|
 |
- Search
| J. Electrochem. Sci. Technol > Volume 13(4); 2022 > Article |
|
Abstract
In the study, Ni2+ (nickel) removal from synthetically prepared wastewater by electrocoagulation method, which is one of the electrochemical treatment processes, was investigated and parameters such as current density, pH, mixing speed, initial Ni2+ concentration, supporting electrolyte type and concentration were determined to determine Ni2+ removal efficiencies effects were studied. Experiment conditions during 30 minutes of electrolysis; the current density was determined as 0.95 mA/cm2, the initial pH of the wastewater was 6, the mixing speed was 150 rpm, and the initial nickel concentration was 250 mg/L. The Ni2+ removal efficiency was obtained as 75.99% under the determined experimental conditions, while the energy consumption was calculated as 3.15 kW-h/m3. In the experiments, it was observed that the type and concentration of the supporting electrolyte did not have a significant effect on the Ni2+ removal efficiency. In the trials where the effect of the support electrolyte concentration was examined, the Ni2+ removal efficiency was 75.99% in the wastewater environment without the supporting electrolyte, while the Ni2+ removal efficiency was 81.55% when 7.5 mmol/L NaCl was used after the 30-minute reaction, and the energy consumption was 2.15 kW-h/m3 obtained as. As a result of the studies, it was concluded that the electrocoagulation process can be applied in the treatment of wastewater containing Ni2+.
The increase in the need for drinking and utility water due to rapid population growth and industrialization causes rapid depletion of water resources. A large amount of water is consumed in industrial activities applied in developed countries. After being treated, the wastewater generated due to water consumption should be discharged to the receiving environment in accordance with the appropriate discharge standards.
Since wastewater is discharged to receiving environments such as seas and lakes, which are generally closest to industries, the pollution they will create can cause the ecological balance to deteriorate in these environments. Industries such as paper, textile, food, leather and chemistry, which are established in or near settlements, give high amounts of pollution to the receiving environment [1]. Heavy metals also play a major role in environmental pollution caused by industrial wastewater. Heavy metals are often found along with other pollutants in wastewater from the electroplating, metal plating and cleaning, iron and steel, textile, auto parts, printing, paint, leather and petrochemical industries [2].
Heavy metals are pollutants that have a toxic effect even at very low concentrations. Heavy metals, which are frequently used in production processes or auxiliary processes in many industries, are called micropollutants and elements such as Hg, Ni, Cr, As, Pb, and Cd can be given as examples of heavy metals. Most of the heavy metals in wastewater are found in sewage sludge. Some of them dissolve and reach water sources such as surface waters, lakes and seas and stay here and can reach the drinking water and food chain. Heavy metals coming into the food chain cannot be removed from the body chemically or biologically and cause accumulation in the body [3,4]. Nickel, which is a heavy metal; While it is mainly used in alloy making, 12% of nickel is used in the production of superalloys, about 65% in the making of stainless steel, and the remaining 23% in rechargeable batteries, coin making, foundry products, and coatings [5,6]. Nickel can enter the human body with cigarette smoke, consumed foods and drinking water. It is natural to take small amounts of nickel, but high amounts can be dangerous for human health. Diseases such as lung, larynx, nose, prostate cancer development, lung and respiratory failure, shortness of breath, asthma and chronic bronchitis can be listed as health problems caused by high amounts of nickel. Since they are chemicals with high toxicity and accumulation tendency, removal of heavy metals is of great importance in terms of environmental as well as living health. Considering the type of heavy metal, the characteristics of the water to be treated and the size of the treatment need in the treatment of waste-water containing heavy metals; Various methods such as sulfur or hydroxide precipitation, adsorption and ion exchange can be applied. Hydroxide precipitation is known as the most suitable treatment method for the treatment of metal-containing wastewater. This method is based on the precipitation of metals as hydroxides at optimum pH values, and active silica or polymers can be added as coagulation aids. Although the chemical precipitation method is very effective in the treatment of industrial wastewater, chemicals added to the environment as coagulants can cause secondary pollutant problems. Considering the secondary pollutant problem and economic concerns, the applicability of the electrocoagulation method in the treatment of different industrial waste-waters has been rapidly investigated recently. Electrocoagulation method is a simple and effective method in water and wastewater treatment. In this method, the coagulant is obtained as a result of the electrooxidation of Al (aluminum) and Fe (iron) anodes, and at the same time, hydrogen gas is formed at the cathode. One of the biggest advantages of the electrocoagulation method is that no chemicals are added as a coagulant. Accordingly, the amount of sludge to be formed in the system is negligible. When the literature is examined, studies on heavy metal removal from wastewater by using electrocoagulation method are very limited, but studies on this subject have been started in recent years [7].
Adhoum et al. (2004) examined the purification of synthetically prepared samples containing Cu2+, Zn2+ and Cr6+ by electrocoagulation method using aluminum electrode. In the experiments carried out to determine the effect of pH on metal removal efficiency at 50 mg/L initial metal concentrations, 50 cm2 anode surface area and 25┬░C temperature, it was concluded that effective metal removal was obtained between pH 4ŌĆō8, and pH 6 was the optimum pH. In addition, it was determined in the studies that the metal removal efficiency increased as the current density increased, and the removal efficiency decreased with the increase of the initial metal concentration [8].
Canizares et al. (2005) investigated the solubility of aluminum electrodes in the electrocoagulation process. It has been stated that the electrical dissolution of the anode surface in the electrocoagulation process is the main mechanism that provides the formation of coagulant. Experiments examining the chemical solubility of aluminum were repeated for pH 3, 7, 12, 13, and NaCl 2450 mg/L or Na2SO4 3000 mg/L conditions. As a result of these experiments, it was observed that the chemical solubility of aluminum was highly affected by pH and alkaline pH increased the solubility. In addition, it was observed that the electrolyte (NaCl or Na2SO4) did not affect the chemical solubility. Experiments examining the electrochemical solubility of the aluminum electrode were carried out under current density 0.5 mA/cm2, flow rate 0.0055 L/sec, pH 6 and NaCl 2450 mg/L or Na2ŌłÆ SO4 3000 mg/L conditions [9].
