|
 |
- Search
| J. Electrochem. Sci. Technol > Volume 13(3); 2022 > Article |
|
Abstract
Silicon (Si) has attracted considerable attention due to its high theoretical capacity compared to conventional graphite anode materials. However, Si-based anode materials suffer from rapid capacity loss due to mechanical failure caused by large volume change during cycling. To alleviate this phenomenon, crosslinked polymeric binders with strong interactions are highly desirable to ensure the electrode integrity. In this study, thermally crosslinked polyimide binders were used for Si-alloy anodes in Li-ion batteries. The crosslinked polyimide binder was found to have high adhesion strength, resulting in enhanced electrode integrity during cycling. Therefore, the Si-alloy anodes with crosslinked polyimide binder provide enhanced electrochemical performance, such as Coulombic efficiency, capacity retention, and cycle stability.
With an expansion of electric vehicles, portable electronic devices, and energy storage systems, the demand for large improvements in the current lithium-ion batteries (LiBs) with high energy density and improved cycle life is ever increasing. Although graphite has been widely used an anode materials for LiBs, its available capacity is limited to around 350 mAh/g [1].
Silicon (Si) has extensively been investigated as promising anode materials due to its high theoretical capacities. However, Si-based anode materials suffers from rapid capacity loss due to the mechanical failure induced by significant volume changes during lithiation and de-lithiation [2]. To address this issue, many innovative methods have been developed, including design and engineering of electrode architectures and functional binders [3,4]. For examples, electrode materials with nanostructures, porous structures, and 3D architectures have been developed to mitigate the stress resulting from volume changes [5].
Except for optimizing the electrode materials, functional polymeric binders with strong interactions between the active materials as well as the current collector are highly crucial to ensure the electrode integrity. So far, polymeric binders, such as sodium alginate, poly (acrylic acid), carboxymethyl cellulose, guar gum, chitosan, poly (vinyl alcohol), polyimide (PI), etc., have investigated. These polymers have polar functional groups (e.g., COOH, OH, and NH2), therefore resulting in interactions between the active material, conductive additives, as well as the current collectors. To further improve the electrode integrity, crosslinking is most promising route. Various crosslinking strategies have been intensively studied to develop crosslinked polymer binders, such as poly (acrylic acid)/carboxymethyl cellulose [6], poly (acrylic acid)/poly (vinyl alcohol) [7], poly (vinyl alcohol)/poly (ethylene imine) [8], carboxymethyl cellulose/poly (ethylene glycol) [9], polyacrylamide [10], and natural rubber/chitosan [11]. These crosslinked polymer binders can provide the mechanical integrity of Si-based anode due to the strong adhesion and the effective alleviation of the large volume change of Si during lithiation/delithiation process, resulting in enhancing the electrochemical performance, such as excellent cycling stability and outstanding Coulombic efficiency at higher current densities. Therefore, crosslinked polymer binders are potential candidates for Si anodes in next-generation LiBs.
In this study, we report thermally crosslinked PI binder for Si-alloy anodes in Li-ion batteries. Organic soluble PI with various amount of carboxylic acid groups was synthesized and used as Si-alloy anode binders. It is well known that PI with carboxylic acid groups undergo decarboxylation-induced crosslinking reaction by thermal treatment [12]. Si-alloy anodes with crosslinked PI binders were characterized in terms of their adhesion strength and electrochemical performance. As a result, Si-alloy anodes with crosslinked PI binders deliver enhanced electrochemical performance, such as Coulombic efficiency, capacity retention, and cycle stability.
Monomers, 4,4′-(hexafluoroisopropylidene) diphthalic anhydride (6FDA), m-phenylenediamine (mPDA), and 3,5-diaminobenzoic acid (DABA) were purchased from Sigma-Aldrich and they were dried at 200°C in a vacuum oven. N-methyl-2-pyrrolidinone (NMP, anhydrous), acetic anhydride (AcAn), and pyridine were purchased from Sigma-Aldrich and dehydrated with molecular sieves. Silicon alloy (Si50Al30Fe20) was prepared according to our previous study [13].
