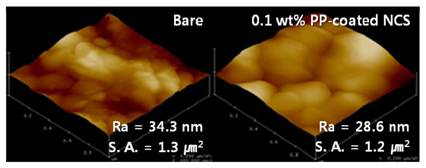
AFM images of bare and 0.1 wt% PP-coated NCS particles are shown in
Fig. 1. From the 0.1 wt% PP-coated NCS particles, the average roughness was reduced from 34.3 nm to 28.6 nm. This means that the 0.1 wt% PP-coated NCS particles had a lower surface area than the NCS particle. Low surface area of the primary particles and indirect contact area between cathode and electrolyte is effective for reducing the contact area with the electrolyte. It will therefore be possible to reduce the chemical side reactions with the electrolyte. The chemical side reactions are among the unnecessary reactions. It is known that commercial LiPF
6-based lithium battery electrolytes always contain a trace amount of water, and it has been established that LiPF
6 can react with this water and produce a very reactive HF species. The surface of the cathode material is attacked by HF species from the electrolyte. The reaction causes elution of transition metal elution in the cycling process
[9]. However, the PP coating reduces the contact with the electrolyte, and improvement of electrochemical properties is thus expected. In order to confirm the existence of the coated layer,
Fig. 2 shows a TEM image of a 0.1 wt% PP-coated NCS particle. The edge of the 0.1 wt% PP-coated NCS particle is covered with a thin layer (average thickness = 8.43 nm) with uniform dispersion. The selected area electron diffraction (SAED) patterns were obtained from the PP-coated layer and the NCS cathode material. The SAED pattern corresponding to the PP-coated layer (right side) exhibited a hollow ring pattern without bright spots, thus indicating that the coated layer consisted of disordered polymer materials. The image in the right inset shows some bright spots in the SAED pattern, which is typical of crystalline NCS cathode material
[2,
8]. These results thus provide conclusive evidence that the NCS particles are uniformly coated by the nano-sized polymer layer.
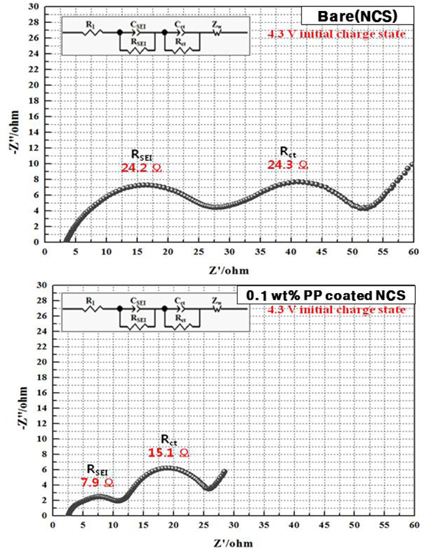
Fig. 3 shows Nyquist plots of the bare and 0.1 wt% PP-coated NCS electrode in the charged state of 4.3 V. The high-frequency semicircle is attributed to the resistance of the solid electrolyte interphase (SEI) film (R
SEI), and the second semicircle that appears at lower frequency is associated with the charge transfer resistance (R
ct)
[10,
11]. The SEI film is formed by contact between the oxygen generated in the charging process and the electrolyte. The reaction of generated oxygen with the electrolyte forms SEI film such as Li
2CO
3, ROLi, or ROCO
2Li on the surface of the cathode material. The SEI film reduces the electrochemical properties that prevent lithium diffusion. The PP-coated layer present on the cathode material surface effectively reduces the contact between the oxygen and the electrolyte. The R
SEI value of the 0.1 wt% PP-coated NCS electrode is smaller than that of the bare NCS electrode. With the reduced R
SEI, the R
ct of the 0.1 wt% PP-coated NCS electrode is lowered from 24.3 Ω to 15.1 Ω.