Electrocoagulation: It is an electrochemical waste-water treatment process in which a soluble and coagulating metal electrode is used. Electrocoagulation is based on the principle of forming coagulants by electrical dissolution of the electrode material used. Iron and aluminum, which are easy to obtain and cheap as electrode materials, are the most preferred materials. The difference between electrocoagulation and chemical coagulation is the way aluminum or iron is added to the medium [10]. In electrocoagulation, coagulation and precipitation mechanisms are not carried out by adding chemicals to the system, but by the electrodes in the electrochemical reactor [11,12].
In this study, the treatment of nickel removal from synthetically prepared wastewater by electrocoagulation method, which is one of the electrochemical treatment processes, was investigated. At the same time, the most effective treatment conditions will be determined in the light of the parameters that will affect the removal efficiency.
The studies were carried out to investigate the treatment of nickel removal from synthetically prepared wastewater by electrocoagulation method, which is one of the electrochemical treatment processes. The synthetic wastewater studied in the experiments was prepared by using the compound NiCl2┬Ę6H2O. In the studies, a stock solution containing 1000 mg/L Ni2+ was prepared, and the experiments were carried out using 1000 mL of synthetic wastewater by making appropriate dilutions.
The electrocoagulation unit is made of 15├Ś8 ├Ś10 cm plexiglass. A total of 10 aluminum (Al) electrodes, 5 anodes and 5 cathodes, were used in the studies. The aluminum plates used are placed in the electrocoagulation cell at 5 mm intervals and are monopolar configurations with a height of 7.5 cm and a width of 7 cm. The total wet surface area is calculated as 1050 cm2. The current is controlled through the power supply (GW GPC-3060D). Nickel analysis was performed using atomic absorption spectrometry (SHIMADZU AA6800). Samples were taken against time and the removal efficiencies and energy consumptions from these samples were calculated with the help of the following equations.
Removal efficiency:
Energy consumption:
Where C0: Initial metal concentration (mg/L), Ct: Metal concentration at any time t (mg/L), V: System voltage (Volts), I: Current intensity (Amperes), t: Time (hours), v: Shown as wastewater volume (m3).
One of the most important process parameters in electrocoagulation is the initial solution pH [8]. Since the pH of the solution affects the stability of the hydroxide species, its effect on the removal efficiency also causes positive or negative results. As a result of the reactions of the anode and cathode reactions in the aqueous solution, changes in the pH value of the solution occur. Therefore, depending on the pH of the solution, electrolytically dissolving metal ions (Al3+ and Fe2+) form various metal hydroxide species at different pH values [13]. At high pH values, heavy metals precipitate as hydroxide. Therefore, pH values higher than 6 have not been studied to investigate heavy metal removal without precipitation.
When aluminum anode is used, the initial pH of the solution was adjusted to pH values varying in the range of 3ŌĆō6 in order to examine the effect of the initial pH of the wastewater on the Ni2+ removal efficiency. Initial Ni2+ concentration was determined as 250 mg/L, mixing speed 150 rpm, current density 0.95 mA/cm2 experimental conditions. Ni2+ removal efficiencies of 62.76, 67.63, 75.15 and 75.99% were obtained for the initial pH values 3, 4, 5 and 6, respectively, during the 30-minute electrolysis period. Aluminum can form different chemical species depending on the pH of the solution and the presence of other elements. At low pH, the coagulation effect of aluminum is insignificant in the form of Al3+, and between pH 5.0ŌĆō7.5, Al(OH)3, Al(OH)2+, Al17(OH)327+, which are effective coagulants for pollutants. It appears to be in the form of aluminum complexes such as the solubility of Al(OH)3 will increase at higher pH values. Therefore, it is considered that the most efficient treatment is achieved due to the presence of sufficient flocculator in the environment in the pH range of 5.0ŌĆō7.5 and the pH being in the range where aluminum has the lowest solubility [7]. Therefore, the next experiments were continued by adjusting the initial pH value to pH 6 [14,15]. The results obtained are shown graphically in Fig. 1 and the calculated energy consumptions are shown graphically in Fig. 2.
The current density applied to each unit area of the electrode is an important parameter in electrochemical processes in terms of controlling the reaction rate [16,17]. According to FaradayŌĆÖs law, as the amount of current increases, the amount of oxidized and reduced substances in the electrodes will also increase.
In order to examine the effect of the current density applied in the electrocoagulation method on the Ni2+ removal efficiency, the current densities were adjusted to 0.24, 0.48, 0.95 and 1.43 mA/cm2 values, and the experimental conditions were kept constant as the initial Ni2+ concentration of 250 mg/L, the initial pH value 6 and the mixing speed 150 rpm. Ni2+ removal efficiencies end of 30 min reaction time for 0.24, 0.48, 0.95 and 1.43 mA/cm2 current densities were obtained as 58.29, 68.71, 75.99 and 92.38%, respectively. Based on these results, which also took into account the energy cost, the experiments continued with a current density of 0.95 mA/cm2, where the most effective removal efficiency was obtained. Similar studies are available in the literature [18,19]. The results obtained are given graphically in Fig. 3 and the calculated energy consumption values are given in Fig. 4.
The purpose of the mixing speed is to ensure that the flocculants formed due to the dissolving electrodes are homogeneously dispersed in the reactor. Failure to provide an effective dispersion causes the lack of a homogeneous environment in the reactor and thus the formation of regional flocculation. In addition, mixing ensures that variable parameters such as pH and temperature in the system are homogenized.
In order to examine the effect of mixing speed on Ni2+ removal efficiency when aluminum anode is used, initial Ni2+ concentration is 250 mg/L, wastewater initial pH value is 6, current density is 0.95 mA/cm2 for 50, 150, 250 and 400 rpm mixing speeds was determined as the experimental conditions. Ni2+ removal efficiencies were obtained as 71.23, 75.99, 67.91 and 64.49% respectively, for 50, 150, 250 and 400 rpm mixing speeds in 30 minutes electrolysis time. As can be seen from the results, the experiments were continued with a mixing speed of 150 rpm, where the highest removal efficiency was obtained. Increasing the mixing speed decreased the nickel removal efficiency. Increasing the mixing speed decreases the flock formation capacity of aluminum ions. Stirring speeds less than 150 rpm decrease the nickel removal efficiency and do not allow a homogeneous mixture to be provided in the reactor. The effect of mixing speed was also investigated in previous studies, and similar results were obtained with the results of this study [20]. Similar studies are available in the literature [21,22]. The results obtained are shown graphically in Fig. 5 and the calculated energy consumption values in Fig. 6.