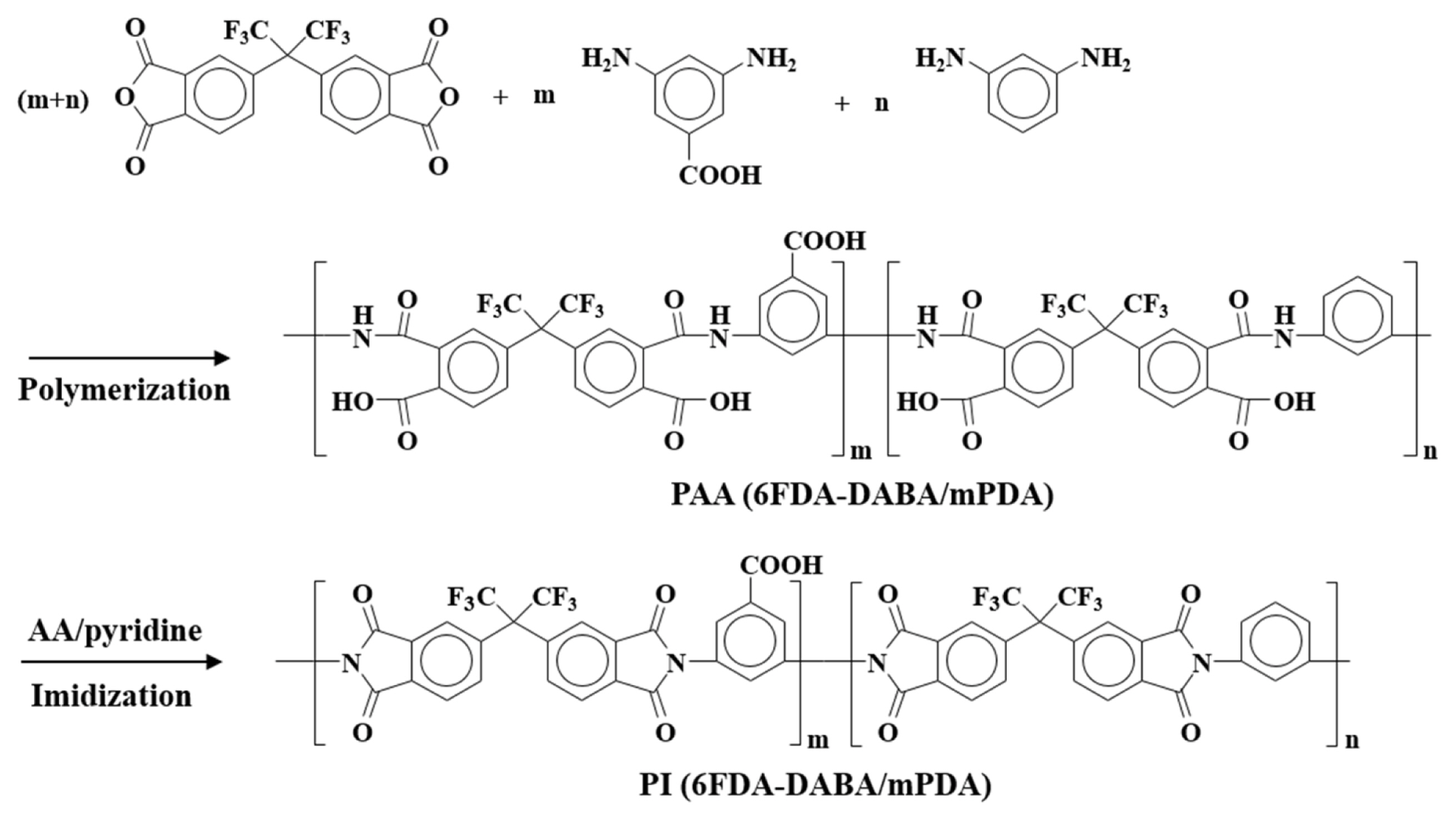
PI was synthesized by general two-step procedures: polycondensation and chemical imidization. Briefly, a stoichiometric amount of DABA or mPDA was added to a mixture of 6FDA in NMP, and then reacted at 25°C for 48 h under N2 atmosphere to obtain poly (amic acid) (PAA) solutions. For chemical imidization, a mixture of AcAn and pyridine (1/1, v/v, with a molar ratio of 1:4 to 6FDA) was added into a PAA solution and then stirred at 80°C for 12 h under N2 atmosphere. The resulting PI was precipitated, washed with methanol several times, and dried at 80°C under vacuum for 24 h.
The chemical structures were analyzed by 1H NMR (Avance 400, Bruker) and FT-IR (Spectrum Two, PerkinElmer) spectrometers. The thermal properties were investigated by thermogravimetric analysis (TGA, TGA-8000, PerkinElmer, USA) at a temperature range of 50 – 800°C with a heating rate of 5°C min−1 under N2 atmosphere. The morphologies were obtained by field emission scanning electron microscopy (FE-SEM, S-4300, Hitachi). The adhesion strength was measured using a 180° peel test apparatus (5848 MicroTester, Instron) using a scotch tape (Magic, 3M) at a 20 mm sec−1 (electrode sample of 20 mm width/80 mm length). The gel content of the thermally treated PI was determined by immersing the pre-weighted samples in NMP at room temperature for 24 h, and the insoluble fractions were dried under vacuum for at least 24 h to a constant weight. The gel content was calculated by the following equation: gel content = W1/W0 × 100 (%), where W0 and W1 are the initial weight of the thermally treated PI and the weight of the insoluble fraction in NMP, respectively.
