation, the increase in the current density will mean an increase in the potential difference applied to the system, so the energy consumption of the system increases. The energy consumption values at pHi=6 were obtained as 33 kW-h m−3 at 5 A, 115 kW-h m−3 at 10 A and 175 kW-h m−3 at 15 A. In this case, applying high current density to the system leads to rapid depletion of the electrodes, a decrease in the removal efficiency and an increase in energy consumption.
Contact time is another effective parameter for COD removal in the electrocoagulation process. In the electrocoagulation reactor, reactants, electrochemical transformation and products occur on the electrode surfaces. As the wastewater flow rate increases, the contact time decreases, and the rate of transport of the reagent to the electrode surface and the products to the solution increases [33]. Experiments were carried out at different flow rates at current density values of 10, 20, 30 and 40 A and wastewater pH=6, where the best removal results were obtained, and the results are shown in Fig. 6.
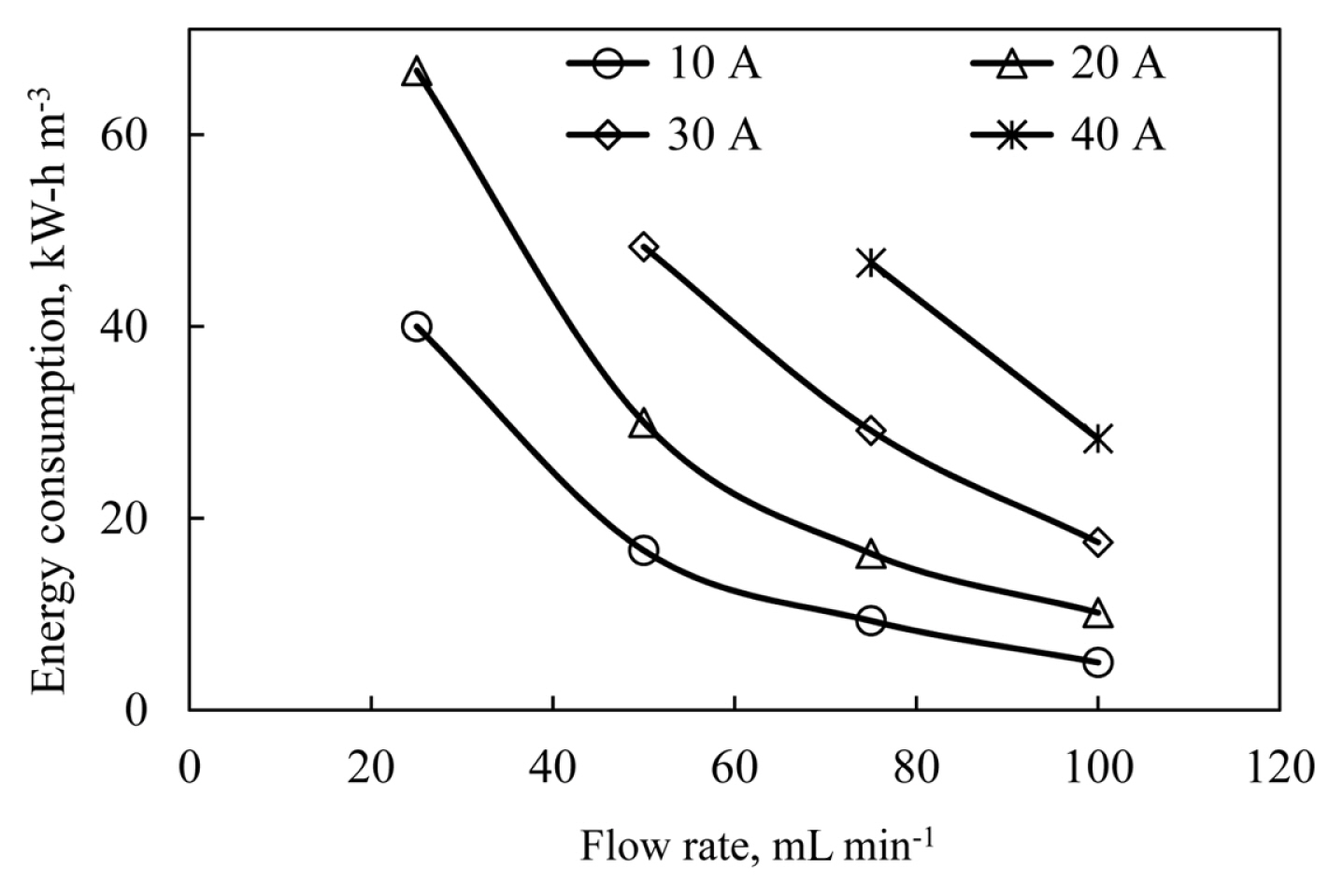
When the initial wastewater pH value is kept constant, the pH value of the effluent increases faster as the current applied to the system increases. The effluent pH values were obtained as 7.75, 8, 8.1 and 8.2 for 10, 20, 30 and 40 A, respectively. As seen in Fig. 6, if the flow rate and the current density are increased together, COD removal efficiencies are greatly increased. The reason for this is the decrease in the residence time at high flow rates, but the increase in the dissolution rate of Al3+ ions dissolved at the anode with the increase of the current density and the more efficient use of the Al(OH)3 flocs formed. The current density flowing through the system affects not only the removal efficiency, but also the electricity consumption. The electricity consumption values obtained as a result of the experiments performed at different current densities and different flow rates are shown in Fig. 7.
Energy consumption values were calculated with the help of potential differences occurring when current density and flow rate were increased simultaneously, and higher energy consumption values occur at lower flow rates. Energy consumption values decrease as the flow rate increases, but also increase as the current density applied to the system increases. Energy consumption values at different flow rates were 67 kW-h m−3 at 25 mL min−1 for 20 A, 48 kW-h m−3 at 50 mL min−1 for 30 A, 46.67 kW-h m−3 at 75 mL min−1 for 40 A and 28 kW-h m−3 at 100 mL min−1 for 40 A. The obtained experimental data for energy consumption are shown in Fig. 7.