As the Ni2+ concentration of the wastewater increases, the heavy metal removal efficiency decreases. But the amount of heavy metal removed increased. The variation of removal of metal by electrocoagulation were investigated at an initial pH of 6, a mixing speed of 150 rpm and current density of 0.95 mA/cm2 for initial metal concentration range (25ŌĆō100 mg/L). The removal efficiencies for 30 minutes of electrolysis; 25, 50, 100, 250, 500 and 1000 mg/L initial Ni2+ concentrations were 95.34, 91.18, 86.72, 75.99, 62.33 and 45.09% respectively. After that, the initial Ni2+ concentration was continued as 250 mg/L. Obtained results are shown graphically in Fig. 7 and energy consumptions are shown in Fig. 8.
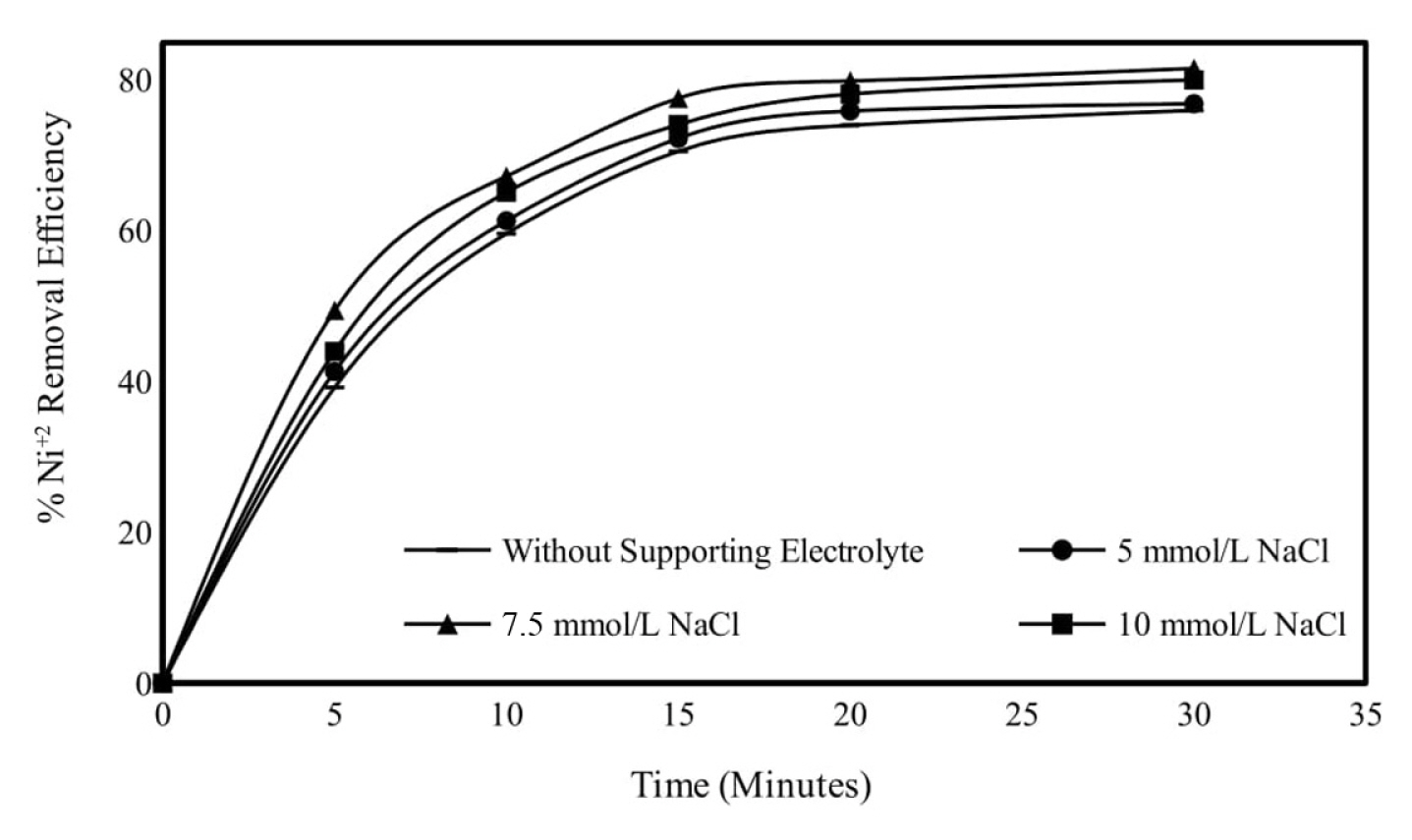
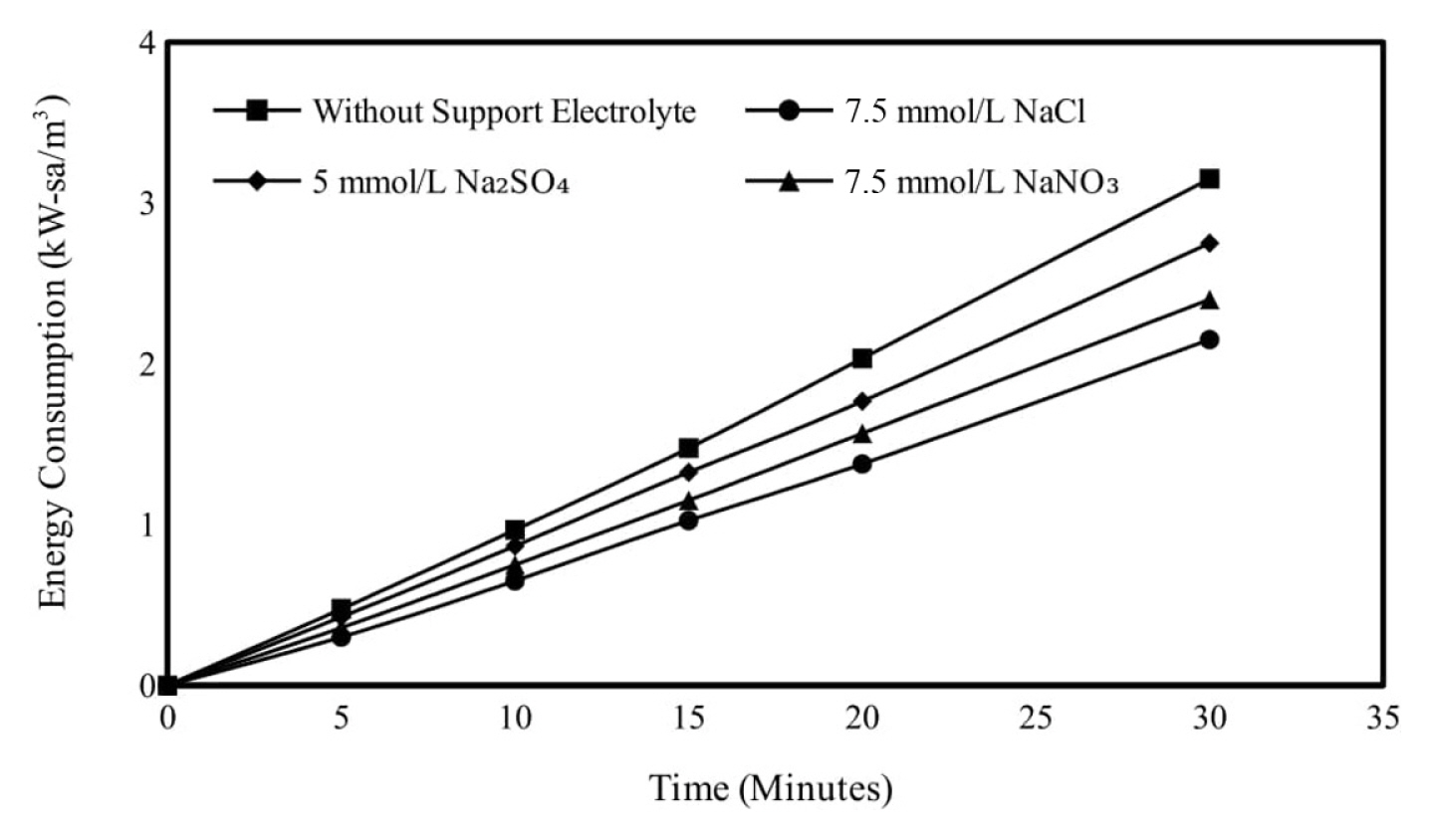
The effect of supporting electrolyte types on Ni2+ removal by electrocoagulation was studied on without supporting electrolyte; 7.5 mmol/L NaCl; for 5 mmol/L Na2SO4 and 7.5 mmol/L NaNO3 salt types at pH 6.0, 150 rpm mixing speed, 0.95 mA/cm2 current density and 250 mg/L initial metal concentration. Removal efficiencies end of 30 minutes of electrolysis for without supporting electrolyte, 7.5 mmol/L NaCl, for 5 mmol/L Na2SO4 and 7.5 mmol/L NaNO3 salt types were obtained as 75.99, 81.55, 77.70 and 79.55%, respectively. NaCl, which is used as a supporting electrolyte type in electrocoagulation applications, both increases conductivity and decreases energy consumption. In addition, NaCl is highly soluble in water compared to other supporting electrolyte types and is an easily accessible and inexpensive supporting electrolyte type. Subsequent experiments were continued by choosing NaCl as the supporting electrolyte type. Obtained results are given graphically in Fig. 9 and calculated energy consumption values in Fig. 10.
