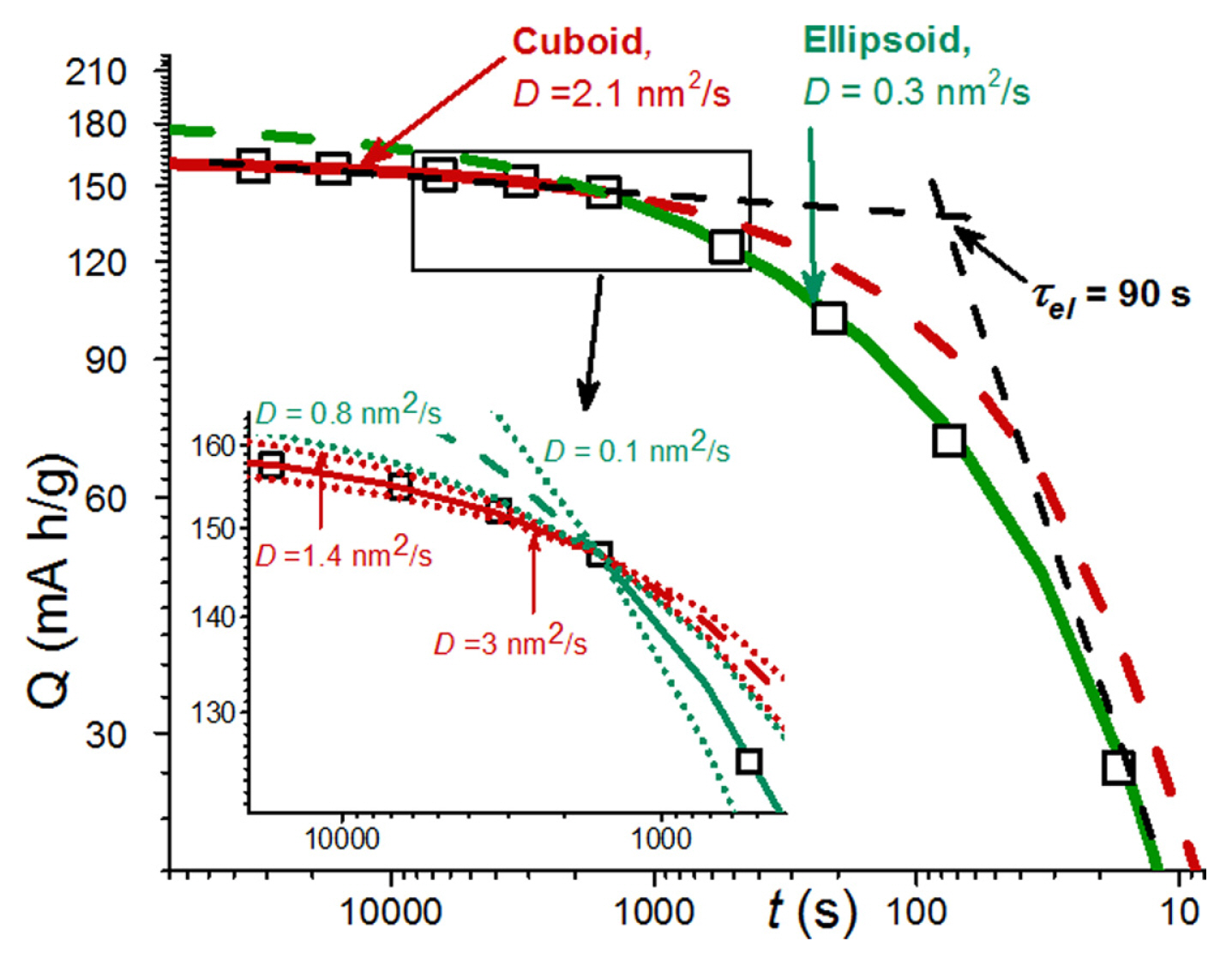
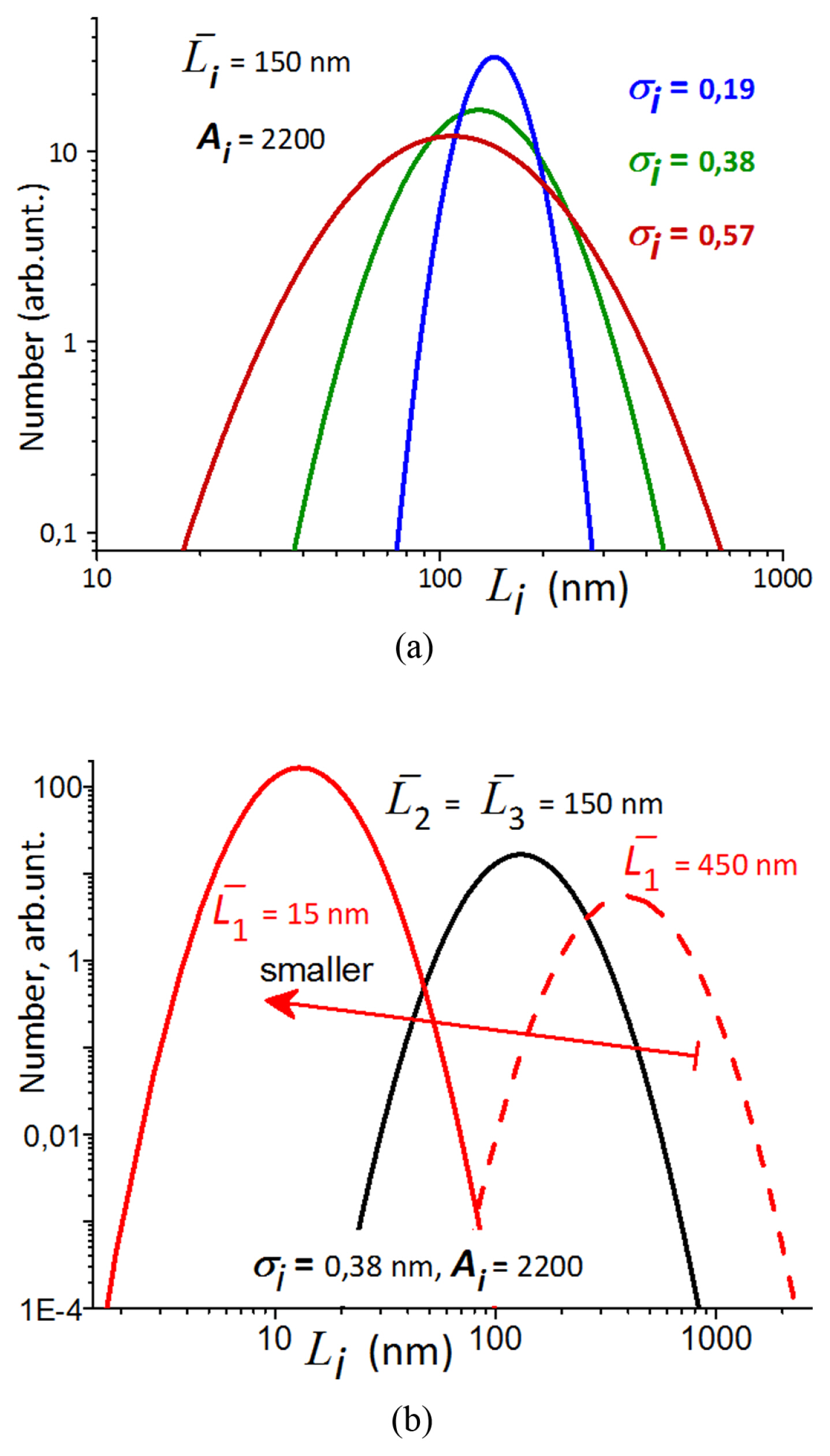
