 |
 |
- Search
| J. Electrochem. Sci. Technol > Volume 13(4); 2022 > Article |
|
Abstract
A chemically sintered and binder-free paste of TiO2 nanoparticles (NPs) was prepared using a binary-liquid mixture of 1-octanol and CCl4. The 1:1 (v/v) complex of CCl4 and 1-octanol easily interacted chemically with the TiO2 NPs and induced the formation of a highly viscous paste. The as-prepared binary-liquid paste (PBL)-based TiO2 film exhibited the complete removal of the binary-liquid and residuals with the subsequent low-temperature sintering (~150°C) and UV-O3 treatment. This facilitated the fabrication of TiO2 photoanodes for flexible dye-sensitized solar cells (f-DSSCs). For comparison purposes, pure 1-octanol-based TiO2 paste (PO) with moderate viscosity was prepared. The PBL-based TiO2 film exhibited strong adhesion and high mechanical stability with the conducting oxide coated glass and plastic substrates compared to the PO-based film. The corresponding low-temperature sintered PBL-based f-DSSC showed a power conversion efficiency (PCE) of 3.5%, while it was 2.0% for PO-based f-DSSC. The PBL-based low- and high-temperature (500°C) sintered glass-based rigid DSSCs exhibited the PCE of 6.0 and 6.3%, respectively, while this value was 7.1% for a 500°C sintered rigid DSSC based on a commercial (or conventional) paste.
Since the invention of dye-sensitized solar cells (DSSCs) in 1991, extensive research has been performed to understand the operation principle and improve the power conversion efficiency (PCE) [1–3]. As a consequence, the PCE of over 14% was achieved for conventional glass/fluorine-doped tin oxide (FTO) substrate and high-temperature (500°C) sintered TiO2 photoanode-based rigid DSSCs (g-DSSCs) [4,5]. The fabrication of TiO2 photoanodes under low-temperature (150°C or lower temperature range) sintering conditions for DSSCs application has attracted substantial interest over the conventional high-temperature sintering process. This is advantageous not only for the fabrication cost reduction but also for developing mobile power sources for wearable electronic and IoT devices with light-weight and flexibility [6,7]. Additionally, the low-temperature fabrication method can be applicable for preparing multilayer structures with different dyes at each layer that can facilitate the PCE enhancement by solar spectrum splitting in the course of light passage through the photoanode [8].
The preparation of TiO2 photoanodes for conventional high-efficiency g-DSSCs fabrication requires the use of organic binders such as polyethylene glycol, ethylcellulose, and hydroxypropyl cellulose. These binders increase the viscosity of the TiO2 pastes and enable the preparation of highly stable, crack-free, and homogenous mesoporous TiO2 (mp-TiO2) films with a thickness of > 10 μm [9,10]. During the sintering process at 500°C, these organic binders are burned out and facilitate the formation of mp-TiO2 layers. In addition, this process induces the development of excellent electrical pathways for transporting photo-injected electrons by improving the interparticle connection of TiO2 and the adhesion of the TiO2 layer with glass/FTO substrates [9–11]. However, the conventional binder containing TiO2 pastes and the high-temperature sintering methods are unsuitable for fabricating transparent conducting oxide-coated plastic substrates-based flexible DSSC (f-DSSC) [8]. This is because flexible plastic substrates, indium tin oxide/polyethylene naphthalate (ITO/PEN) and ITO/polyethylene terephthalate (ITO/PET), cannot withstand a process at a temperature exceeding 150°C [12].
So far, mechanical pressing [13,14], electrophoretic deposition [15–17], microwave sintering [18], spray-deposition [19], chemical sintering [20,21], and film transfer (lift-off) [22] methods have been demonstrated for fabricating low-temperature processed TiO2 film onto the flexible substrates for DSSCs application. Among these methods, mechanical methods for TiO2 film fabrication required complicated multi-step fabrication processes with additional post-treatment, and they were generally ineffective for preparing a TiO2 film with a thickness of > 10 μm [7]. Additionally, these TiO2 film fabrication methods result in imperfect TiO2 NPs interconnections and poor adhesion of the TiO2 film with the flexible plastic substrates, resulting in inefficient charge transfer and collection in f-DSSCs [7,23]. Contrary to the mechanical methods, the solution-processed chemical sintering approach is promising for fabricating low-temperature sintered TiO2 pastes and the corresponding TiO2 films onto flexible plastic substrates with reduced fabrication steps, low cost, and high enough film thickness [21,23]. Furthermore, the chemical sintering approach offers the advantages of large-scale production of TiO2 film with improved mechanical stability [23]. Accordingly, chemical sintering approaches were reported to prepare binder-free TiO2 pastes using nano-glue (5 nm TiO2 particles), ethanol, ammonia, acid-base mixture, acids, and water [21,23–26].