Wastewater conductivity is an important parameter for electrochemical treatment processes. Generally, a higher conductivity value means lower resistance in the electrochemical cell and the efficiency of the process is higher [23]. Under alkaline conditions, hypochlorite, chloride ions, and hydroxyl radicals are important, while in acidic conditions free chloride is the predominant oxidizing agent [24].
The removal of nickel by electrocoagulation was studied by varying the concentration of supporting electrolyte without supporting electrolyte, 5 mmol/L NaCl, 7.5 mmol/L NaCl and 10 mmol/L NaCl in 0.95 mA/cm2 current density, pH 6, 250 mg/L the initial metal concentration and 150 rpm mixing speed. The removal of nickel was equal to 75.99, 76.87, 81.55 and 81.14% for having supporting electrolyte concentration of without supporting electrolyte, 5, 7.5 and 10 mmol/L, respectively. Since no significant effect was observed on the treatment efficiencies obtained from the added support electrolyte type and concentration, the next experiments were continued without using the supporting electrolyte. Obtained results are given graphically in Fig. 11 and energy consumption values in Fig. 12.
The following results were obtained in the studies examining the removal of nickel from wastewater containing heavy metals by electrocoagulation method. In the study, the effects of parameters such as current density, pH, mixing speed, initial Ni2+ concentration, supporting electrolyte type and concentration on Ni2+ removal efficiencies were investigated. Experiment conditions during 30 minutes of electrolysis; The current density was determined as 0.95 mA/ cm2, the initial pH of the wastewater was 6, the mixing speed was 150 rpm, and the initial nickel concentration was 250 mg/L. The Ni2+ removal efficiency was obtained as 75.99% in the determined experimental conditions, while the energy consumption was calculated as 3.15 kW-h/m3. In the experiments, it was observed that the type and concentration of the supporting electrolyte did not have a significant effect on the Ni2+ removal efficiency.
