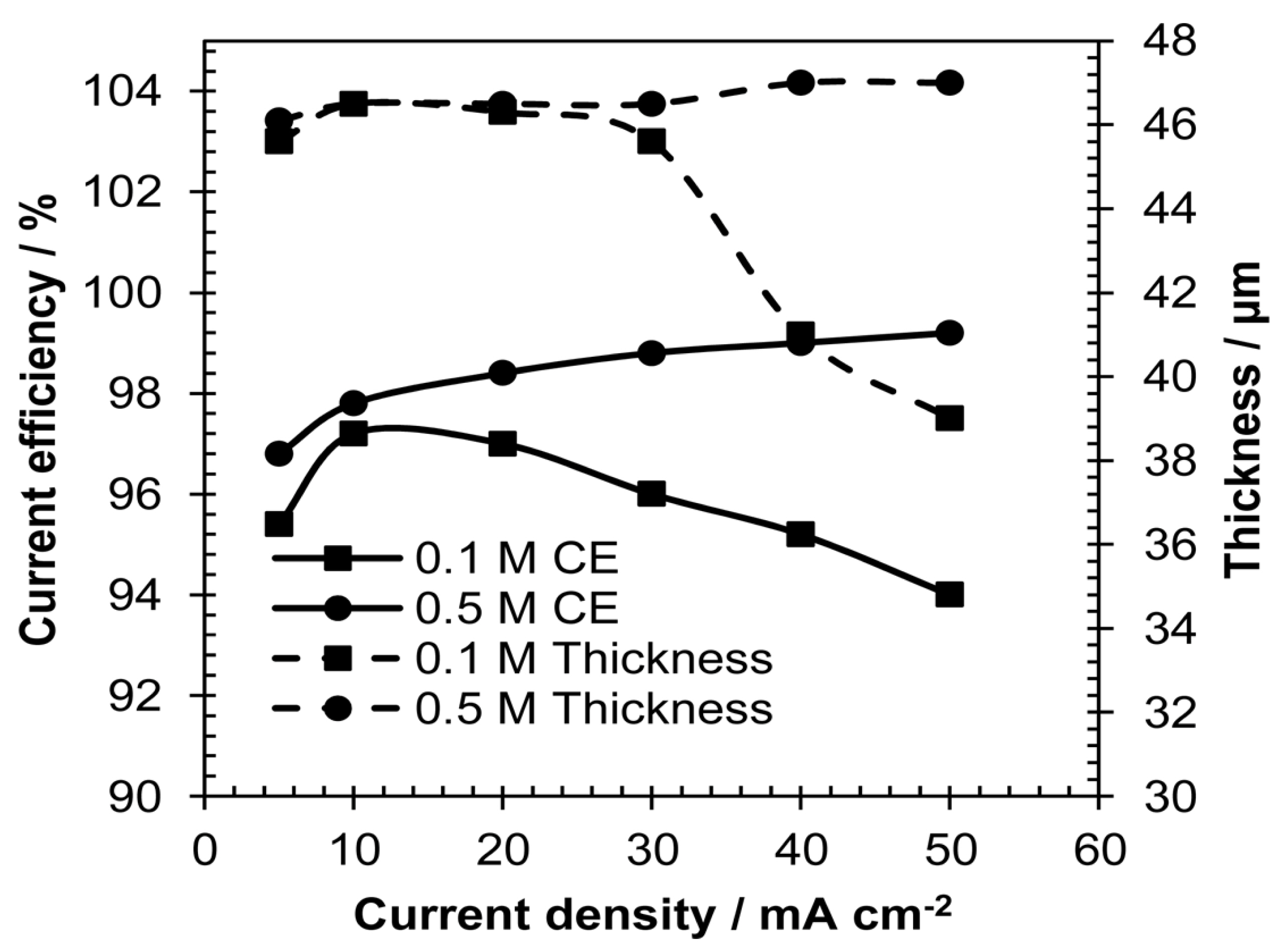
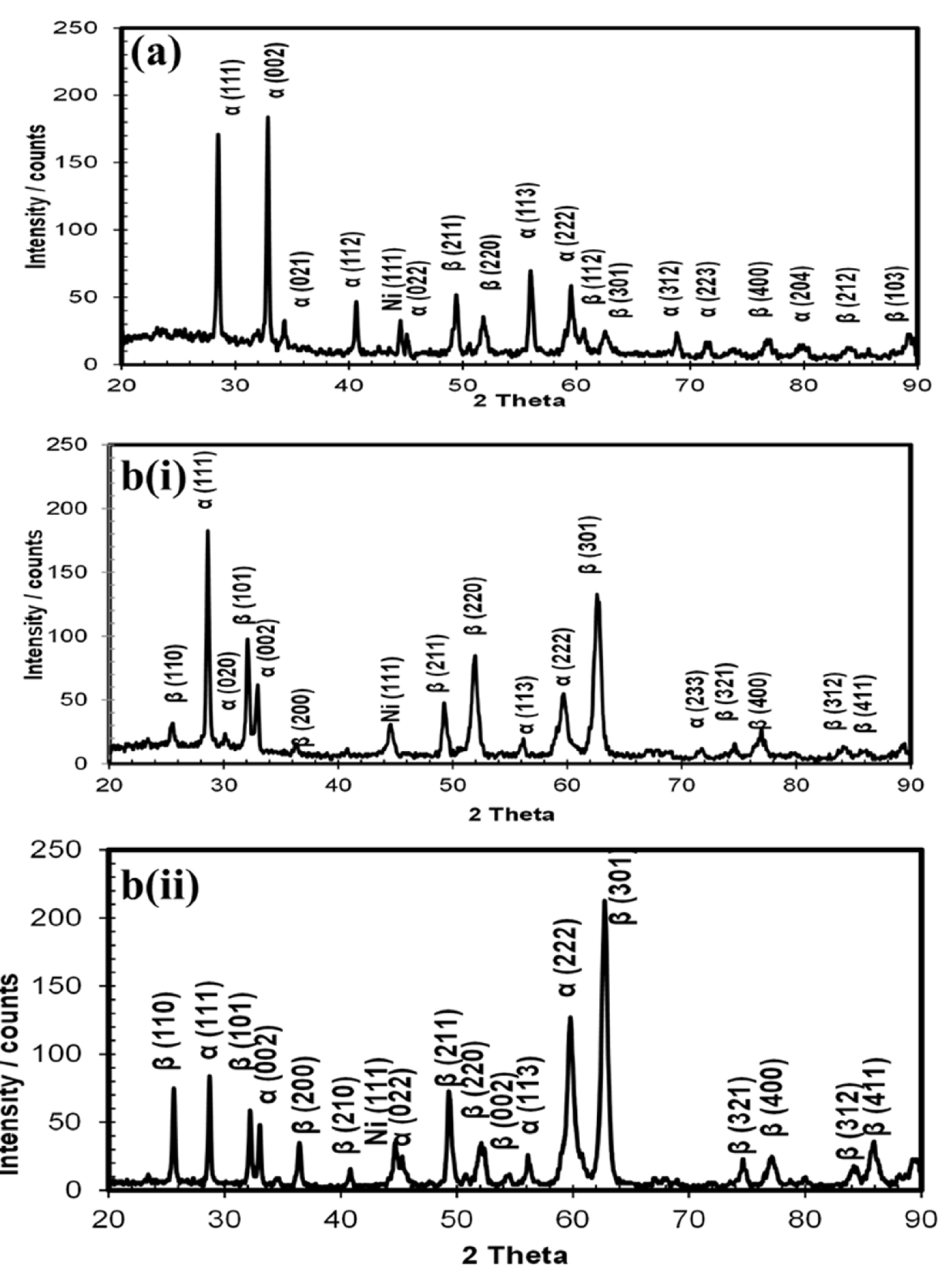
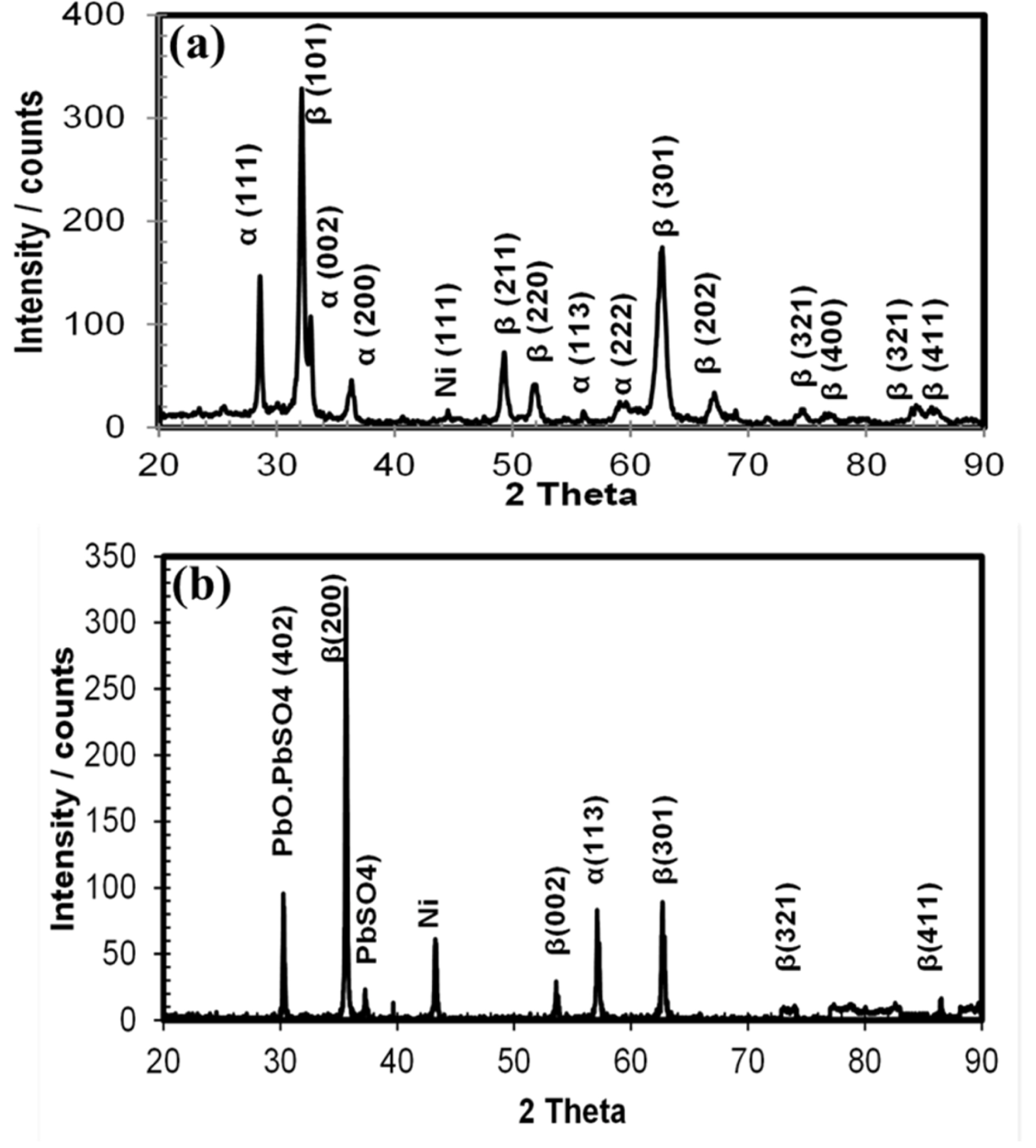
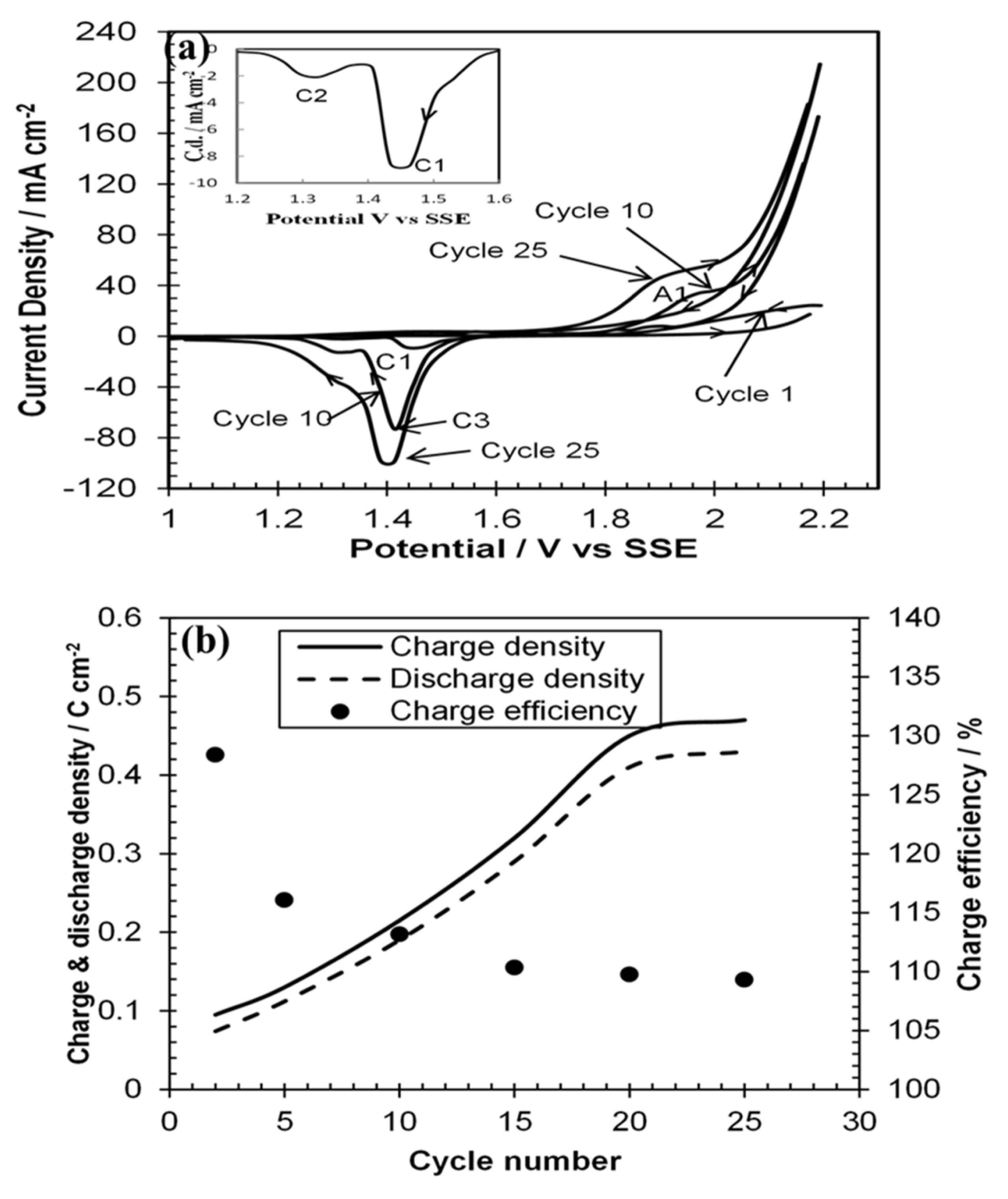
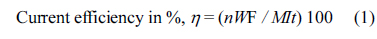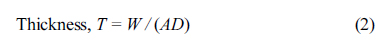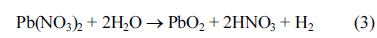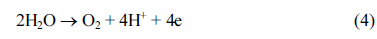Cyclic voltammetric investigations were conducted on the deposited PbO
2 in 4.7 M H
2SO
4 solution (lead-acid battery environment) between 1000 to 2200 mV at 30 mV S
-1 scan rate. All potentials were measured with respect to SSE. For voltammograms shown in
Fig. 3 deposition was made from 0.1 M Pb(NO
3)
2 solution for 20 minutes at 50 mA cm
-2 current density. In
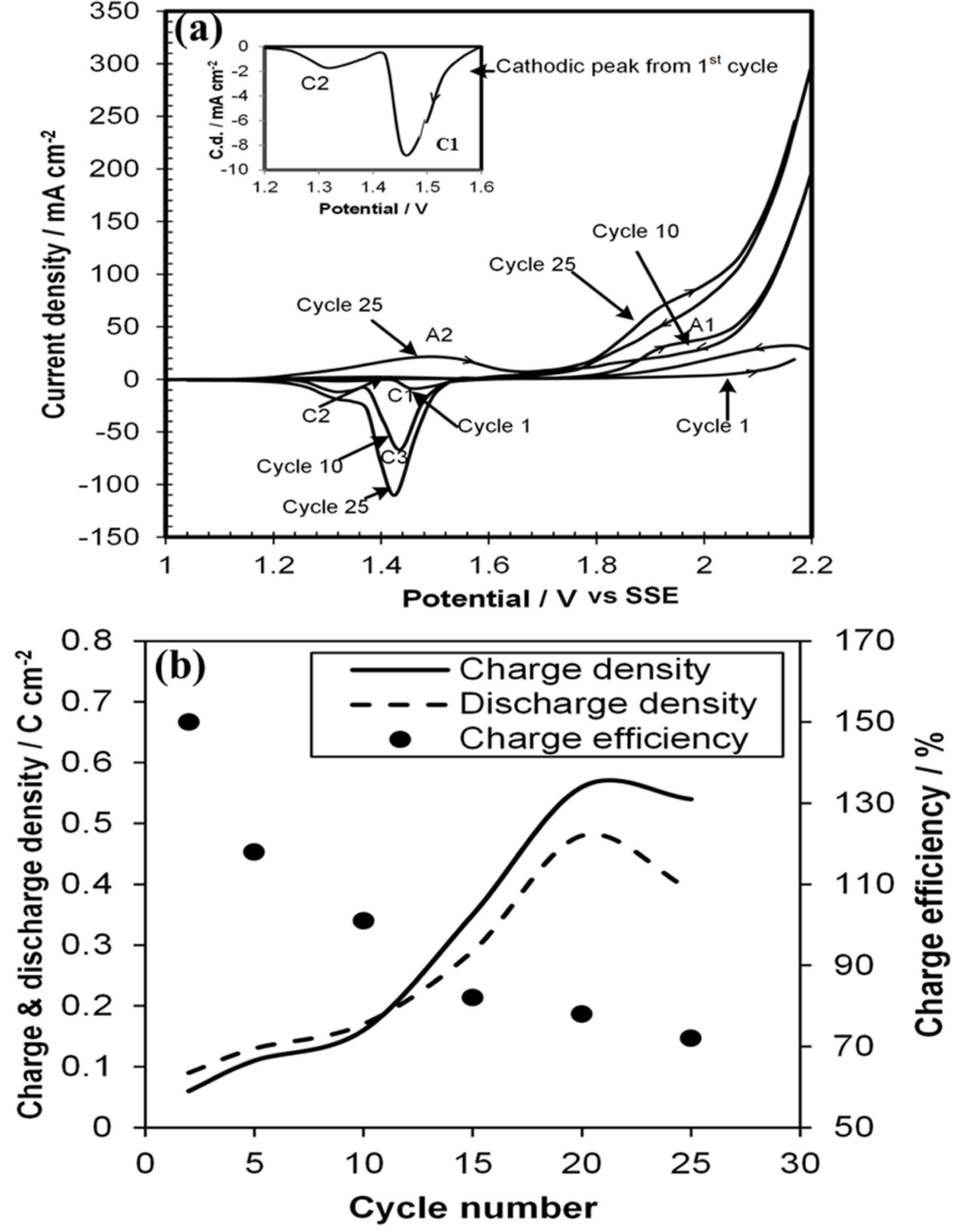
Fig. 3a, the first cycle for the three voltammograms (1
st, 10
th and 25
th cycles) started anodically at 1000 mV shows a small anodic current rise after approximately 1950 mV, and two cathodic peaks C
1 and C
2 (inset) at1460 mV and 1320 mV respectively. It is known