In this study, the usability of the continuous system electrocoagulation process for domestic wastewater treatment was investigated. In the study, firstly, experiments were performed for different flow rates and different wastewater pHs and it was determined that the most suitable wastewater initial pH was around 6. In addition, regardless of wastewater pH, the pH measured at the system outlet never exceeded 9.5. This provides great flexibility for the treatment of domestic wastewater with the electrocoagulation system. At all flow rates studied, the highest removal efficiencies were obtained in the pH range of 6–7, where the solubility of Al(OH)3 is lowest.
In the studies about the effect of current density on COD removal efficiency, the increase in the current density passing through the system at constant flow rate affected the COD removal efficiency negatively. The reason for this is that the aluminum produced cannot be used efficiently since the increase in the current density increases the amount of dissolved aluminum but increases the pH too fast. Besides, it was determined that the COD removal efficiencies increase if the current density that passes through the system is increased together with the flow rate. In studies about the effect of flow rate on COD removal efficiency, the highest result was obtained with a flow rate of 75 mL min−1 under 40 A. It was also observed that the amount of energy per unit volume of treated wastewater decreased. For this reason, it is important to increase the flow rate and current density together, taking into account the pH values that allow the highest floc formation rate to ensure optimum operating conditions.
One of the biggest problems encountered in the biological treatment of domestic wastewater is that it is difficult to treat due to low organic matter content. As a result of these studies, the domestic wastewater of enterprises with low flow rate and organic matter content can be easily treated with a continuous flow electrocoagulation system instead of large and complex treatment systems that are difficult to operate.
Fig. 3
The change in COD removal efficiencies vs initial pH as a function of flow rate at 10 A and 15°C.