An anode slurry was prepared by mixing the Sialloy particles (87 wt%), ketjen black (3 wt%), and PI binder (10 wt%) in NMP using a planetary mixer (Corona Mix CRM I.5L TSI Co. Ltd.,). The slurry was coated on the Cu foil current collector using a casting knife, and then dried at 110°C for 2 h in a drying oven. The loading level and electrode density were 1.0 – 1.1 mg cm−2 and 0.8 – 0.9 g cm−3, respectively. The prepared anodes were thermally treated at 400°C for 1 h in a tubular furnace (Ajeon Heating Industrial) under an inert atmosphere to crosslink the PI, and then naturally cooled to room temperature. The 2016 coin cells were assembled in an Ar-filled glove box. The cells were composed of lithium metal as a counter electrode, a polypropylene separator (Celgard 2400), and the prepared anode. An electrolyte solution composed of 1 M LiPF6 in ethylene carbonate (EC)/ethyl methyl carbonate (EMC) (EC:EMC, 3:7 v/v) with a fluoroethylene carbonate of 5 wt% (Dongwha Electrolyte, Korea) was injected between the two electrodes. As a comparative experiment, an anode was prepared using a commercial PVDF binder in the same manner without any thermal treatment. The constant current cycle experiment was performed in the voltage range of 0.005 – 2.0 V vs. Li/Li+ using the battery charge and