[27] that between two polymorphic forms reduction of orthorhombic α-PbO
2 to PbSO
4 takes place at more positive potential than that for the reduction of tetragonal β-PbO
2. So, peak C
1 and C
2 are due to reduction of α-PbO
2 and β-PbO
2 respectively. Normally α-PbO
2 deposits from the basic solution and β-PbO
2 deposits from the acidic solution as reported in the literature
[19]. In fact, 0.1 M HNO
3 is weak acidic solution. This may be reason for both α-PbO
2 and β-PbO
2 deposition in this acidic environment. The 10
th cycle shows an anodic peak at around 1940 mV which merged with steep current rise after 2080 mV. It has been reported
[28,
34] that the oxidation of PbSO
4 to PbO
2 on Ni surface takes place at around 1800 mV, and oxygen evolution on PbO
2 takes place at further positive potential. Thus, this peak was due to PbO
2 formation and subsequent current rise was due to oxygen evolution. During cathodic scan a new big peak C
3 (α+β-PbO
2) appeared at more negative potential (1450 mV) than C
1 with a shoulder of C
2 peak. This peak was due to gradual shifting of C
1 peak potential with the increase of cycle number i.e., α-PbO
2 gradually converted to β-PbO
2 which increases oxygen evolution properties from the start of 2
nd cycle. All peak current densities and currents are several times bigger than that of the first cycle. The anodic scan of the 25
th cycle shows a broad and shallow peak A
2 at around 1450 mV for oxidation of substrate nickel to ultimate NiO(OH) formation as reported elsewhere
[28,
35,
36]. The cathodic scan shows shifting of C
3 peak potential to more negative value. All current rises are higher than those of 10
th cycle. Increase of oxygen evolution current and other currents suggest formation of porous PbO
2 due to repeated PbO
2-PbSO
4-PbO
2 reactions. Noticeable nickel oxidation peak current indicates extension of pores up to Ni surface or falling apart part of PbO
2 film from surface. The influence of cycle number on charge (PbSO
4 → PbO
2) and discharge (PbO
2 → PbSO