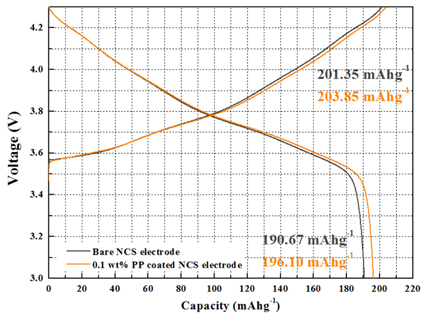
Fig. 4 presents the initial charge and discharge curves of the cells with bare NCS and 0.1 wt% PP-coated NCS electrodes in a voltage range of 3.0 to 4.3 V at a constant current rate of 17 mA/g. The bare NCS cathode material coated with PP delivered a slightly higher discharge capacity (196.10 mAhg
−1) than the bare NCS cathode material (190.67 mAh/g
−1), and this is attributed to the effect of SEI film. In the case of the 0.1 wt% PP-coated NCS electrode, the R
ct of the R
SEI reduction decreases after the initial charge process. Therefore, the initial discharge capacity increase relative to the bare NCS electrode, and thus, when calculating initial efficiency based on the results, initial coulomb efficiency increases from 94.7 % to 96.2 %.
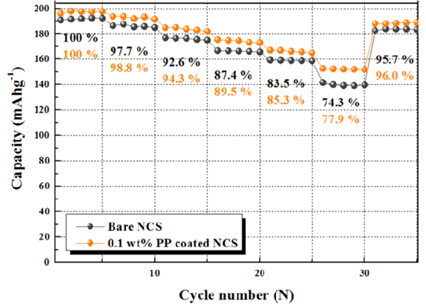
Fig. 5 shows the cycle performance of bare NCS and 0.1 wt% PP-coated NCS electrodes at a current density of 17 mA/g between 3.0 and 4.3 V. The initial discharge capacity of the bare NCS electrode decreased from 190.67 mAh/g to 171.22 mAh/g after 40 cycles. (40
th cycle efficiency = 89.8 %). The 0.1 wt% PP-coated NCS electrode delivered an initial discharge capacity of 196.10 mAh/g, which was reduced to 182.77 mAh/g after 40
th cycle. (40
th cycle efficiency = 93.2 %). The cycle efficiency is a result of suppressing the SEI film and HF attack, and the suppressed SEI film reduces the R
ct. Furthermore, the insertion and extraction of Li
+ ions proceeds smoothly in the charge and discharge process. The contact area undergoing HF attack is reduced. Therefore, it is possible to improve structural instability by reducing transition metals elution
[12,
13].
Fig. 6 shows the electrochemical properties of various current densities of the bare NCS and 0.1 wt% PP-coated NCS electrode. The PP-coated electrode exhibited a higher discharge capacity at all the rates compared with that of the bare electrode, further enhancing the rate capability. Polypyrole has been known as an inherent electrical conductive polymer due to the conjugation of the single and double bonds alternating within macromolecular architecture
[14]. The extra electrons of the double bond in a conjugated system are free to move or roam through the polymer chain. Therefore, the superior rate performance of PP-coated electrode is obtained with the high electronic conductivity of polypyrole layer although reducing surface area. These results indicated an easier lithium intercalation/deintercalation owing to reduced charge transfer resistance
[14,
15].
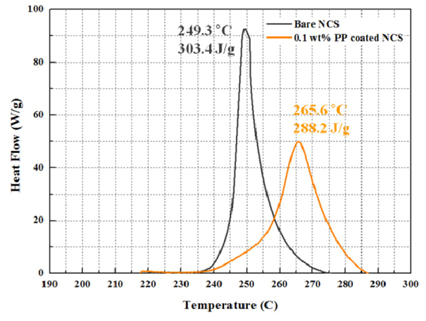
Fig. 7 shows DSC curve of bare NCS and 0.1 wt% PP-coated NCS cathode materials in the charged state of 4.3 V. The bare NCS material had a large exothermic peak at 249.3 ℃ and the reaction released heat of 303.4 J/g. However, for the 0.1 wt% PP-coated NCS material, the reaction took place at a higher temperature of 265.6 ℃ with reaction released heat of 288.2 J/g. This indicates that the PP coating layer with high surface coverage effectively prevents the NCS from being directly exposed to the liquid electrolyte, which in turn alleviates the exothermic reaction between the delithiated NCS and liquid electrolyte. The similar DSC result of PATEP coated NCM cathode was published
[19].