Fig. 4
The change in energy consumption vs initial pH of wastewater as a function of flow rate at 10 A and 15°C.

Fig. 5
The effect of current density on COD removal and energy consumption, pHi= 6, T=15°C, flow rate=10 mL min−1.
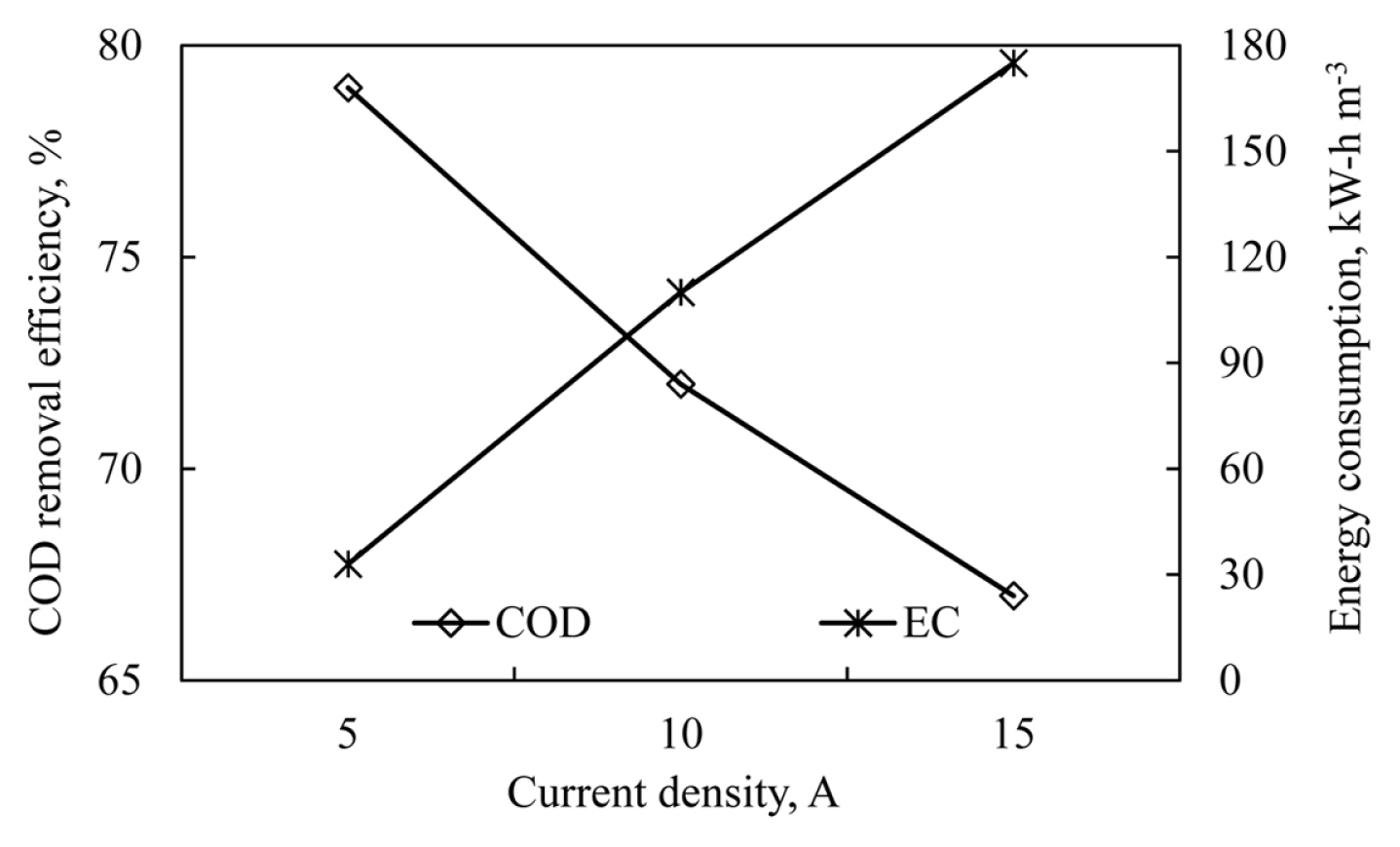
Table 1
Parameters affecting the electro-coagulation process for domestic wastewater treatment
References
[2] Q. H. Zhang, W. N. Yang, H. H. Ngo, W. S. Guo, P. K. Jin, S. J. Yang, Q. Wang, X. C. Wang and D. Ao, Environ. Int, 2016, 92, 11–22.

[8] N. T. Thuy, N. X. Hoan, D. V. Thanh, P. M. Khoa, N. T. Tai, P. Q. H. Hoang and N. N. Huy, J. Electrochem. Sci. Technol, 2021, 12(1), 21–32.


[9] M. A. Mamelkina, S. Cotillas, E. Lacasa, C. Sáez and M. A. Rodrigo, Sep. Purif. Technol, 2017, 182, 87–93.

[13] M. Dolati, A. A. Aghapour, H. Khorsandi and S. Karimzade, J. Environ. Chem. Eng, 2017, 5(5), 5150–5156.

[14] G. F. S. Valente, R. C. S. Mendonça, J. A. M. Pereira and L. B. Felix, Sep. Purif. Technol, 2014, 132, 627–633.

[17] R. Perumalsamy, C. Kumaran and V. Rajamanickam, J. Electrochem. Sci. Technol, 2021, 12(1), 92–100.


[19] S. Irdemez, N. Demircioglu, Y. S. Yildiz and Z. Bingul, Sep. Purif. Technol, 2006, 52, 218–223.

[21] D. Valero, J. M. Ortiz, V. Garcia, E. Espósito, V. Montiel and A. Aldaz, Chemosphere, 2011, 84, 1290–1295.

[22] APHA, Standarts Methos of Examination of Water and Wastewater. 23rd. Edition. 2017.
[24] A. S. Koparal, YŞ. Yildiz, B. Keskinler and N. Demircioğlu, Sep. Purif. Technol, 2008, 59(2), 175–182.

[27] M. Kobya, F. Ulu, U. Gebologlu, E. Demirbas and S. Oncel, Sep. Purif. Technol, 2011, 77, 281–293.
[30] D. T. Moussa, M. H. El-Naas, M. Nasser and M. J. Al-Marri, J. Environ. Manage, 2017, 186, 24–41.