Park et al. developed a binder-free TiO2 paste using acid-base chemistry of CH3COO− and NH4+ in solution [21]. The flocculation formation of TiO2 nanoparticles (NPs) induced by CH3COO− and NH4+ could enhance the viscosity by improving the interaction between the TiO2 NPs. The resulting binder-free and chemically sintered TiO2 photoanode exhibited a PCE of 2.55% in g-DSSCs [21]. In another report, Li et al. developed a nano-glue-based TiO2 paste with a well-interconnected NPs network [23]. The nano-glue acts as a chemical sintering agent for TiO2 NPs, in which the ample surface hydroxyl groups (-OH) of nano-glue are well mixed with the TiO2 NPs and increase the viscosity of the paste. The corresponding low-temperature sintered DSSC, fabricated with a plastic substrate-based TiO2 photoanode and a platinized glass/FTO substrate-based counter electrode (CE), exhibited a PCE of 5.43%.
Strong acids such as hexa-fluorotitanic acid (H2TiF6) and hydrochloric acid (HCl) were used for preparing low-temperature chemically sintered TiO2 pastes for f-DSSCs [25,26]. The oxidizing nature of acids can increase the amounts of -OH groups on the TiO2 surface and improve the TiO2 interparticle connection with the subsequent dehydration reaction at the low-temperature sintering conditions. Holliman et al. prepared a low-temperature sintered TiO2 paste using H2SiF6 as a chemical sintering agent with a PCE of 4.2% in DSSC based on ITO/PET photoanode substrate and glass/FTO based CE substrate [26]. In another report, Weerasinghe et al. used HCl as a chemical sintering agent for preparing TiO2 film with a PCE of 5% in f-DSSC [26]. Although these acids pastes-based TiO2 films showed good adhesion with the substrates, the possible chemical etching of the ITO and FTO conducting layer by the H2TiF6 and HCl can lower the fill factor (FF) and photocurrent of the DSSCs devices [26]. Therefore, it is essential to develop ITO and FTO corrosion-resistive TiO2 pastes, which exhibit high enough viscosity with sufficient mechanical strength and thickness of the TiO2 layers and strong adhesion to the conductive flexible plastic substrates after low-temperature sintering.
In this study, a novel binary-liquid mixture of carbon tetrachloride (CCl4) and 1-octanol was used for preparing chemically sintered low-temperature TiO2 paste for f-DSSCs. The unfavorable physical and chemical properties of CCl4, including high volatility, low-boiling temperature (Tb =76.72°C), and low viscosity (0.965 cP), were not suitable for the preparation of TiO2 pastes. Thus, 1-octanol with high Tb (194.5°C) and high-viscosity (7.36 cP) was introduced into the CCl4 for preparing highly viscous, binder-free, and chemically and low-temperature sintered TiO2 pastes. According to Raoult’s law [27], the high Tb of 1-octanol could decrease by the low Tb of CCl4 and facilitate the preparation of TiO2 films sintered at low temperature (≤ 150°C). The formation of the 1-octanol-CCl4 complex could easily interact with the TiO2 NPs via hydrogen bonding. This induced the reduction of TiO2 interparticle distance and enabled the preparation of high-viscous pastes. The as-prepared chemically sintered and low-temperature TiO2 films exhibited good inter-particle connection and porosity, improved mechanical strength, and strong adhesion to the ITO/PEN and glass/FTO substrates. The 150°C sintered g-DSSCs and f-DSSCs fabricated with this binary-liquid paste exhibited PCEs of 6.0 and 3.5%, respectively.
All the reagents and solvents were purchased from Sigma-Aldrich (St. Louis, MO, USA). Cis-diisothiocyanato–bis(2,2′-bipyridyl-4,4′-dicarboxylato) ruthenium (II) bis(tetrabutylammonium) (N719) dye was purchased from Solaronix, Switzerland. ITO/PEN (13 Ω/sq.) and glass/FTO (8 Ω/sq.) were purchased from Peccell Technologies Inc (Japan) and Pilkington (USA), respectively.