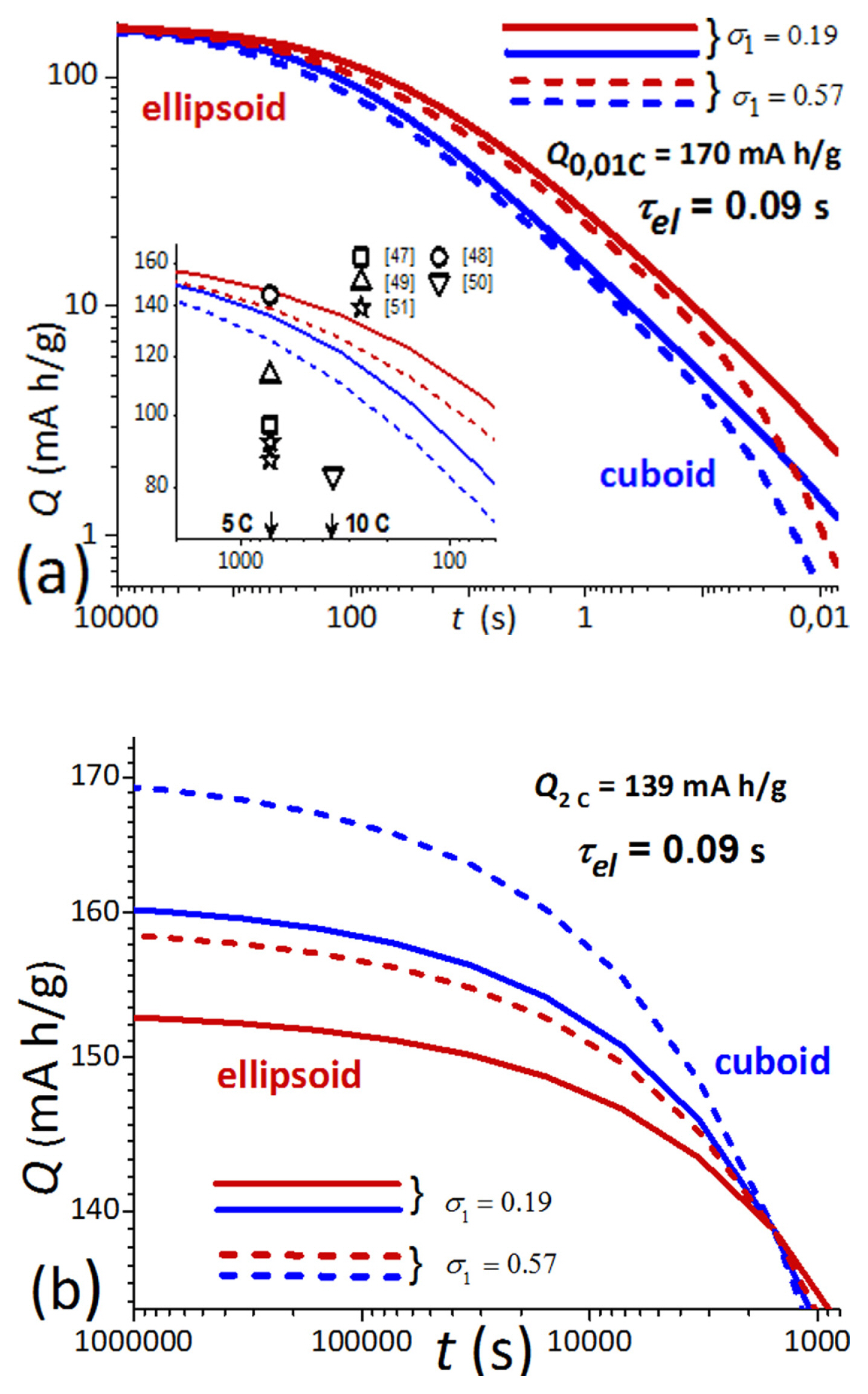
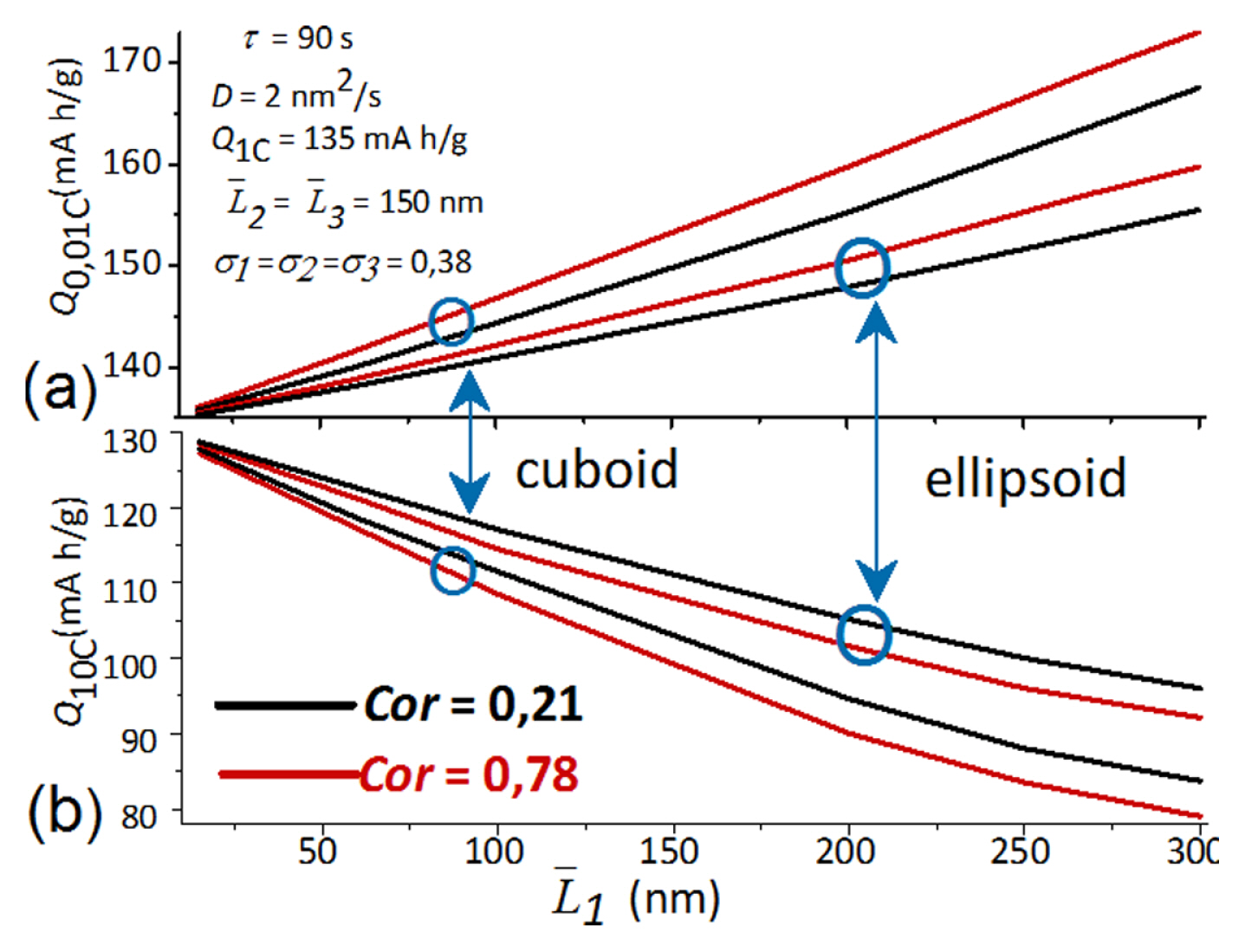
[1] W. Peukert, Elektrotechnische Zeitschrift, 1897, 20, 20ŌĆō21.
[2] N.F. Compagnone,
J. Power Sources,
1991,
35(
2), 97ŌĆō111.