Acknowledgments
This work is supported by the Scientific Research Project Fund of Balikesir University under the project number 2018/183
Fig.┬Ā1
The effect of initial pH value of wastewater on Ni2+ removal efficiency in electrocoagulation trials. Initial Ni2+ concentration: 250 mg/L, mixing speed: 150 rpm, current density: 0.95 mA/cm2
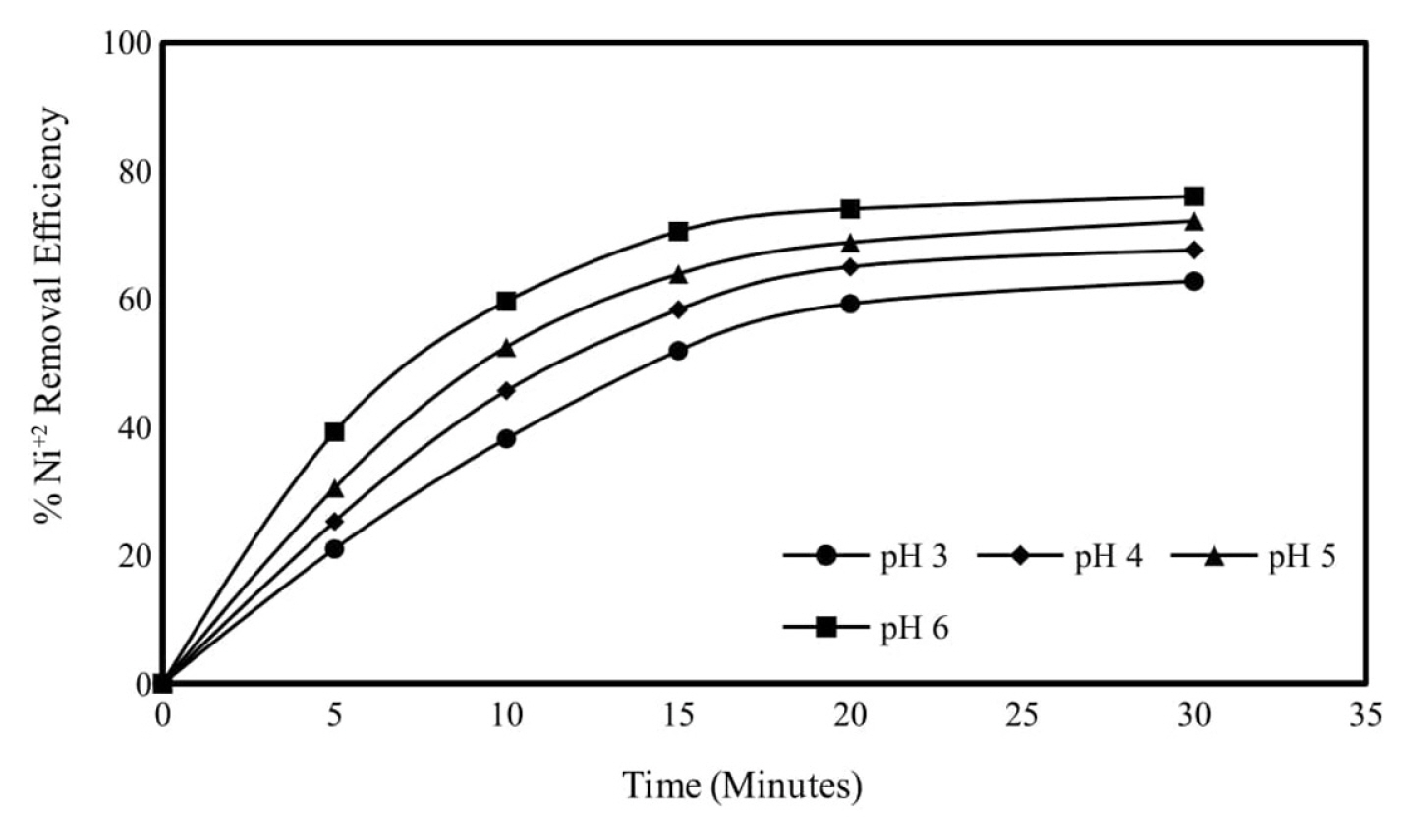
Fig.┬Ā2
The effect of initial pH value of wastewater on energy consumption in electrocoagulation trials. Initial Ni2+ concentration: 250 mg/L, mixing speed: 150 rpm, current density: 0.95 mA/cm2

Fig.┬Ā3
Effect of current density applied in electrocoagulation trials in Ni2+ removal efficiency. Initial Ni2+ concentration: 250 mg/L, mixing speed: 150 rpm, pH: 6
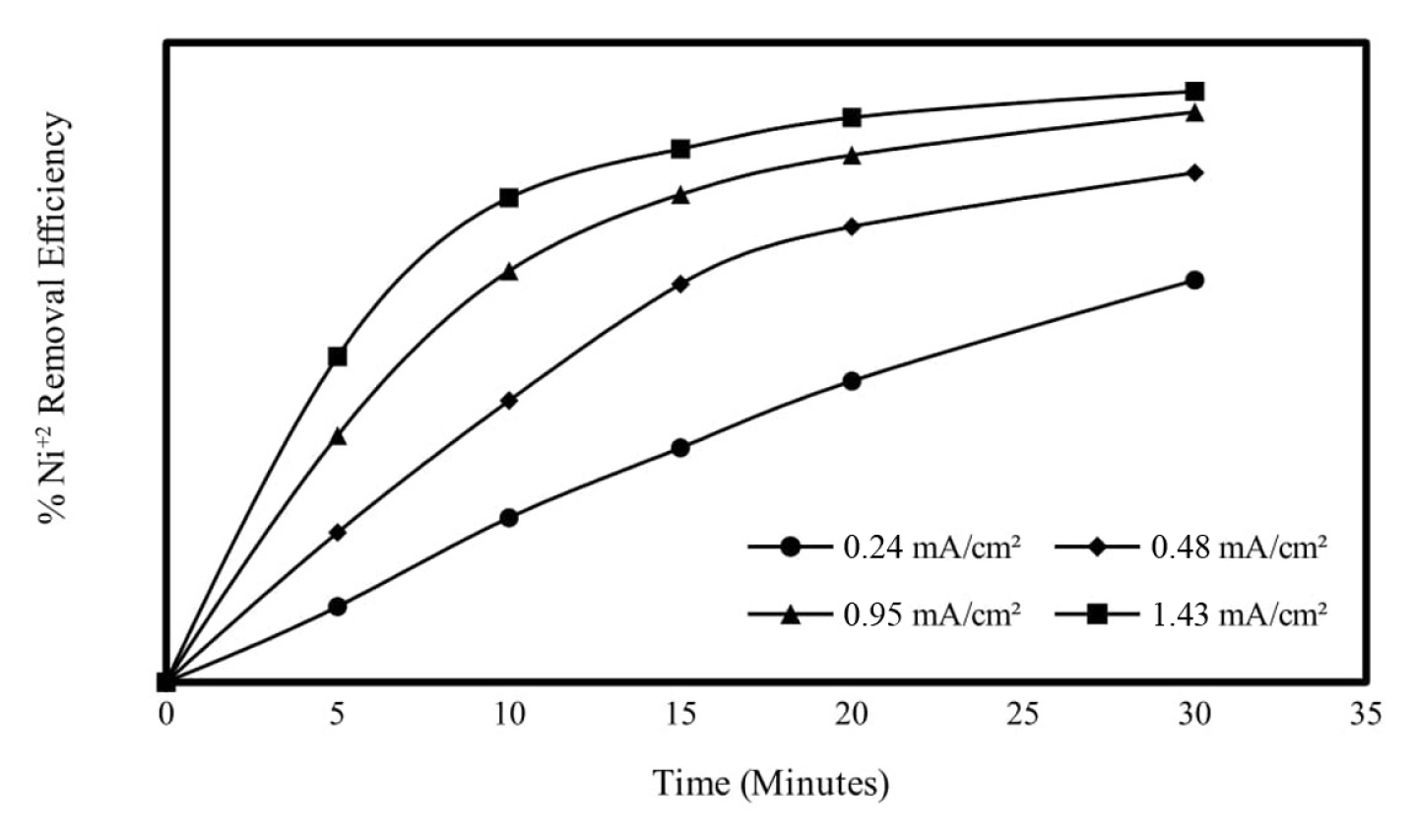
Fig.┬Ā4
The effect of current density applied in electrocoagulation experiments on energy consumption. Initial Ni2+ concentration: 250 mg/L, mixing speed: 150 rpm, pH: 6
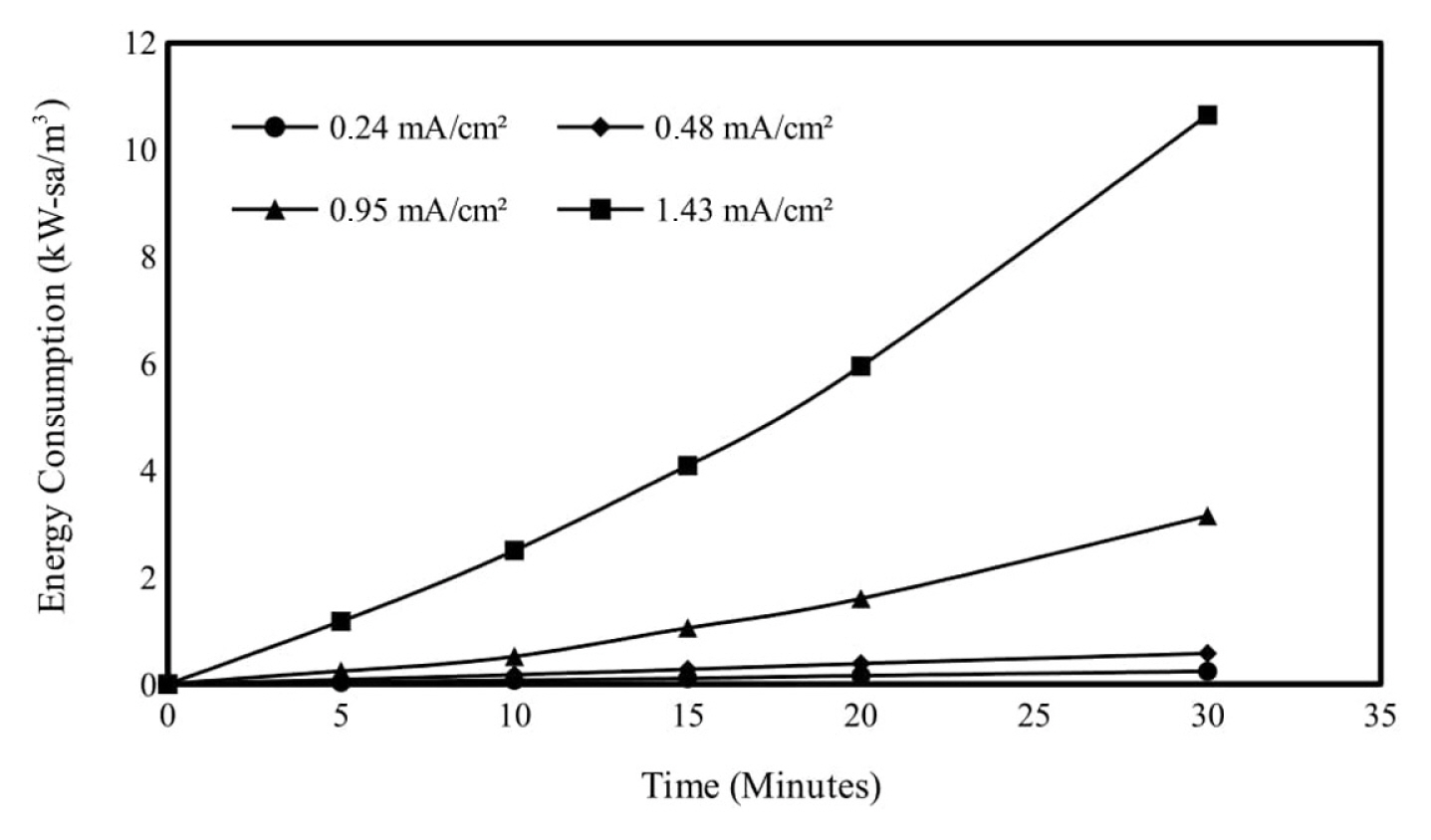
Fig.┬Ā5
Effect of mixing speed on Ni2+ removal efficiency in electrocoagulation trials. Initial Ni2+ concentration: 250 mg/L, pH: 6, current density: 0.95 mA/cm2