Before preparing the TiO2 pastes, the TiO2 powder (Degussa P25) was pre-treated with acetylacetone to minimize NPs aggregation [9]. Then, 1 g of pretreated TiO2 powder was mixed with two compositions of 1-octanol and CCl4 (1:1 and 1:0 (v/v)) and stirred at room temperature (RT) for 6 h. Subsequently, the mixture was homogenized for another 30 min to obtain homogenous dispersion of TiO2 NPs by using an ultrasonic horn. The mixture was heated again at 70°C with stirring (300 rpm/s) for 24 h to obtain the high viscous pastes. The pastes were denoted hereafter as PBL and PO, where the composition of 1-octanol and CCl4 were 1:1 and 1:0 (v/v), respectively.
The as-prepared TiO2 pastes were coated on flexible ITO/PEN and glass/FTO substrates by the doctor blade method and dried at RT. Then, the TiO2 films were sintered at 150°C for 60 min in an electric oven in the presence of air. Subsequently, the films were treated with UV-O3 for 30 min. For comparison, the PBL and a commercial binder containing TiO2 paste (PC) (TTPH-20N, ENBKOREA Co., Ltd., Korea, particle size 20 nm) were used to prepare 500°C sintered TiO2 films on glass/FTO substrate according to a previously reported method [28]. The thickness of the PBL and PO-based TiO2 films was ca. 15.8 and 15.3 μm, respectively, while it was ca. 15.4 μm for PC-based TiO2 photoanode. All the TiO2 electrodes were dipped into the N719 dye solution (0.3 mM) in ethanol for 18 h. For the preparation of f-DSSCs CE, Pt was sputtered on ITO/PEN substrate for about 10 s. While for the preparation of g-DSSCs CEs, 5 mM chloroplatinic acid hexahydrate (H2PtCl6·6H2O) solution in ethanol was drop cast onto the glass/FTO substrate and sintered in an electric muffle furnace at 400°C for 20 min in air. The dye-loaded photoelectrodes (active area ca. 0.25 cm2) and Pt-CEs were sandwiched using 60-μm thick Surlyn film (Solaronix SA, Switzerland) as a spacer and sealing agent using a hot press. The electrolyte solution with the composition of 0.6 M 1,2-dimethyl-3-propylimidazolium iodide (DMPII), 0.1 M LiI, 0.1 M I2, and 0.5 M 4-tert-butylpyridine (tBP) and 1 mM 4′,6-diamidino-2-phenylindole (DAPI) as an efficient energy relay dye (ERD) [29] were dissolved in 3-methoxypropionitrile (MPN) and injected into the cells through the drilled holes on the CEs. The holes were sealed by using surlyn film with cover glass.
The viscosity of the TiO2 pastes was measured with a viscometer (μVisc, Rheosense, Inc., USA). The thickness of the TiO2 films was measured by a surface profilometer (Accretech, Japan), and the adhesion of TiO2 film with FTO was evaluated by a scotch-tape scratch test. The strength of adhesion of the films was evaluated using a peel-off tester (Top-Tac 2000, Korea). The crystallographic pattern of TiO2 was characterized with an X-ray diffractometer (XRD, Philips, X’pert, Netherland) using Cu Ka radiation. A field-emission scanning electron microscope (FE-SEM, JSM-6700F, JEOL) equipped with energy-dispersive X-ray spectroscopy (EDS) was used (EDS, INCAx-sight7421, Oxford Instruments) to characterize the surface morphology and elemental analysis of TiO2 films. The amount of adsorbed dye onto the TiO2 film was determined by desorbing the dye in a 0.1 M NaOH aqueous solution, and the absorption spectra of the desorbed dye solutions were measured using a UV–Vis absorption spectrophotometer (Perkin-Elmer Lamda 35). A solar simulator (Polaronix® K201, McScience, Korea) with a 200 W Xenon lamp with the incident light intensity of 100 mW/cm2 (AM 1.5 G) was used for the photovoltaic (PV) measurements of the DSSCs. A PV power meter (Polaronix® K101 LAB20, McScience, Korea) was used to measure the current density-voltage (J-V) characteristics of DSSCs. Incident photon-to-electron conversion efficiency (IPCE) spectra were obtained by an IPCE measurement system (Polaronix, K3100 Spectral IPCE, McScience) with a 300 W xenon lamp source. Electrochemical impedance spectra (EIS) were measured using an impedance analyzer (IM6ex, Zahner Zahner-Elektrik GmbH & Co. KG, Germany) in the frequency range from 105 to 0.1 Hz with the ac amplitude of 5 mV under open-circuit potential and dark conditions. The EIS spectra were analyzed with an appropriate equivalent circuit using Z-view software (Scribner Associates Inc., version 3.1). Thermogravimetric (TGA) analyses were performed with a Scinco TGA-N 1000 analyzer (Seoul, Korea) under ambient atmospheric conditions.