[3] N.E. Galushkin, N.N. Yazvinskaya and D.N. Galushkin,
J. Electrochem. Soc,
2020,
167, 013535.


[4] D.H. Jeon, J.-H. Song, J.-P. Hong and S.H. Lee,
J. Ind. Eng. Chem,
2019,
76, 524ŌĆō531.

[5] J. Kim, S. Park, S. Hwang and W.-S. Yoon,
J. Electrochem. Sci. Technol,
2022,
13(
1), 19ŌĆō31.


[6] A.V. Ivanishchev, I.A. Ivanishcheva, S.-C. Nam and J. Mun,
Rus. J. Electrochem,
2021,
57(
7), 706ŌĆō720.


[7] F. Jiang and P. Peng, Sci. Rep, 2016, 6, 32639.
[8] R. Tian, S. Park, P. King, G. Cunningham, J. Coelho, V. Nicolosi and J. Coleman, Nat. Commun, 2019, 10, 1933.
[9] C. Heubner, M. Schneider and A. Michaelis,
Adv. Energy Mater,
2020,
10(
2), 1902523.


[10] J.-S. Lee, K. Heo, H.-S. Kim, M.-Y. Kim, J. Kim, S.-W. Kang and J. Lim,
J. Alloys Compd,
2019,
781, 553ŌĆō559.

[11] Z. Ahsan, B. Ding, Z. Cai, C. Wen, W. Yang, Y. Ma, S. Zhang, G. Song and M. Javed, J. Electrochem. En. Conv. Stor, 2021, 18(1), 010801.
[12] J. Choi, N. Voronina, Y. Sun and S. Myung,
Adv. Energy Mater,
2020,
10(
42), 2002027.


[13] I.-S. Seo, G.C. Hwang, J.-K. Kim and Y. Kim,
Electrochim. Acta,
2016,
193, 160ŌĆō165.

[14] X. Huang, Y. Yao, F. Liang and Y. Dai,
J. Alloys Compd,
2018,
743, 763ŌĆō772.

[15] Z. Ma, G. Shao, Y. Fan, G. Wang, J. Song and T. Liu,
ACS Appl. Mater. Interfaces,
2014,
6(
12), 9236ŌĆō9244.

[16] Y. Zhang, J.A. Alarco, J.Y. Nerkar, A.S. Best, G.A. Snook and P.C. Talbot,
J. Electrochem. Soc,
2019,
166(
16), A4128ŌĆōA4135.

[17] A. Bobyl and I. Kasatkin,
RSC Adv,
2021,
11, 13799ŌĆō13805.

[18] C.E. Kril and R. Birringer,
Philos. Mag. A,
1998,
77(
3), 621ŌĆō640.

[19] R.J. Matyi, L.H. Schwartz and J.B. Butt,
Catal. Rev. Sci. Eng,
1987,
29(
1), 41ŌĆō99.

[20] T.B. ┼Įuni─ć and J. Dohrup,
Powder Diffr,
1999,
14(
3), 203ŌĆō207.

[21] T. Lahrs,
Quality driven: PhostechŌĆÖs advanced LiFePO
4 cathode
. In: International power supply conference and exhibition; French Riviera. 2009; pp 1ŌĆō27.
[22] H. Al-Shammari and S. Farhad,
Energies,
2022,
15(
2), 410.

[23] K. Heo, J. Im, J.-S. Lee, J. Jo, S. Kim, J. Kim and J. Lim,
J. Electrochem. Sci. Technol,
2020,
11(
3), 282ŌĆō290.


[24] L. Wen, X. Wang, X. Liu, J. Sun, L. An, X. Ren, Z. Li, G. Liang and S. Jiang,
Ionics,
2019,
25, 5269ŌĆō5276.