discharge cycler (WBCS3000Le32, WonATech, Korea). To measure it, we charged it at the constant current of current density 80 mA g−1 (0.1 C) until it reached 40 mA g−1 (0.05 C) with constant voltage CC-CV mode at 0.005 V, and discharged it at the 80 mA g−1 (0.1 C) constant current mode.
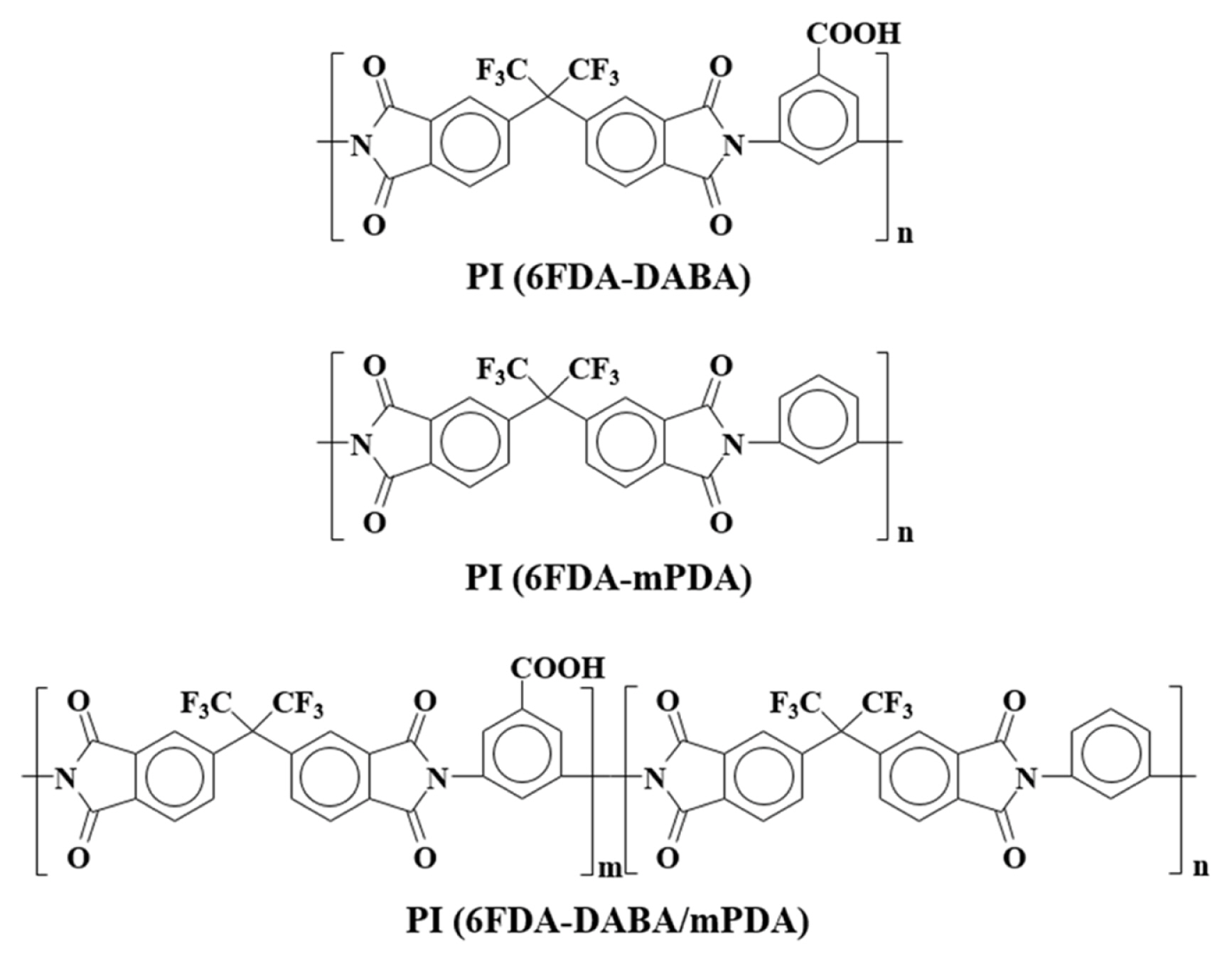
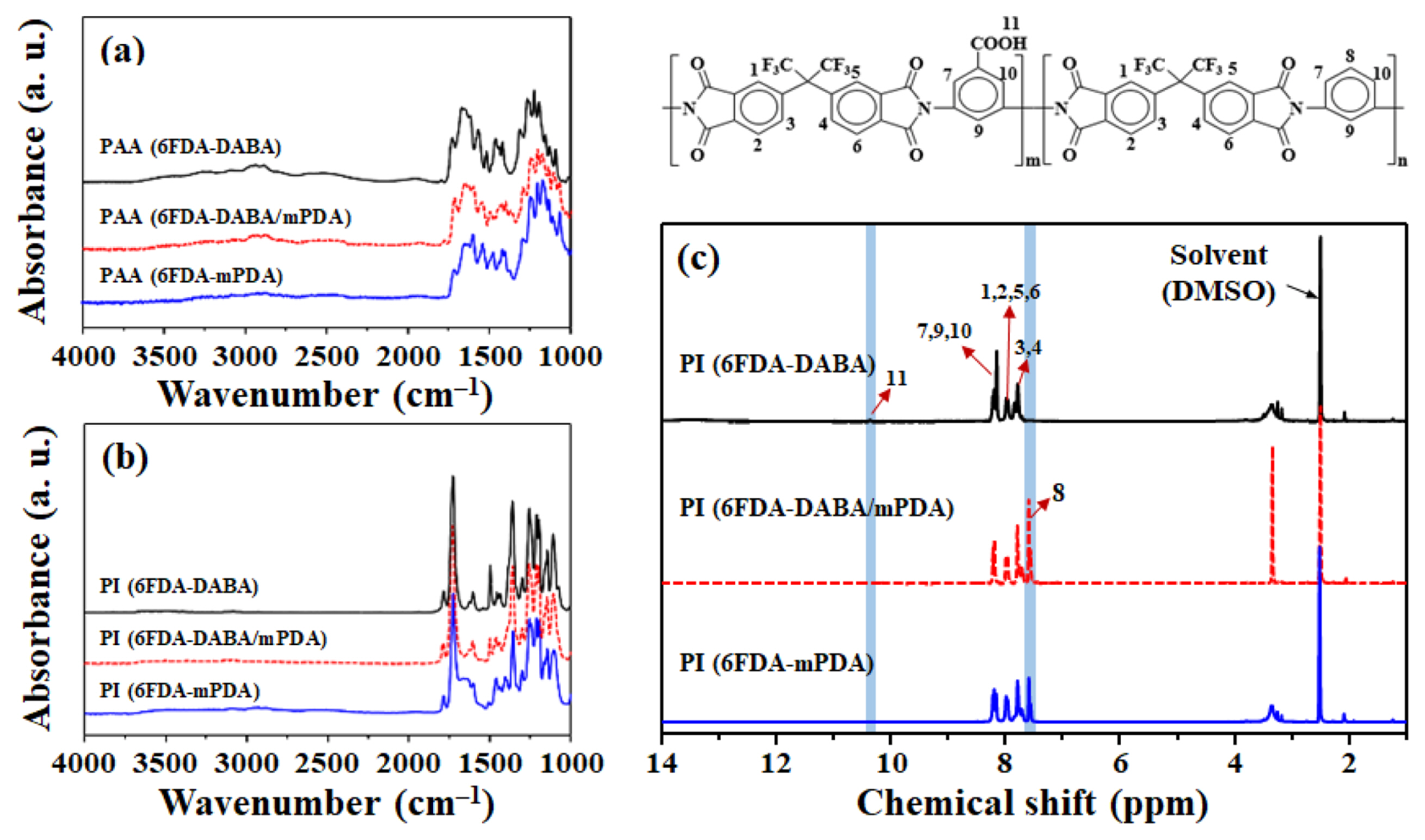
The synthetic scheme and chemical structure of the PI are presented in Figs. 1 and 2. The complete imidization of PAA to PI was confirmed by FT-IR and NMR spectroscopy. As shown in Fig. 3(a), all the PAA samples show characteristic peaks for amide groups such as C=O (1,666 cm−1), C-C (1,600 and 1,410 cm−1), and C-N (1,550 cm−1), respectively [14]. After imidization (Fig. 3(b)), these peaks were disappeared, while characteristic peaks for imide groups, such as C=O (1,780 and 1,721 cm−1), C-N (1,377 cm−1), and C-H (1,094 cm−1), appeared newly, indicating the successful imidization [15]. The 1H-NMR spectroscopy was performed to further confirm the imidization, and the results are shown in Fig. 3(c). All the peaks can be assigned clearly according to the integral values of the intensity and the 1H-NMR spectra completely agree with the proposed PI structures. These results confirmed the successful imidization of PAA to PI.