Fig. S1 presents the bar diagram of the Tb variation of the CCl4 and 1-octanol mixture against their volume fraction. The Tb of the binary mixture was increased by increasing the volume fraction of 1-octanol that is in accordance with Raoult’s law [27]. Considering the preparation of binder-free, low-temperature chemically sintered TiO2 paste, and the composition of CCl4 and 1-octanol complex formation [30], a 1:1 volume ratio is selected as the optimized composition, where the Tb of 1-octanol:CCl4 (1:1 v/v) was 97°C. The schematic illustration of the TiO2 NPs inter-connections in the mp-TiO2 film based on PBL is presented in Scheme 1. In the mixture of 1-octanol, CCl4, and TiO2 NPs, 1-octanol and CCl4 could easily form a complex with a 1:1 (v/v) ratio via hydrogen bonding (O-H·····C1-C) [30]. Subsequently, this complex could interact with the -OH groups on the TiO2 NPs surface and form Ti-O-C-O-C8H17 and Ti-O-C-O-Ti bonds by the elimination of HCl. This enabled to decrease in the TiO2 interparticle distance and formed a viscous paste. In the subsequent heat treatment of the as-prepared TiO2 NPs film at 150°C, CO2 and H2O can be eliminated by the decomposition of hydrocarbon chains and the dehydration of -OH groups on the surfaces of TiO2 NPs, respectively. This induces to interconnect the TiO2 NPs through the Ti-O-C-O-Ti bonds. Finally, upon the UV-O3 treatment, Ti-O-C-O-Ti bonds can be broken and the residual carbon can be eliminated in the form of CO2 and O2 [31], resulting in the further reduction of TiO2 interparticle distance and forming a highly stable mp-TiO2 network through Ti-O-Ti bonds. This induces the decoloring of the film, which can be attributed to the complete removal of the remaining organic residues, as shown in Fig. S2. Meanwhile, for the case of PO, TiO2 NPs are homogeneously distributed into the 1-octanol due to the physical adsorption as well as the hydrogen bonding interaction between the -OH groups of TiO2 and 1-octanol. Upon heat treatment of the as-prepared PO-based TiO2 film at 150°C, 1-octanol can effectively bridge the gap between TiO2 NPs by dehydration process. Finally, in the UV-O3 treatment, the residual 1-octanol and carbon can be eliminated as CO2 and O2 (Fig. S2), thus, facilitating the formation of TiO2 film with moderate stability.
Fig. 1a shows the viscosity of the TiO2 pastes (PBL, PO, and PC) and the mechanical strength of their corresponding TiO2 films prepared onto the glass/FTO substrates by the doctor blade method. The corresponding photo-image of PBL and PO pastes is presented in Fig. 1b. The rheological behavior of the TiO2 pastes revealed that the addition of CCl4 with 1-octanol significantly increased the viscosity of the TiO2 paste from 38,214 (PO) to 53,158 (PBL) cPs. This can be attributed to the hydrogen bonding between the TiO2 NPs and the complex of CCl4 and 1-octanol [30,32], which concurrently increase the maximum strength of adhesion of the 150°C sintered TiO2 film from 192 (PO) to 229 (PBL) gf/mm2. These values were 98245 cPs and 282 gf/mm2 for PC under 500°C sintering conditions. These results are consistent with the tape test results for the mechanical adhesion of PBL compared to PO (Fig. S3) on a glass/FTO substrate upon sintering at 150°C followed by subsequent UV-O3 treatment. PBL showed the highest level of adhesion without affecting the cross-cut areas (~0%), while PO exhibited a much lower degree of adhesion by detaching the cross-cut areas of ~10%.
The morphology of TiO2 films prepared from the PBL and PO was studied with FE-SEM. The TiO2 film of PBL, before heat treatment at 150°C, exhibited crack-free homogenous distribution of TiO2 NPs with a relatively homogenous distribution of particle aggregation (Fig. S4a). In contrast, the TiO2 film of PO showed some cracks over the TiO2 film with an enhanced particle aggregation by forming bigger local clusters over the film (Fig. S4b). After sintering at 150°C and subsequent UV-O3 treatment, the TiO2 film of PBL showed high porosity without the presence of NPs aggregation (Fig. 2a), while the TiO2 film of PO exhibited lower porosity with the presence of bigger local clusters over the film (Fig. 2b). The carbon content in the TiO2 film of PBL was analyzed by EDS under different treatment conditions (e.g., drying at 25°C, 150°C, 150°C followed by UV-O3 treatment, and 500°C), as shown in Fig. 2c. The corresponding EDS spectra are presented in Fig. S5. It was observed that the carbon content in PBL decreased significantly after both sintering at 150°C and subsequent UV-O3 treatment with a carbon content of 1.76%. This value for the RT dried and 150°C sintered PBL-based TiO2 films