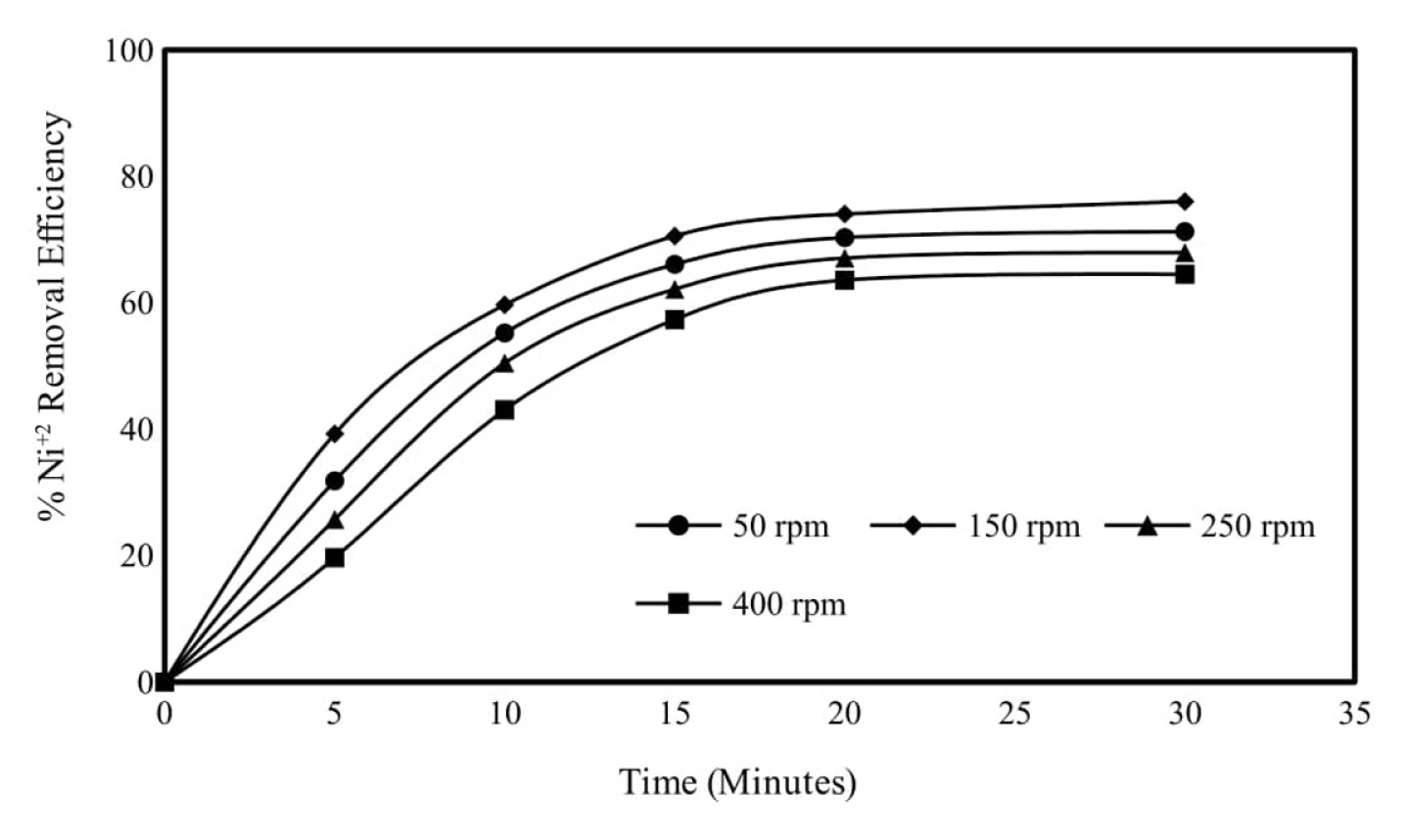
Fig.┬Ā6
Effect of mixing speed in energy consumption in eletrocoagulation trials. Initial Ni2+ concentration: 250 mg/L, pH: 6, current density: 0.95 mA/cm2

Fig.┬Ā7
Effect of initial concentration on Ni2+ removal efficiency in electrocoagulation trials. pH: 6, mixing speed: 150 rpm, currnet density: 0.95 mA/cm2

Fig.┬Ā8
Effect of initial concentration on energy consumption in electrocoagulation trials. pH: 6, mixing speed: 150 rpm, current density: 0.95 mA/cm2
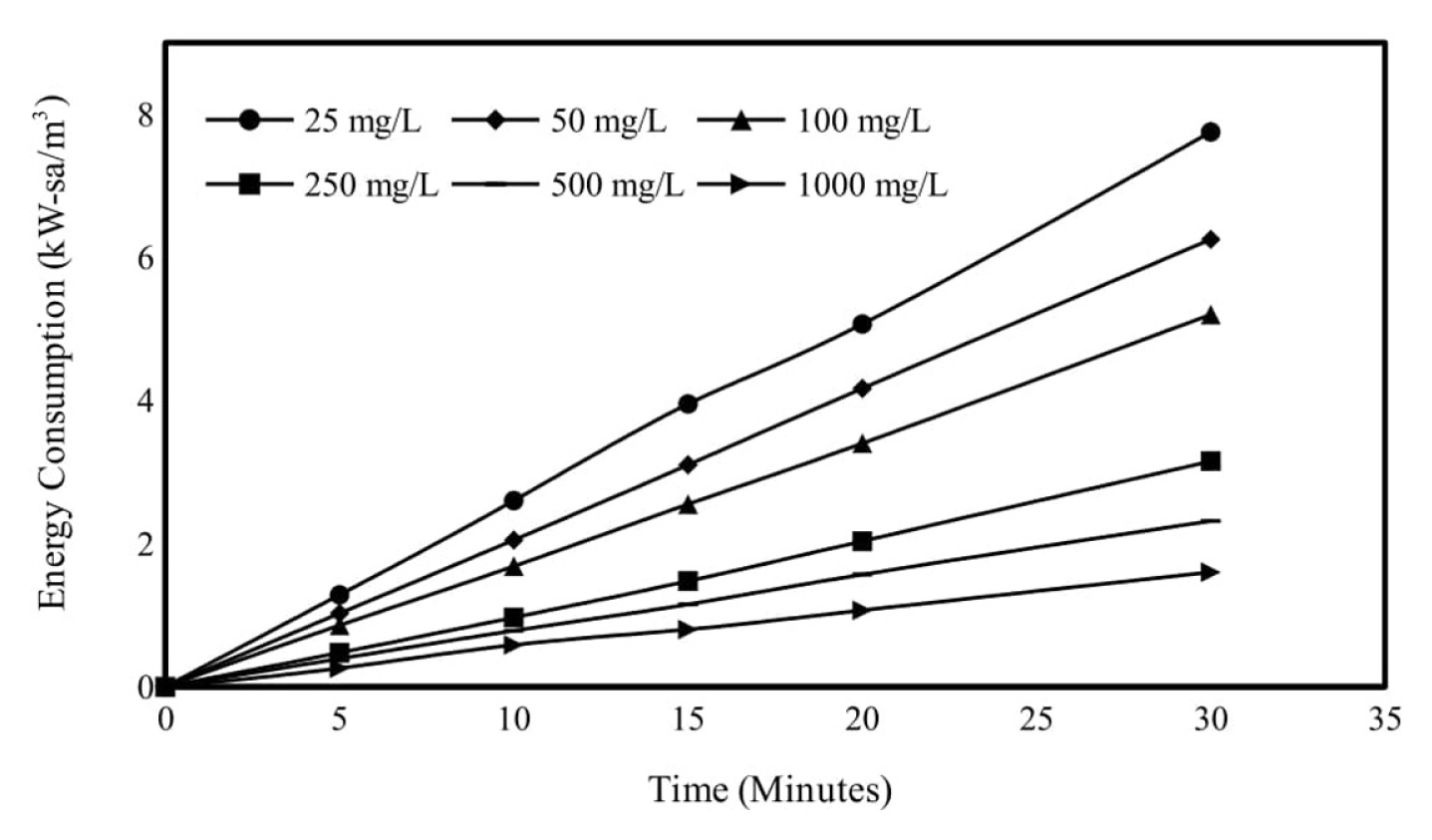
Fig.┬Ā9
Effect of supporting elecrolyte type on Ni2+ removal efficiency in electrocoagulation trials. Initial Ni2+ concentration: 250mg/L, supporting electrolyte concentraion: 7.5mmol/L, pH: 6, mixing speed: 150 rpm, current density: 0.95 mA/cm2

Fig┬Ā10
Effect of supporting electrolyte type on energy consumption in electrocoagulation trials. Initial Ni2+ concentration: 250 mg/L, supporting electrolyte concentration: 7.5 mmol/L, pH: 6, mixing speed: 150 rpm, current density: 0.95 mA/cm2
References
[1] B.A. Fil, Treatment of pistachio processing wastewater by electrooxidation method. PhD Thesis. Department of Environmental Engineering, Graduate School of Natural and Applied Sciences, Ataturk University, Erzurum, Turkey, 2014.