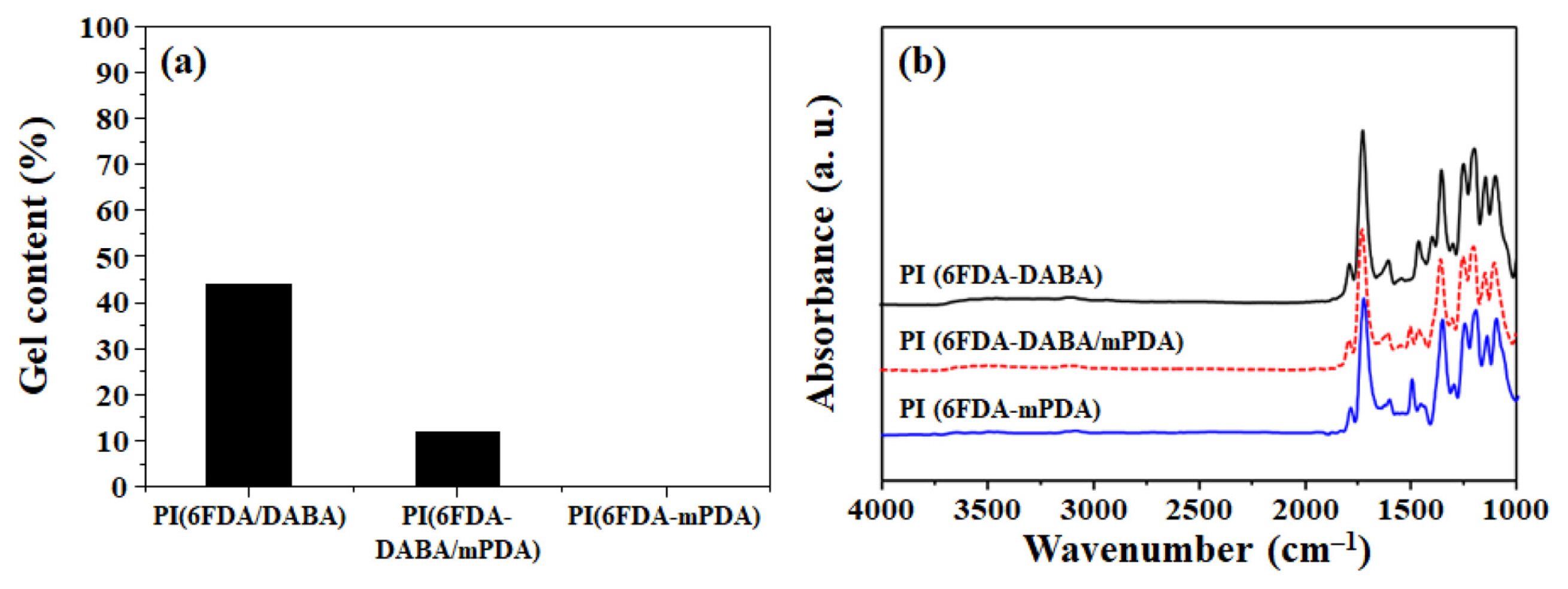
The decarboxylation-induced crosslinking of the PI was confirmed by gel content measurement, and FT-IR and TGA analyses, and the results are shown in Figs. 4 to 5. As shown in Fig. 4(a), the gel content increased from 0 to 44% with an increasing DABA content, indicating the crosslinking of the PI with DABA units. As can be seen in Fig. 4(b), the peak intensities for O-H band at 1260 cm−1 and C-O band of COOH groups at 1317 cm− 1 significantly decreased after thermal treatment. These results confirm the decarboxylation of the PI after thermal treatment. As seen in Fig. 5(a), there is the initial weight loss probable caused by the removal of residual solvents and moistures. Besides, DABA-containing PI shows significant weight loss in temperatures ranging from 100 to 400°C, caused by the decarboxylation. These results are generally observed for PI containing DABA units [16]. Above that temperature, the thermal decomposition of the PI backbones occurred. To further investigate the decarboxylation, TGA thermograms of the PI after thermal treatment at 400°C were obtained as shown in Fig. 5(b). The weight loss corresponding to the decarboxylation diminished after thermal treatment, indicating the complete decarboxylation and crosslinking [17].