was 3.0 and 2.48%, respectively. Further, a small reduction of the carbon content at 500°C sintering condition (1.47 wt%) evidently showed that the additional UV-O3 treatment is very critical to eliminating the carbon residues after sintering at relatively low temperatures. The thermogram in Fig. 2d exhibited a continuous weight loss up to 62 and 51.75%, respectively, for PBL and PO at 138°C, while no further significant weight loss was observed until 500°C (63.50 and 53.15%, respectively, for PBL and PO), consistent with the previous weight loss data. The little high residual percentage of PO compared to PBL can be ascribed to the incomplete removal of 1-octanol from PO induced by its high Tb (194.5°C). These results demonstrated that both PBL and PO pastes are suitable for the preparation of binder-free f-DSSCs photoanodes under low-temperature sintering conditions with subsequent UV-O3 treatment. Additionally, the crystallographic pattern of the TiO2 NPs in the PBL and PO-based TiO2 films exhibited that anatase is the major crystal phase (JCPDS card no. 21-1272) (Fig. S6) [33]. This is advantageous for improved dye-loading and the development of high-performance DSSCs [34].
Fig. 3a presents the photo-current density-voltage (J-V) plots of the g-DSSCs based on the TiO2 photoanodes of PBL and PO, sintered at 150°C. The resulting PV parameters are summarized in Table 1. It was observed that the PO cell showed the lowest PV performance with the short-circuit current density (Jsc), open-circuit potential (Voc), and PCE of 8.45 mA/ cm2, 0.73 V, and 4.2%, respectively. Meanwhile, the PBL cell exhibited the PCE enhancement of 30% compared to the PO cell with the Jsc, Voc, and PCE of 12.07 mA/cm2, 0.73 V, and 6.0%, respectively. The increase in the PCE of PBL cell compared to the PO device is mostly due to the increase in Jsc. This can be attributed to the improved dye loading in PBL (2.51 × 10−8 mol/cm2) compared to the PO (ca. 1.8 × 10−8 mol/ cm2) (Fig. S7a). The high visible transparency of the TiO2 film of PBL compared to the PO film (Fig. S7b) is another possible reason for increased Jsc. Meanwhile, the 500°C sintered PBL-based g-DSSC exhibited a PCE of 6.3%, which is comparable to the PCE of PC cell (7.10%) (Fig. 3b and Table 1). The improved PV performance of the PC-based g-DSSCs is due to the enhanced TiO2 interparticle connection compared to PBL, which corroborates a 24.32% higher Jsc. This result suggests that PBL is promising to develop conventional high-temperature sintered g-DSSCs.
Finally, f-DSSCs were fabricated by utilizing PBL and PO on ITO/PEN substrate, and the resulting J-V plots and PV parameters are summarized in Fig. 3c and Table 1, respectively. Similar to the g-DSSCs, the fully flexible cell with PBL exhibited a PCE of 3.5% with the Jsc, Voc, and FF of 9.42 mA/cm2, 0.70 V, and 52.3%, respectively, while the PCE was 2.0% for PO with the Jsc, Voc, and FF of 6.42 mA/cm2, 0.68 V, and 46.8%, respectively. By inspection, the 75% PCE enhancement for PBL-based f-DSSC compared to the PO device is mainly due to the significant increase of Jsc (46.72% enhancement). This can be attributed to the improved TiO2 interparticle connection and dye loading of the PBL-based TiO2 film compared to the TiO2 film based on PO. Nevertheless, The PCE of the PBL-based f-DSSC is 71.40% lower than the g-DSSC prepared under similar conditions. This can be ascribed to the high ohmic resistance of the ITO/PEN substrate as well as the internal resistance of the devices, which corroborated a significantly low FF. The PCE of the PBL DSSC is higher or comparable to the previously reported g-DSSCs and f-DSSCs prepared under low-temperature sintering conditions (Table S1), suggesting that PBL can be effectively utilized for developing high-performance DSSCs with additional electrode engineering. Fig. 3d shows the IPCE spectra of the corresponding f-DSSCs based on PBL and PO. It can be observed that the external quantum efficiency (EQE) of the PBL cell exhibited an upward shift over the wide wavelength range (400–800 nm) with the maximum EQE value of 54% at 540 nm, while this value for the PO device was 41% at 540 nm. This result is consistent with the Jsc values of the corresponding f-DSSCs devices.
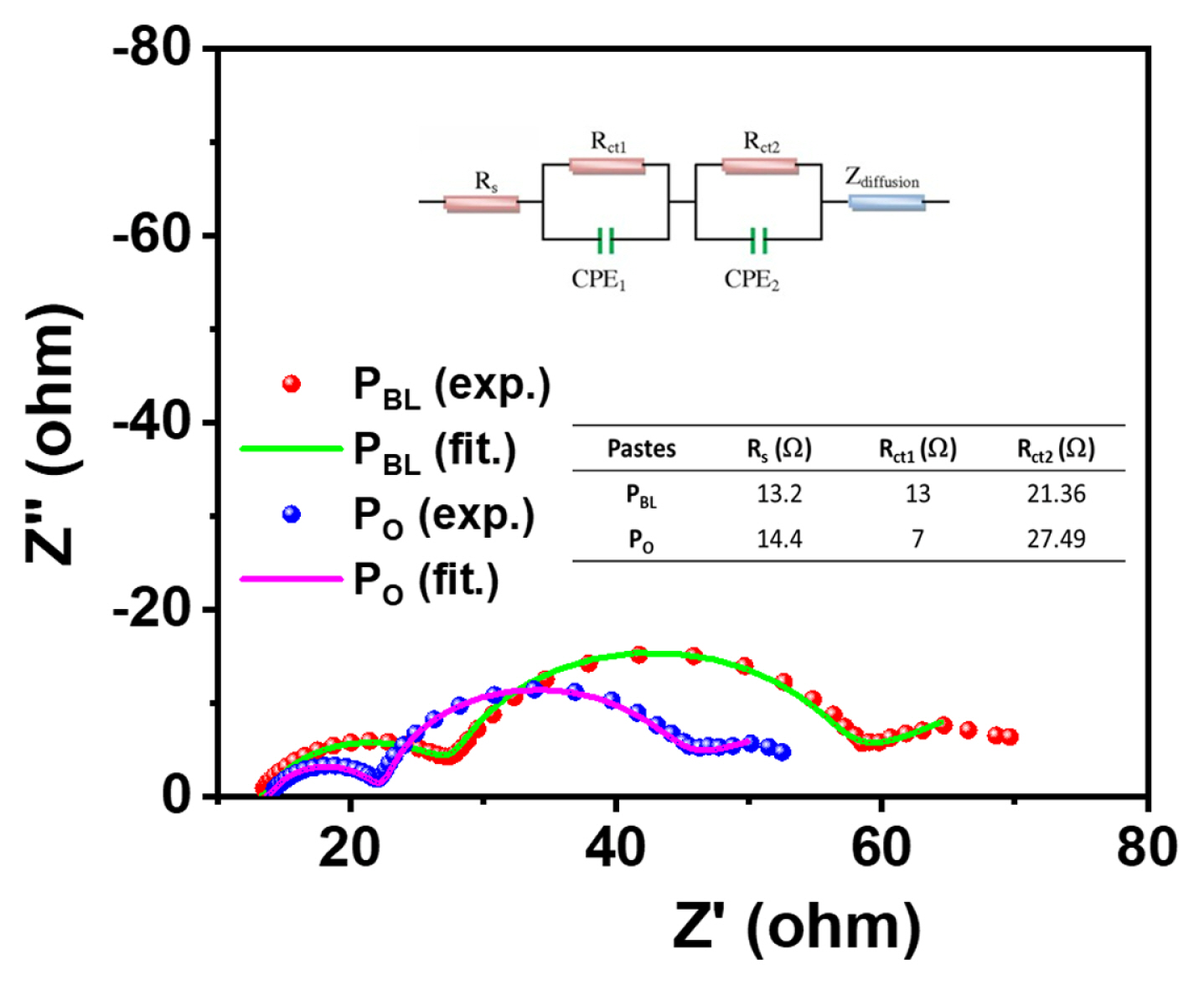