4) densities and charge efficiency are shown in
Fig. 3b. The charge and discharge densities was obtained by calculating the area under the anodic and cathodic peaks in
Fig. 3a divided by scan rate respectively. However, the charge efficiency was calculated from the ratio of discharge and charge current densities. This figure shows charge and discharge densities increased up to 20 cycles and then decreased. The decrease was due to parting away of PbO
2 from the Ni surface. Initially, charge efficiency over was 140% which steadily decreased with the increase of cycle number. This indicates initially discharge density of PbO
2 film was greater than charge density. This is similar result with literature
[21] as they also obtained charge efficiency more than 100% for initial few cycles. This was due to increase of PbSO
4 formation with cycle number, which was difficult for conversion to PbO
2 due to non-conductivity. Densities of α-PbO
2, β-PbO
2 and PbSO
4 are 9.87 g cm
-3, 9.30 g cm
-3 and 6.29 g cm
-3 respectively
[37]. Conversion of PbO
2 to PbSO
4 increased volume and produced internal stress for expansion. When PbSO
4 was transformed to PbO
2 volume contraction produced porosity resulting more surface area for charge-discharge as well as oxygen evolution reactions.
Cyclic voltammetric results for PbO
2 electrodeposition from 0.5 M Pb(NO
3)
2 are shown in
Fig. 4. The ratio of C
1 and C
2 height is 3.7 as obtained from
Fig. 4a (inset of the 1
st cycle) which is 5.9 for electrodeposition from 0.1 M Pb(NO
3)
2 solution. Which suggests that more α-PbO
2 (approximately 85%) formed when deposition was made from 0.1 M Pb(NO
3)
2 solution compared to that (78%) for deposition from 0.5 M Pb(NO
3)
2 solution under identical deposition conditions. Density of α-PbO
2 is higher than that of β-PbO
2. This is one of the reasons for obtaining less thick deposit film from 0.1 M Pb(NO
3)
2 solution. However, all anodic and cathodic peak current heights are smaller in this case. Moreover, the 25
th cycle does not show Ni oxidation currents, and the potential for C
3 is more negative than that electrodeposition from electrolyte containing 0.1 M Pb(NO
3)
2. The charge efficiency attained a constant value (~112%) after 10
th cycle in
Fig. 4b. This revealed that electrodeposited of PbO
2 from 0.5 M Pb(NO
3)
2 solution has better charge efficiency cycle number behavior. The oxygen evolution current density i.e., currents after 2000 mV (not shown) was always lower for deposition from 0.5 M Pb(NO
3)
2 solution. All of these suggest the presence of more β phase in the mixture of (α+β)-PbO
2, and more compact PbO
2 deposition from 0.5 M Pb(NO
3)
2 solution compared to that deposited from 0.1 M solution. Although β-PbO
2 favors oxygen evolution reaction, nonetheless due to more compact film formation for deposition from 0.5 M Pb(NO
3)
2 solution less surface was exposed for oxygen evolution and charge-discharge reactions. This compact film was comparatively more adherent as evident from the absence of Ni oxidation peak.