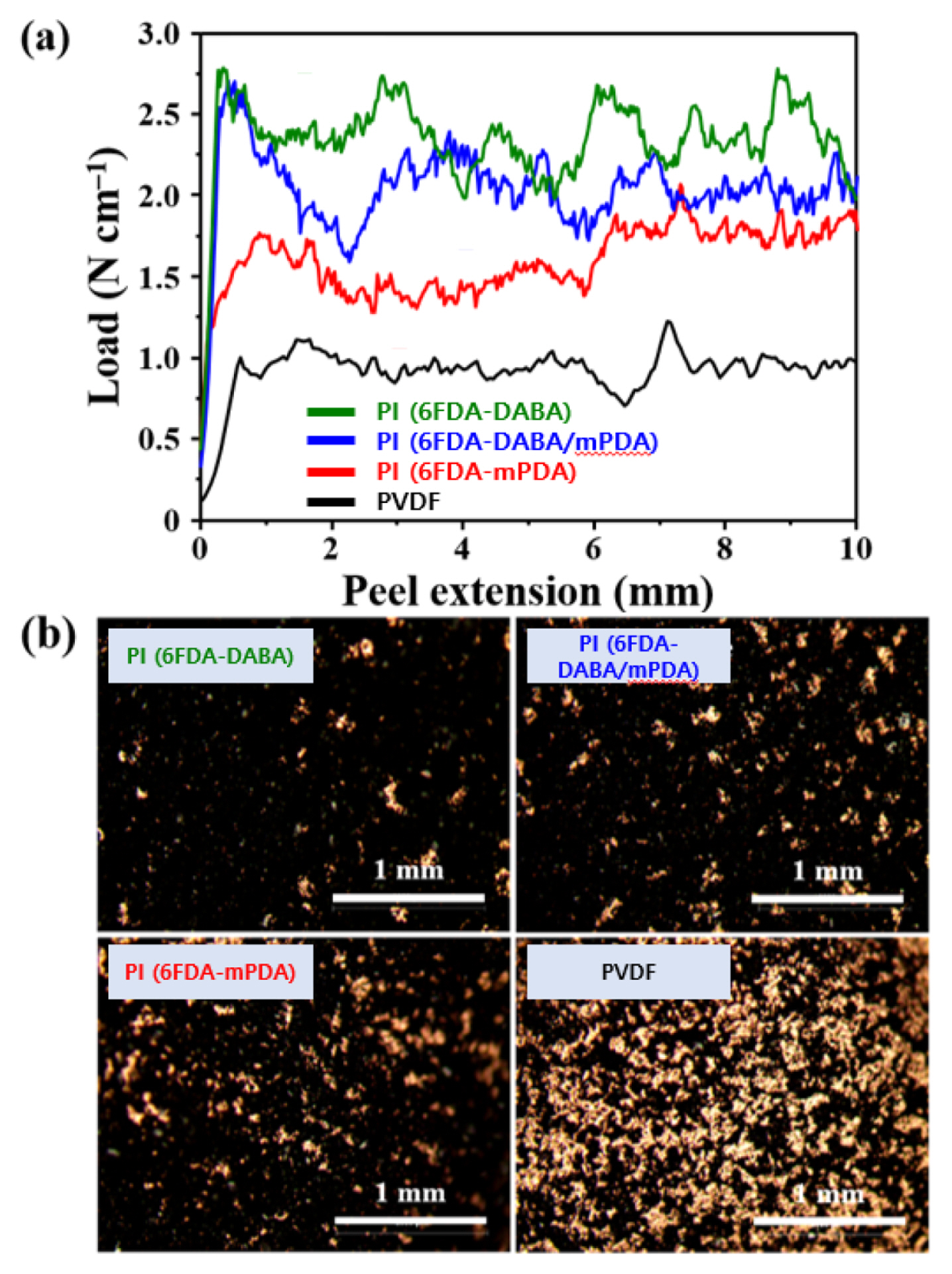
The peel strength of the anode with different binders was measured, and the results are shown in Fig. 6 and Table 1. As can be seen in Fig. 6(a), the anodes with the PI binders exhibit higher strength than the anode with PVDF binder. In addition, the strength increased with increasing the content of DABA possibly due to the strong interactions of the PI binders with the Si-alloy particles as well as the current collectors and the thermal crosslinking of the PI chains. Changes in the surface morphology of the anodes after peel test were observed by optical microscopy, and the results are shown in Fig. 6(b). The surface of the anodes can be divided into two regions. The black and gold-colored regions show the surfaces of the anode materials and Cu current collector, respectively. The anode with PVDF binder was almost detached from the Cu current collector due to the adhesion failure during the peel test. In contrast, the anode with PI (6FDA-DABA) binder was found to be well attached to the current collector due to the enhanced adhesion strength of the binder. These results revealed that the crosslinked PI (6FDA/DABA) binder enhanced the adhesion between active materials, conductive carbon particles, and current collectors, enabling the electrode to maintain good mechanical integrity of the anodes during cycling.