EIS spectra of the PBL and PO-based f-DSSCs were measured to investigate the electron transport mechanism and the interfacial properties. Fig. 4 shows the EIS spectra of the devices in the form of Nyquist plots, which exhibited three semicircles. The semicircle at the high-, mid-, and low-frequency regions corresponds to the charge transfer resistance at the CE/ electrolyte interface (Rct1), charge transfer resistance at the TiO2/dye/electrolyte interface (Rct2), and the diffusion resistance (Zdiffusion) within the electrolyte, respectively [35]. The intercept with the real axis at the highfrequency region corresponds to the contact resistance (Rs) between the TiO2/substrate or the ohmic resistance of the cells [35,36]. Generally, the Rs values do not alter much in a DSSC with similar substrates and paste. However, a little lower Rs for the PBL cell (13.2 W) than the PO-based f-DSSC (14.4 W) suggests the lower contact resistance between the PBL TiO2 film and ITO/PEN substrates. This can be attributed to the strong adhesion of the PBL TiO2 film with the substrate [36], consistent with the scotch tape and mechanical adhesion tests. The Rct2 values for PBL and PO devices are 21.36 and 27.49 W, respectively. The lower Rct2 designates the faster interfacial charge transfer process and lower recombination in the PBL device compared to the PO device [35], which is consistent with the PV parameters of their corresponding f-DSSC.
This study described the utilization of 1-octanol with high viscosity and high Tb together with CCl4 with low viscosity and low Tb to prepare a binder-free TiO2 paste for low-temperature sintered f-DSSCs. The 1:1 (v/v) complex of CCl4 and 1-octanol could interact with the TiO2 NPs via hydrogen bonding, which reduced the TiO2 interparticle distance and formed a vicious TiO2 paste. This binary-liquid-based TiO2 paste, PBL, exhibited strong adhesion to the FTO/glass and ITO/PEN substrates with high mechanical stability. The PBL-based f-DSSC and g-DSSC, sintered at 150 and 500°C, respectively, exhibited PCEs of 3.5 and 6.3%, respectively. The PCE for f-DSSC and g-DSSC based on pure 1-octanol paste and sintered at 150°C, were 2.0 and 4.0%, respectively, while this value for commercial 500°C sintered PC-based g-DSSC was 7.1%. The significantly improved PCE of PBL-based f-DSSC and g-DSSC compared to the pure 1-octanol paste-based cells can be attributed to the strong adhesion and high mechanical stability of the corresponding TiO2 film with improved porosity and dye loading. This result demonstrated the applicability of the PBL paste for the development of both low- and high-temperature sintered DSSCs. We strongly believe that the present methodology is promising for developing high-efficiency f-DSSCs by choosing other highly viscous alcohols and low boiling temperature liquids.
Supporting information
Supporting Information is available at https://doi.org/10.33961/jecst.2022.00262
Acknowledgements
This research was supported by the Technology Development Program to Solve Climate Changes of the National Research Foundation, funded by the Ministry of Science, ICT & Future Planning (NRF-2016M1A2A2940912).
Fig. 1
(a) Viscosity of PBL, PO, and PC pastes and the maximum strength of adhesion of their corresponding TiO2 films onto glass/FTO substrates. (b) The photographic images of the as-prepared PBL and PO.
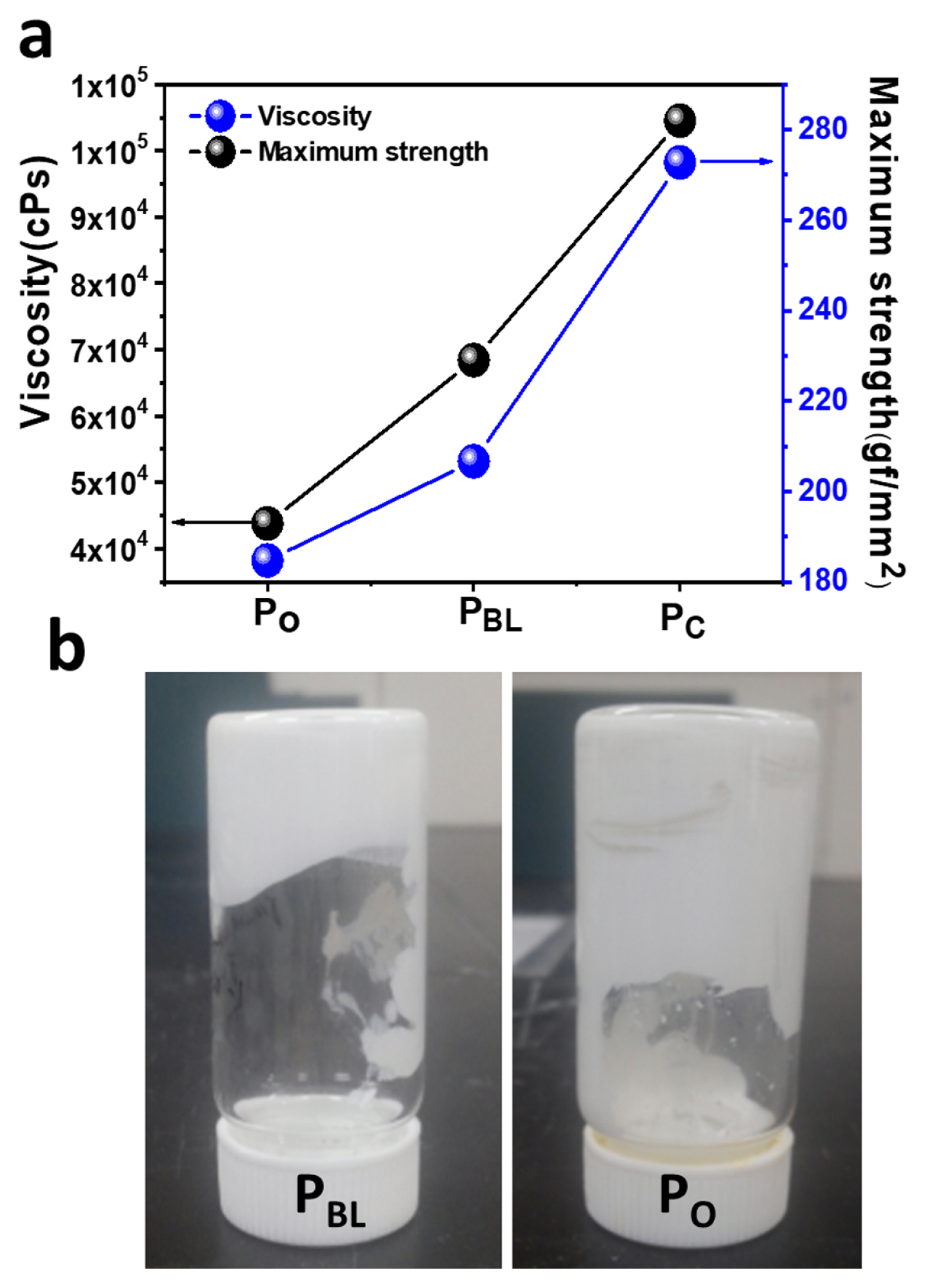
Fig. 2
Top-view FE-SEM images of (a) PBL and (b) PO pastes-based TiO2 films after sintering at 150°C and subsequent UV-O3 treatment. (c) Weight (%) of carbon in the TiO2 film of PBL with different treatment conditions. (d) TGA curve of RT dried TiO2 pastes with a heating range of 3°C/min.
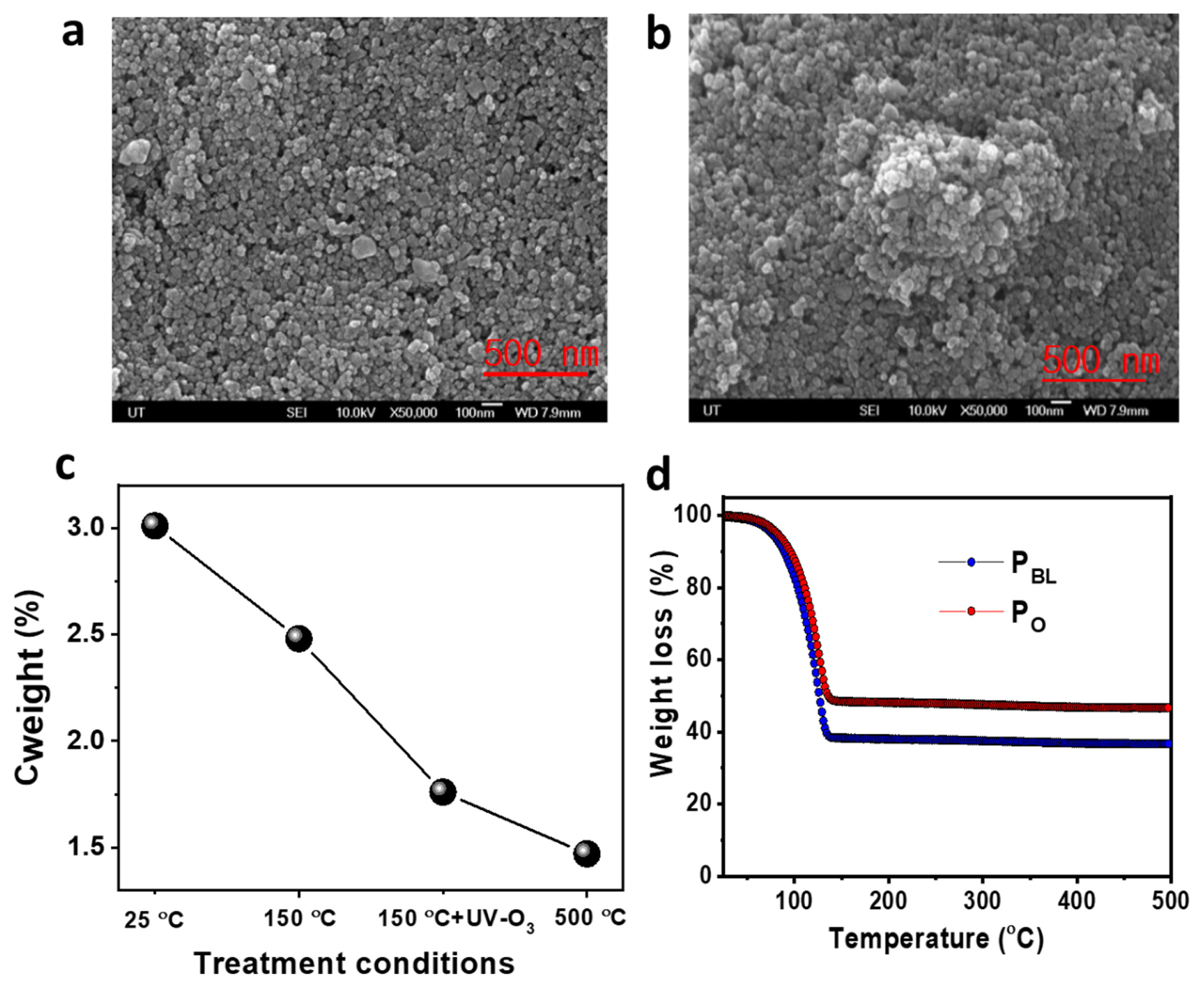
Fig. 3
Photo-current density (J) - voltage (V) characteristics of different TiO2 paste based g-DSSCs sintered at (a) 150°C and (b) 500°C, and (b) f-DSSCs sintered at 150°C. (d) The IPCE spectra of the corresponding f-DSSCs.
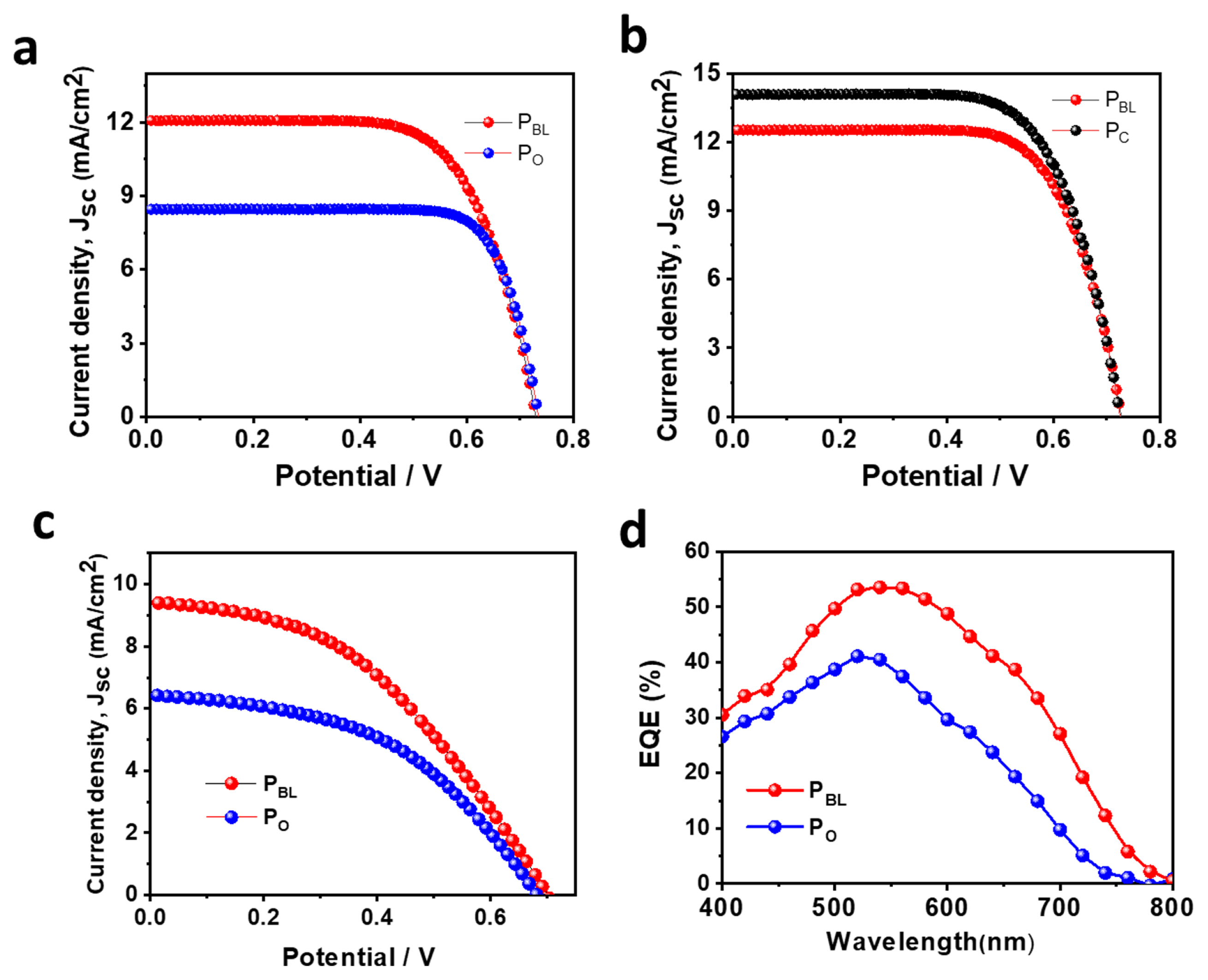
Fig. 4
Nyquist plots of f-DSSCs measured under open circuit and dark conditions. The upper inset shows the equivalent circuit model to fit the spectra, and the table in the lower inset displays the fitted results of EIS parameters.
Scheme 1
Schematic illustrations of the TiO2 interparticle connection in binary liquid-based TiO2 paste via a dehydration process at 150°C and subsequent UV-O3 treatment.
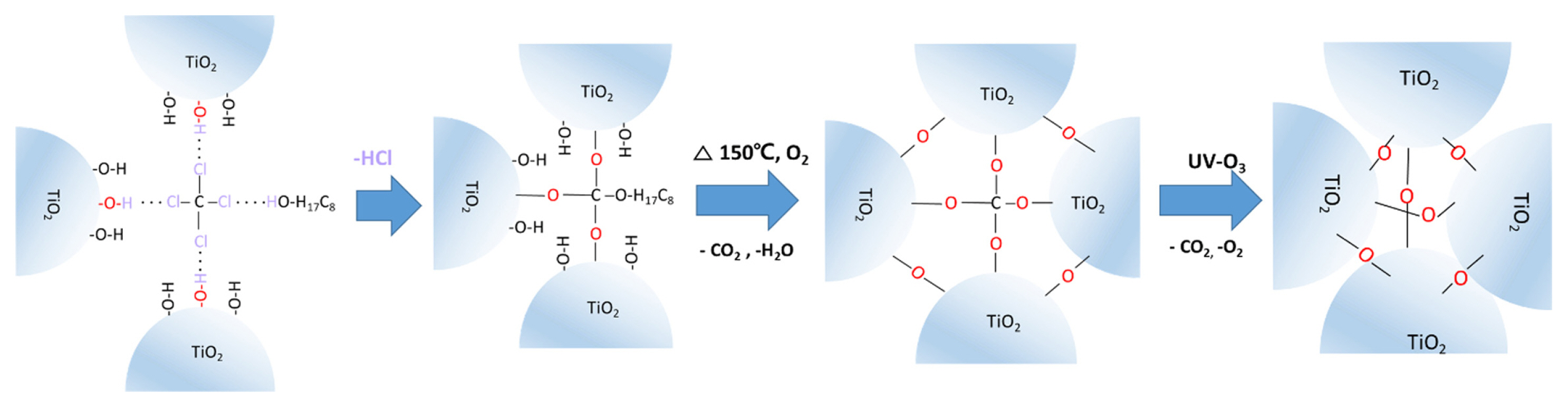
Table 1
Photovoltaic parameters of different TiO2 pastes based DSSCs.
References
[2] K. Sharma, V. Sharma and S.S. Sharma, Nanoscale Res. Lett, 2018, 13, 381.
[4] K. Kakiage, Y. Aoyama, T. Yano, K. Oya, J. Fujisawa and M. Hanaya, Chem. Commun, 2015, 51, 15894–15897.