[25] N.C. Popa,
J. Appl. Cryst,
1998,
31, 176ŌĆō180.

[26] N.C. Popa and G.A. Lungu,
J. Appl. Cryst,
2013,
46, 391ŌĆō395.

[27] L. Lutterotti, S. Matthies and H.R. Wenk, Newslett. CPD, 1999, 21, 14ŌĆō15.
[28] P. Boullay, L. Lutterotti, D. Chateignera and L. Sicard, Acta Cryst, 2014, A70, 448ŌĆō456.
[29] A. Nalbach-Leniewska,
Series Statistics,
1979,
10(
3), 381ŌĆō387.

[31] In: F Scholz editors. Electroanalytical methods: Guide to experiments and applications. Springer, Berlin, 2010.
[32] M.F. Triola, Elementary statistics technology update. 11th ed. Pearson Education, 2012.
[33] V.F. Lvovich, Distributed impedance models, Impedance spectroscopy: Applications to electrochemical and dielectric phenomena. John Wiley & Sons, 2012.
[34] K. Yang and M. Tang,
J. Mater. Chem. A,
2020,
8(
6), 3060ŌĆō3070.


[35] J. Baek, K. Kim and W. Shin,
J. Electrochem. Sci. Technol,
2018,
9(
2), 85ŌĆō92.


[36] H.W. Kim, P. Manikandan, Y.J. Lim, J.H. Kim, S.-C. Nam and Y. Kim,
J. Mater. Chem A,
2016,
4(
43), 17025ŌĆō17032.

[37] I.N. Beckman, Higher mathematics: The mathematical apparatus of diffusion. Yurayt Publishing House, Moscow, 2017.
[38] A.S. Hammood, Corrosion mechanism of cupronickel alloys. Lambert Academic Publishing, German, 2014.
[39] In: H.A Wiltsche, P Berghofer editors. Phenomenological approaches to physics. Springer, Cham, 2020.
[40] M. Tran, N. Ertugrul and T. Miller, World Appl. Sci. J, 2017, 35(8), 1561ŌĆō1567.
[41] M. Farag, M. Fleckenstein and S. Habibi,
J. Power Sources,
2017,
342, 351ŌĆō362.

[42] R. Malik, D. Burch, M. Bazant and G. Ceder,
Nano Lett,
2010,
10(
10), 4123ŌĆō4127.

[43] D.A. Cogswell and M.Z. Bazant,
Electrochem. Commun,
2018,
95, 33ŌĆō37.

[44] A.V. Churikov, A.V. Ivanishchev, A.V. Ushakov and V.O. Romanova,
J. Solid State Electrochem,
2014,
18, 1425ŌĆō1441.


[45] T. Kim, W. Choi1, H.-C. Shin, J.-Y. Choi, J.M. Kim, M.-S. Park and W.-S. Yoon,
J. Electrochem. Sci. Technol,
2020,
11(
1), 14ŌĆō25.


[46] J. Li, B.L. Armstrong, J. Kiggans, C. Daniel and D.L. Wood,
J. Electrochem. Soc,
2013,
160(
2), A201.

[47] S. Ju, H. Peng, G. Li and K. Chen,
Mater. Lett,
2012,
74, 22ŌĆō25.

[48] M. Chen, Q. Ma, C. Wang, X. Sun, L. Wang and C. Zhang,
J. Power Sources,
2014,
263, 268ŌĆō275.

[49] F. Wang, Z. Fang and Y. Zhang,
J. Electroanal. Chem,
2016,
775, 110ŌĆō115.

[50] R.R. Kapaev, S.A. Novikova, T.L. Kulova, A.M. Skundin and A.B. Yaroslavtsev,
Nanotechnol. Russia,
2016,
11, 757ŌĆō760.


[51] J. Seher and M. Fr├Čba,
ACS Omega,
2021,
6, 24062ŌĆō24069.