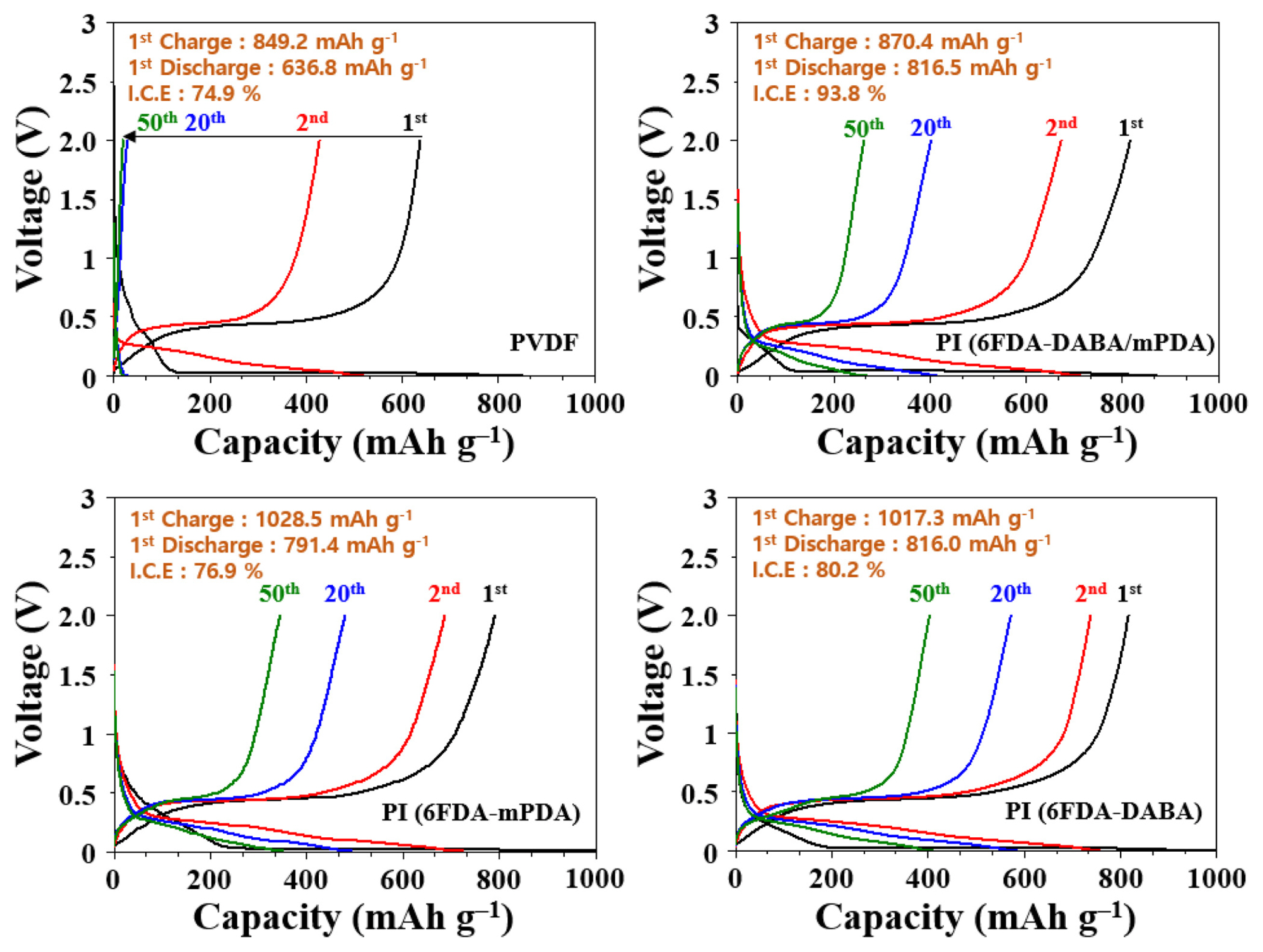
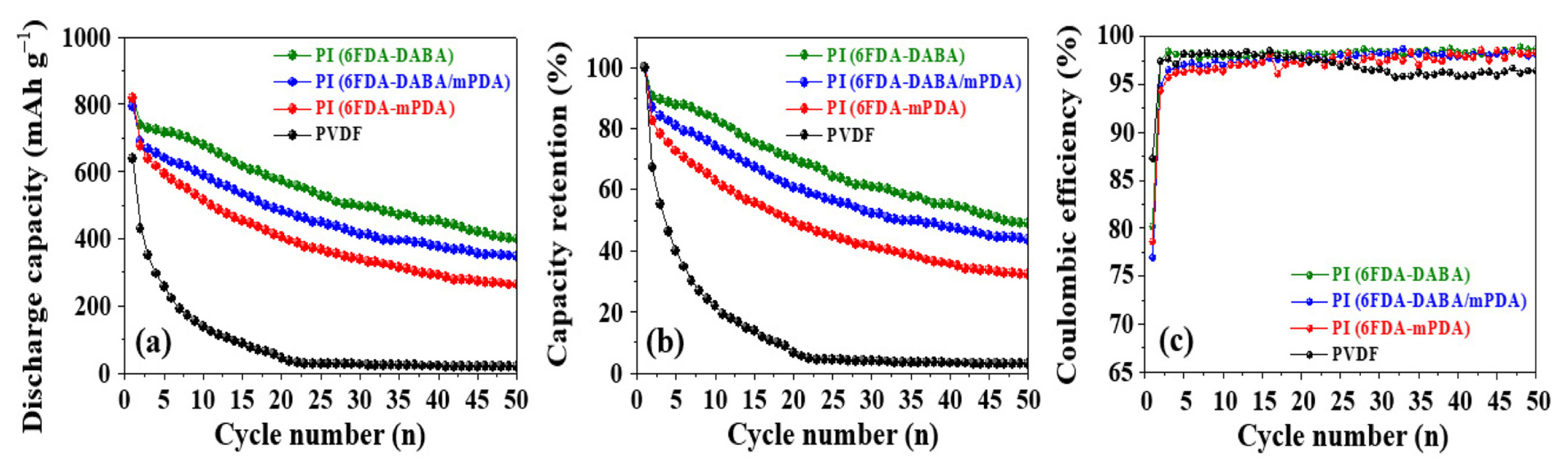
Galvanostatic measurements were conducted to compare the effect of binders on cell performance. The anodes with different binders showed different electrochemical performance in terms of initial discharge capacity and capacity retention. As shown in Fig. 7 and Fig. 8, the initial discharge capacity (636 mAh g−1 with an initial Coulombic efficiency (ICE) of 74.9%) of the anode with PVDF binder significantly decreased to 427 mAh g−1 after the 1st cycle. The initial discharge capacities of anodes with PI (6FDA-DABA/mPDA), PI (6FDA-mPDA), and PI (6FDA-DABA) binders were 816, 791, and 816 mAh g−1, respectively. Consequently, the ICE values were 93.8% (6FDA-DABA/mPDA), 76.9% (6FDA-mPDA), and 80.2% (6FDA-DABA). The ICE values for PI binders were higher than that of the PVDF binder, the binders. In addition, the PI binder containing DABA shows a higher ICE value. After 50 cycles, the capacity retentions of all the anodes are 48.7% PI (6FDA-DABA), 43.59% PI (6FDA-DABA/mPDA), 32.16% PI (6FDA-mPDA), and 2.94% (PVDF), respectively, indicating that the anode with a PI (6FDA-DABA) binder shows the superior electrochemical performance compared to other anodes.