[8] K. Kim, G.W. Lee, K. Yoo, D.Y. Kim, J.K. Kim and N.G. Park, J. Photochem. Photobiol. A: Chem, 2009, 204(2–3), 144–147.

[9] S. Sarker, N.C.D. Nath, M.M. Rahman, S.-S. Lim, A.J.S. Ahammad, W.-Y. Choi and J.-J. Lee, J. Nanosci. Nanotechnol, 2012, 12(7), 5361–5366.

[10] M.M. Rahman, H.-Y. Kim, Y.-D. Jeon, I.-S. Jung, K.-M. Noh and J.-J. Lee, Bull. Korean Chem. Soc, 2013, 34(9), 2765–2768.

[11] M.M. Rahman, H.-S. Son, S.-S. Lim, K.-H. Chung and J.-J. Lee, J. Electrochem. Sci. Technol, 2011, 2(2), 110–115.


[12] H.-P. Jen, M.-H. Lin, L.-L. Li, H.-P. Wu, W.-K. Huang, P.-J. Cheng and E.W.G. Diau, ACS Appl. Mater. Interfaces, 2013, 5(20), 10098–10104.

[13] T. Yamaguchi, N. Tobe, D. Matsumoto, T. Nagai and H. Arakawa, Sol. Energy Mater. Sol. Cells, 2010, 94(5), 812–816.