[2] K. Sezer, Investigation of the adsorption of single and binary mixtures of Cadmium(II) and Nickel(II) ions in wastewaters on chitosan, clay and chitosan-clay composite in batch and continuous systems. PhD Thesis Department of Chemical Engineering, Graduate School of Natural and Applied Sciences, Haccettepe University. Ankara, Turkey, 2015.
[3] F.R. Siegel, Environmental geochemistry of potentially toxic metals. Springer, 2002.
[4] E. Filiz, Heavy metal removal from water with adsorbents obtained from natural sources. Master Thesis. Department of Chemical Engineering, Graduate School of Natural and Applied Sciences, Istanbul Technical University, Istanbul, Turkey, 2007.
[8] N. Adhoum, L. Monser, N. Bellakhal and J.-E. Belgaied, J. Hazard. Mater, 2004, 112(3), 207ŌĆō213.

[9] P. Canizares, M. Carmona, J. Lobato, F. Martinez and M. Rodrigo, Ind. Eng. Chem. Res, 2005, 44(12), 4178ŌĆō4185.

[10] P. Drogui, M. Asselin, S.K. Brar, H. Benmoussa and J.-F. Blais, Sep. Purif. Technol, 2008, 61(3), 301ŌĆō310.

[11] A. Koparal, S. Gokcen and U. Ogutveren, Symposium on Environmental Pollution Priorities in Turkey-III, 1999, 1, 370ŌĆō379.