Fig. 9 exhibits the differential capacity vs. potential plots (dQ/dV) after 1 cycle, 2 cycles, and 50 cycles, respectively. The sharp peaks centered at 0.45 V are due to the delithiation process, which is assigned to the presence of the crystalline Li15Si4 phase and a two-phase reaction with an amorphous Li~2Si [18]. After 50 cycles, the peaks located at a 0.45 V decreased, indicating the decrease in the discharge capacity due to the degradation of the PVDF anode. However, the peak of the PI (6FDA-DABA) anode at 0.45 V was well-maintained. These results indicate that the PI (6FDA-DABA) is an effective binder and good consistent with the results of adhesion strength (Fig. 6 and Table 1).
Fig. 10 shows rate capabilities of all the anodes in the range of 0.1 – 2 C-rate. Compared with anodes, the anode with a PI (DABA) binder has a highest reversible capacity. The discharge capacities of anode with a PI (DABA) binder were ~900 and ~600 mAh g−1 at C-rates of 0.1 and 2 C, respectively. These outstanding electrochemical behaviors of the anode with a PI (DABA) binder are mainly due to due to the superior mechanical integrity induced by highest adhesive strength.
Fig. 11 indicates the SEM results for anodes before and after 50 cycles. Fig. 11(a) is nothing wrong with the surface of the anode before cycle. In Fig. 11(b), the cracks were generated after 50 cycles due to microstructure change by the weak adhesion of binder. However, the PI (DABA) with an excellent adhesion strength shows the well-maintained microstructure without a crack after 50 cycles.
Thermally crosslinked PI binders were synthe-sized and characterized as robust binders for Si-alloy anodes in Li-ion batteries. The thermal crosslinking of the PI was confirmed by gel content measurement, and FT-IR and TGA analyses. The adhesion strength increased with an increasing DABA units in PI binders, leading to higher electrode integrity with better cycling stability. The Si-alloy anode with PI (6FDADABA) showed the highest electrochemical performance, such as Coulombic efficiency, capacity retention, and cycle stability. This strategy opens up the new design of crosslinked polymeric binders for high-performance anode materials in Li-ion batteries.
Acknowledgements
This work was supported by the Korea Institute of Energy Technology Evaluation and Planning (KETEP) grant funded by the Ministry of Trade, Industry & Energy (MOTIE) of the Republic of Korea (No. 20164010201070), and the Korea Institute for Advancement Technology (KIAT) grant funded by the Ministry of Trade, Industry & Energy (MOTIE) of the Republic of Korea (No. P0012748).
Fig. 6
(a) Peel strength results and (b) optical microscopic images of anodes with different binders.
References
[1] Q. Liu, S. Li, S. Wang, X. Zhang, Si. Zhou, Y. Bai, J. Zheng, X. Lu and . Jung, J. Phys. Chem. Lett, 2018, 9(18), 5567–5573.

[2] S. G. Ji, N. Umirov, H. Y. Kim and S. S. Kim, Journal of the Korean Battery Society, 2021, 1(1), 1–5.

[4] A. Gonzalez, N. Yang and R. Liu, J. phys. Chem, 2017, 121(150), 27775–27787.
[6] B. Koo, H. Kim, Y. Cho, K. T. Lee, N. -S. Choi and J. Cho, Angew. Chem. Int. Ed, 2012, 51(35), 8762–8767.

[7] J. Song, M. Zhou, R. Yi, T. Xu, M. Grodin, D. Tang, Z. Yu, M. Regula and D. Wang, Adv. Funct. Mater, 2014, 24(37), 5904–5910.

[8] Z. Liu, S. Han, C. Xu, Y. Luo, N. Peng, C. Qin, M. Zhou, W. Wang, L. Chen and S. Okada, RSC Adv, 2016, 6(72), 68371–38378.

[9] D. S. Lee, H. J. Park, A. Goliaszewski, Y. K. Byeun, T. S. Song and U. G. Paik, Ind. Eng. Chem. Res, 2019, 58(19), 8123–8130.

[10] H. S. Woo, K. I. Park, J. W. Kim, A. Yun, S. H. Nam and B. W. Park, Adv. Mater. Interfaces, 2020, 7(2), 1901475.

[11] S. H. Lee, J. H. Lee, D. H. Nam, M. S. Cho, J. H. Kim, C. Chanthad and Y. K. Lee, Appl. Mater. Interfaces, 2018, 10(19), 16449–16457.

[13] D. H. Seo, H. Y. Kim and S. S. Kim, J. Korean Inst. Electr. Electron. Mater. Eng, 2019, 32(4), 341–348.
[14] Y. J. Lee, B. N. Chae, Y. J. Park, Y. M. Jung and S. W. Lee, J. Mol. Struct, 2020, 1217, 128383.

- TOOLS
-
METRICS

- Related articles in J. Electrochem. Sci. Technol
-
Recent Progress on Polymeric Binders for Silicon Anodes in Lithium-Ion Batteries2015 June;6(2)