[14] G. Boschloo, H. Lindstrom, E. Magnusson, A. Holmberg and A. Hagfeldt, J. Photochem. Photobiol. A, 2002, 148(1–3), 11–15.

[16] L. Grinis, S. Kotlyar, S. Ruhle, J. Grinblat and A. Zaban, Adv. Funct. Mater, 2010, 20(2), 282–288.

[17] H.W. Chen, C.P. Liang, H.S. Huang, J.G. Chen, R. Vittal, C.Y. Lin, C.W.W. Kevin and K.C. Ho, Chem. Commun, 2011, 47, 8346–8348.

[18] S. Uchida, M. Tomiha, H. Takizawa and M. Kawaraya, J. Photochem. Photobiol. A, 2004, 164(1–3), 93–96.

[20] X. Li, H. Lin, J. Li, N. Wang, C. Lin and L. Zhang, J. Photochem. Photobiol. A, 2008, 195(2–3), 247–253.

[21] N.-G. Park, K.M. Kim, M.G. Kang, K.S. Ryu, S.H. Chang and Y.-J. Shin, Adv. Mater, 2005, 17(19), 2349–2353.

[22] M. Dürr, A. Schmid, M. Obermaier, S. Rosselli, A. Yasuda and G. Nelles, Nat. Mater, 2005, 4, 607–611.
[24] J.H. Yune, I. Karatchevtseva, G. Triani, K. Wagner and D. Officer, J. Mater. Res, 2013, 28(3), 488–496.

[25] H.C. Weerasinghe, P.M. Sirimanne, G.V. Franks, G.P. Simon and Y.B. Cheng, J. Photochem. Photobiol. A: Chem, 2010, 213(1), 30–36.

[26] P.J. Holliman, A. Connell, M. Davis, M. Carnie, D. Bryant and E.W. Jones, Mater. Lett, 2019, 236, 289–291.

[27] F.-M. Raoult, C. R. Hebd. Séances Acad. Sci, 1887, 104, 1430–1433.
[31] L.N. Lewis, J.L. Spivack, S. Gasaway, E.D. Williams, J.Y. Gui, V. Manivannan and O.P. Siclovan, Sol. Energy Mater. Sol. Cells, 2006, 90(7–8), 1041–1051.

[32] Y.-S. Lin, M.-T. Chen, Y.-F. Lin, S.-J. Yang and J.-L. Lin, Appl. Surf. Sci, 2006, 252(16), 5892–5899.

[33] K.-H. Chung, M.M. Rahman, H.-S. Son and J.-J. Lee, Int. J. Photoenergy, 2012, 2012, 215802.
[34] J.-J. Lee, M.M. Rahman, S. Sarker, N.C. Deb Nath, A.J.S. Ahammad, J.K. Lee and B. Attaf(Ed), Composite materials for medicine and nanotechnology. Intech, Croatia, 2011. p.181.
[35] M.M. Rahman, N.C.D. Nath and J.-J. Lee, Isrl. J. Chem, 2015, 55(9), 990–1001.