[12] S. Bayar, R. Boncukcuoglu, B.A. Fil and A.E. Yilmaz, J. Inst. Sci. Technol, 2012, 2(2), 21ŌĆō28.
[13] M.Y.A. Mollah, R. Schennach, J.R. Parga and D.L. Cocke, J. Hazard. Mater, 2001, 84(1), 29ŌĆō41.

[14] S. Camci, Treatment of metal plating wastewater by electrocoagulation method. Master Thesis Department of Environmental Engineering, Graduate School of Natural and Applied Sciences, Ondokuz May─▒s University. Samsun, Turkey, 2008.
[15] T. Arslan, I. Kabdasli, ─░. Arslan-Alaton, T. Olmez and O. Tunay, Su Kirlenmesi Kontrol├╝ Dergisi, 2008, 18(1), 42ŌĆō52.
[19] S. Yi─¤it, Electrochemical treatment of rinsing bath effluent from the nickel-cyanide metal plating process. Master Thesis Department of Environmental Engineering, Institute of Engineering and Science, Gebze Institute of Technology. Gebze, Turkey, 2018.
[20] J-w. Feng, Y-b. Sun, Z. Zheng, J-b. Zhang, L. Shu and Y-c. Tian, J. Environ. Sci, 2007, 19(12), 1409ŌĆō1415.

[21] C. Akarsu, Akademik Platform. Karab├╝k, 2014.
[22] N. Tzanetakis, W. Taama, K. Scott, R. Jachuck, R. Slade and J. Varcoe, Sep. Purif. Technol, 2003, 30(2), 113ŌĆō127.